Advanced Copper-Catalyzed Synthesis of 3-Methylthioindole Derivatives for Commercial Scale-up
Advanced Copper-Catalyzed Synthesis of 3-Methylthioindole Derivatives for Commercial Scale-up
The pharmaceutical and fine chemical industries are constantly seeking robust methodologies for constructing biologically active heterocyclic scaffolds, particularly 3-substituted indole derivatives which serve as critical intermediates in drug discovery. Patent CN109942478B discloses a groundbreaking synthetic approach that utilizes dimethyl sulfoxide (DMSO) as a methylthio reagent in the presence of a copper catalyst, offering a transformative alternative to traditional sulfenylation protocols. This technology addresses long-standing challenges regarding reagent toxicity, odor, and waste generation, positioning it as a highly viable route for the industrial production of high-purity pharmaceutical intermediates. By leveraging the dual functionality of DMSO as both a sulfur source and an oxidant, the process achieves remarkable atom economy and operational simplicity. For procurement and supply chain leaders, this innovation represents a significant opportunity to optimize manufacturing costs while adhering to increasingly stringent environmental regulations. The following analysis details the technical merits and commercial implications of adopting this copper-catalyzed C-H activation strategy for large-scale applications.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the introduction of methylthio groups onto the indole scaffold has relied heavily on the use of volatile and malodorous thiols or disulfides, which pose severe safety and environmental hazards in a manufacturing setting. As illustrated in prior art such as the Selectfluor-mediated reaction shown below, these conventional pathways often require stoichiometric amounts of expensive oxidants and generate substantial quantities of sulfur-containing waste streams that are difficult to treat. 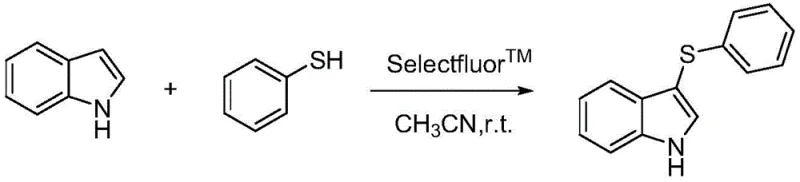 The reliance on such reagents not only complicates the engineering controls required for worker safety but also drastically increases the cost of waste disposal and regulatory compliance. Furthermore, many existing metal-catalyzed methods necessitate harsh reaction conditions, including extremely high temperatures or the use of corrosive halogenated oxidants, which can degrade sensitive functional groups on the indole ring. The inability to recycle excess oxidants and solvents in these traditional processes further exacerbates the environmental footprint, making them less attractive for green chemistry initiatives. Consequently, there is a pressing industrial demand for a cleaner, more efficient sulfenylation protocol that eliminates the need for noxious sulfur sources.
The reliance on such reagents not only complicates the engineering controls required for worker safety but also drastically increases the cost of waste disposal and regulatory compliance. Furthermore, many existing metal-catalyzed methods necessitate harsh reaction conditions, including extremely high temperatures or the use of corrosive halogenated oxidants, which can degrade sensitive functional groups on the indole ring. The inability to recycle excess oxidants and solvents in these traditional processes further exacerbates the environmental footprint, making them less attractive for green chemistry initiatives. Consequently, there is a pressing industrial demand for a cleaner, more efficient sulfenylation protocol that eliminates the need for noxious sulfur sources.
The Novel Approach
The methodology described in CN109942478B revolutionizes this transformation by employing dimethyl sulfoxide (DMSO) as a benign and economically accessible methylthio source, effectively bypassing the need for hazardous thiols. As depicted in the general reaction scheme below, the process utilizes copper acetate as a catalyst and diethyl phosphite as a crucial additive to facilitate the direct C-H activation of indole derivatives in toluene. 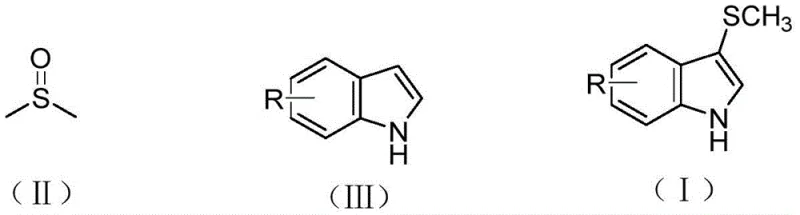 This novel approach operates at a moderate temperature of 110°C and achieves exceptional conversion rates, with specific examples demonstrating isolated yields reaching up to 93% for unsubstituted indole. The elimination of external oxidants is a key feature, as the DMSO molecule itself participates in the redox cycle, thereby simplifying the reagent profile and reducing the overall material cost. Additionally, the reaction exhibits excellent regioselectivity for the 3-position of the indole ring, minimizing the formation of unwanted isomers and streamlining the downstream purification process. This combination of high efficiency, safety, and selectivity makes the technology ideally suited for the commercial scale-up of complex pharmaceutical intermediates.
This novel approach operates at a moderate temperature of 110°C and achieves exceptional conversion rates, with specific examples demonstrating isolated yields reaching up to 93% for unsubstituted indole. The elimination of external oxidants is a key feature, as the DMSO molecule itself participates in the redox cycle, thereby simplifying the reagent profile and reducing the overall material cost. Additionally, the reaction exhibits excellent regioselectivity for the 3-position of the indole ring, minimizing the formation of unwanted isomers and streamlining the downstream purification process. This combination of high efficiency, safety, and selectivity makes the technology ideally suited for the commercial scale-up of complex pharmaceutical intermediates.
Mechanistic Insights into Copper-Catalyzed C-H Activation
The catalytic cycle likely initiates with the coordination of the copper(II) species to the indole substrate, facilitating the activation of the electron-rich C3-H bond through a concerted metalation-deprotonation or radical pathway. The presence of diethyl phosphite plays a pivotal role in this mechanism, potentially acting as a ligand or a radical initiator that assists in the homolytic cleavage of the C-S bond in DMSO to generate the active methylthio radical species. This radical intermediate subsequently attacks the activated indole ring, forming the new carbon-sulfur bond with high fidelity. The copper catalyst is then regenerated through a redox process involving the sulfoxide moiety, ensuring that the catalytic turnover is sustained throughout the 18-hour reaction period without the accumulation of inactive metal species. Understanding this mechanistic nuance is critical for R&D directors aiming to adapt this protocol to novel substrates, as the electronic properties of the indole ring can influence the rate of C-H activation.
From an impurity control perspective, the specificity of this copper-catalyzed system ensures that side reactions such as over-oxidation or polymerization of the indole core are kept to a minimum. The use of toluene as a solvent provides a non-polar environment that favors the solubility of the organic intermediates while allowing for easy removal during the workup phase. The reaction conditions are sufficiently mild to preserve sensitive functional groups such as nitriles, chlorides, and esters, as evidenced by the successful synthesis of diverse derivatives like 6-chloro-3-methylthioindole and indole-5-carbonitrile analogs. This robustness against functional group incompatibility is a major advantage for multi-step synthesis campaigns where intermediate stability is paramount. Furthermore, the absence of halogenated oxidants prevents the formation of halogenated organic byproducts, resulting in a cleaner crude reaction mixture that requires less aggressive purification techniques.
How to Synthesize 3-Methylthioindole Efficiently
Implementing this synthesis route requires precise control over stoichiometry and thermal parameters to maximize the yield and purity of the final product. The patent specifies a molar ratio of 3:1 for DMSO to indole derivative, ensuring that the sulfur source is in sufficient excess to drive the equilibrium forward without creating unmanageable waste volumes. Copper acetate is employed in a 1:1 molar ratio relative to the substrate, which, while stoichiometric in some contexts, acts effectively to promote the transformation under the specified thermal conditions. Diethyl phosphite is added in a 2:1 ratio, serving as an essential promoter for the reaction kinetics. The detailed standardized synthesis steps, including specific workup procedures and chromatographic conditions, are outlined in the guide below.
- Combine indole derivatives, dimethyl sulfoxide (3: 1 molar ratio), copper acetate (1:1 molar ratio), and diethyl phosphite (2:1 molar ratio) in excess toluene solvent.
- Heat the reaction mixture to 110°C and maintain stirring for 18 hours under appropriate atmospheric conditions to ensure complete conversion.
- Dilute the reaction liquid with ethyl acetate, filter, remove solvent via reduced pressure distillation, and purify the crude product using column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this DMSO-based methodology offers tangible benefits in terms of cost reduction and supply chain resilience. By replacing expensive and hazardous thiols with commodity-grade DMSO, the raw material costs are significantly lowered, and the logistical complexities associated with handling dangerous goods are virtually eliminated. The simplified reaction profile reduces the dependency on specialized equipment for odor containment and corrosion resistance, allowing for production in standard glass-lined or stainless steel reactors. This flexibility enhances the agility of the supply chain, enabling faster response times to market demands for key pharmaceutical building blocks. Moreover, the high conversion rates reported in the patent minimize the loss of valuable starting materials, directly contributing to improved overall process economics.
- Cost Reduction in Manufacturing: The substitution of costly oxidants and smelly thiols with inexpensive DMSO and copper acetate leads to a drastic reduction in the bill of materials for each batch. Since the reaction does not require external oxidants like persulfates or Selectfluor, the expenditure on auxiliary reagents is minimized, and the waste treatment costs are substantially decreased due to the lower toxicity of the effluent. The high yields achieved, often exceeding 80% and reaching up to 93%, mean that less raw material is wasted, further driving down the cost per kilogram of the active intermediate. This economic efficiency is compounded by the ability to recover and recycle the toluene solvent, adding another layer of cost savings to the overall manufacturing process.
- Enhanced Supply Chain Reliability: Sourcing stable and non-hazardous reagents like DMSO and copper salts is far more reliable than managing the supply of volatile thiols, which often face strict transportation regulations and availability fluctuations. The robustness of the reaction conditions ensures consistent batch-to-batch quality, reducing the risk of production delays caused by failed runs or out-of-specification products. This reliability is crucial for maintaining continuous supply to downstream API manufacturers who depend on timely deliveries of high-quality intermediates. Additionally, the simplicity of the purification process reduces the lead time for releasing batches, allowing for a more responsive and agile supply chain operation.
- Scalability and Environmental Compliance: The process is inherently scalable, as demonstrated by the use of common solvents and standard heating protocols that translate easily from laboratory to pilot and commercial scales. The avoidance of heavy metal catalysts like palladium or platinum, and the use of relatively benign copper salts, simplifies the regulatory approval process for the final drug substance by reducing residual metal concerns. The green chemistry attributes of this method, specifically the reduction of hazardous waste and the use of safer solvents, align perfectly with global sustainability goals and corporate environmental mandates. This compliance advantage mitigates regulatory risks and enhances the corporate social responsibility profile of the manufacturing entity.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this copper-catalyzed sulfenylation technology. These answers are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity on process capabilities and limitations. Understanding these details is essential for technical teams evaluating the feasibility of integrating this route into their existing manufacturing portfolios. The responses highlight the specific advantages of the DMSO/copper system over legacy methods.
Q: Why is dimethyl sulfoxide preferred over thiols for this synthesis?
A: Dimethyl sulfoxide (DMSO) serves as both the sulfur source and the oxidant, eliminating the need for hazardous, foul-smelling thiols and external oxidants, thereby significantly improving operational safety and environmental compliance.
Q: What represents the key advantage of the copper acetate catalyst system?
A: The copper acetate catalyst facilitates efficient C-H activation at the 3-position of the indole ring under relatively mild thermal conditions (110°C), achieving high conversion rates without requiring expensive noble metals or extreme pressures.
Q: How does this method impact downstream purification costs?
A: By avoiding the formation of complex sulfur-based byproducts common in thiol-based routes and utilizing a simple toluene solvent system, the workup procedure is streamlined, reducing the burden on purification infrastructure and lowering overall processing time.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Methylthioindole Supplier
At NINGBO INNO PHARMCHEM, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that complex synthetic routes like this copper-catalyzed indole functionalization are executed with precision and efficiency. Our rigorous QC labs and commitment to stringent purity specifications guarantee that every batch of 3-methylthioindole derivatives meets the exacting standards required by the global pharmaceutical industry. We understand the critical nature of supply continuity and have optimized our operations to deliver high-purity intermediates with consistent quality profiles. Our technical team is well-versed in the nuances of C-H activation chemistry and can provide expert support for custom synthesis requirements.
We invite potential partners to contact our technical procurement team to request specific COA data and route feasibility assessments tailored to your project needs. By collaborating with us, you can leverage our expertise to achieve a Customized Cost-Saving Analysis that identifies opportunities for optimizing your supply chain and reducing overall manufacturing expenses. Let us help you secure a reliable supply of these vital building blocks for your next-generation therapeutics.
