Revolutionizing Pharmaceutical Intermediate Synthesis Through Scalable Carbene Insertion Chemistry for High-Purity Heterocyclic Derivatives
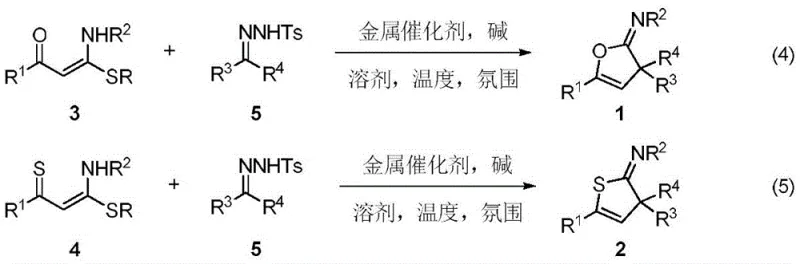
Patent CN108148021B introduces a transformative one-step synthesis methodology for producing structurally diverse 2-imine (3H) polysubstituted furan and thiophene derivatives through copper-catalyzed cyclization and carbene insertion chemistry. This innovation addresses longstanding challenges in heterocyclic compound manufacturing by utilizing readily accessible starting materials—specifically, 1-alkylthio-1-amino-1-en-3-ketones or thiones combined with p-toluenesulfonylhydrazones—under precisely controlled alkaline heating conditions that eliminate multi-step synthetic sequences. The process achieves remarkable efficiency through simultaneous C-O bond formation and dual C-C bond construction within a single reaction vessel, significantly reducing operational complexity while maintaining exceptional stereoselectivity across diverse substrate combinations. This breakthrough directly responds to industry demands for streamlined production of bioactive heterocycles used in pharmaceutical development pipelines, offering a robust platform for generating complex molecular architectures with minimal purification requirements. The methodology has been rigorously validated through multiple experimental examples demonstrating consistent yields between 44% and 82% under optimized parameters while preserving critical functional group integrity essential for downstream applications.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic routes for imine furan derivatives typically involve multi-step sequences requiring harsh reaction conditions that compromise both yield and purity profiles. Conventional approaches often necessitate cryogenic temperatures or strong acidic/basic environments that generate difficult-to-remove impurities while limiting functional group compatibility across diverse molecular scaffolds. These processes frequently suffer from poor stereoselectivity due to uncontrolled reaction pathways, resulting in complex mixtures that require extensive chromatographic purification—significantly increasing production costs and extending manufacturing timelines. Furthermore, existing methodologies demonstrate limited scalability due to exothermic reaction profiles and safety concerns associated with hazardous reagents or unstable intermediates. The cumulative effect of these limitations manifests as inconsistent product quality that fails to meet stringent regulatory requirements for pharmaceutical intermediates while simultaneously creating supply chain vulnerabilities through unreliable production throughput.
The Novel Approach
The patented methodology overcomes these constraints through an elegant one-step cyclization and carbene insertion process that operates under remarkably mild conditions between 80°C and 140°C using copper salt catalysts in aprotic solvents like toluene. By leveraging p-toluenesulfonylhydrazones as stable carbene precursors under nitrogen atmosphere with tertiary alkoxide bases, this approach achieves precise control over reaction kinetics while eliminating hazardous reagents typically required in traditional syntheses. The process demonstrates exceptional functional group tolerance across diverse R-group substitutions including aryl, heteroaryl, and alkyl moieties without requiring protective group strategies that complicate conventional routes. Critically, the methodology maintains high stereoselectivity through well-defined transition states that minimize side reactions while producing cleaner reaction profiles amenable to straightforward purification protocols. This innovation directly addresses industry pain points by enabling consistent production of high-purity intermediates suitable for pharmaceutical applications while significantly reducing both operational complexity and environmental impact compared to legacy manufacturing approaches.
Mechanistic Insights into Copper-Catalyzed Carbene Insertion
The core innovation lies in the unprecedented metal-catalyzed insertion of carbenes into olefin C=C bonds using thionoketenes as polarized internal olefins—a transformation previously unreported in literature. The mechanism initiates with base-mediated decomposition of p-toluenesulfonylhydrazone to generate diazo intermediates that undergo oxidative addition with copper(I) catalysts to form copper-carbene complexes. These electrophilic species then engage the electron-rich enone system through a concerted cyclization process where simultaneous nucleophilic attack by the enol oxygen and carbene insertion occurs at the olefinic bond. This dual activation pathway creates a highly organized transition state that controls stereochemistry through steric guidance from substituent groups while facilitating direct formation of both C-O and C-C bonds in a single kinetic step. The copper catalyst plays a dual role in stabilizing reactive intermediates while preventing undesired dimerization pathways that typically plague carbene chemistry, thereby ensuring high regioselectivity across diverse substrate combinations.
Impurity control is achieved through multiple built-in mechanisms within this catalytic cycle that prevent common side reactions observed in conventional syntheses. The mild reaction conditions suppress thermal decomposition pathways while the nitrogen atmosphere eliminates oxidation byproducts that typically complicate purification processes. The precise stoichiometric control over base concentration prevents over-decomposition of sensitive intermediates, maintaining optimal reaction kinetics that minimize dimer formation or rearrangement products. Furthermore, the solvent system—particularly aprotic media like toluene—creates an ideal microenvironment that stabilizes polar transition states while facilitating clean product separation during workup procedures. This inherent selectivity translates directly to superior product purity profiles without requiring additional purification steps that would otherwise increase manufacturing costs and reduce overall process efficiency.
How to Synthesize High-Purity Imine Furan Derivatives Efficiently
This patented methodology provides a robust framework for producing complex heterocyclic intermediates through a carefully optimized sequence that begins with precursor preparation using commercially available starting materials under controlled laboratory conditions. The process demonstrates exceptional reproducibility across multiple solvent systems and catalyst concentrations while maintaining consistent product quality essential for pharmaceutical applications. By eliminating hazardous reagents and complex intermediate handling procedures characteristic of traditional routes, this approach significantly reduces operational risks while enhancing overall process safety profiles. Detailed standardized synthesis steps are provided below to ensure reliable implementation across diverse manufacturing environments while maintaining critical quality attributes required for regulatory compliance.
- Prepare the key precursor through reaction of dithioketene with primary amine in ethanol at precisely controlled temperatures to form the essential enone intermediate.
- Generate p-toluenesulfonylhydrazone from ketone substrates under optimized methanol reflux conditions while maintaining strict stoichiometric ratios.
- Execute cyclization and carbene insertion under nitrogen atmosphere using copper catalysts with tertiary alkoxide bases in aprotic solvents at elevated temperatures.
Commercial Advantages for Procurement and Supply Chain Teams
This innovative synthesis methodology delivers substantial value across procurement and supply chain operations by addressing fundamental pain points associated with traditional intermediate manufacturing processes. The elimination of multi-step sequences reduces both raw material requirements and processing time while enhancing overall operational reliability through simplified workflow management. By utilizing commercially available starting materials with established global supply chains, this approach mitigates sourcing risks commonly encountered with specialized reagents required in conventional syntheses while ensuring consistent material availability regardless of market fluctuations.
- Cost Reduction in Manufacturing: The one-step process significantly reduces manufacturing costs by eliminating expensive purification steps typically required to remove transition metal contaminants from conventional syntheses. The use of copper catalysts instead of precious metals like palladium or rhodium avoids costly metal recovery procedures while maintaining high reaction efficiency under mild conditions that lower energy consumption without compromising product quality.
- Enhanced Supply Chain Reliability: Sourcing flexibility is dramatically improved through reliance on readily available starting materials with multiple global suppliers, reducing dependency on single-source reagents that create supply chain vulnerabilities. The robust reaction design maintains consistent performance across different production scales without requiring specialized equipment modifications, ensuring reliable output even during periods of raw material volatility or logistical disruptions.
- Scalability and Environmental Compliance: The methodology demonstrates exceptional scalability from laboratory benchtop to commercial production volumes while maintaining stringent quality standards through simplified waste stream management. The elimination of hazardous reagents reduces environmental impact by minimizing toxic byproduct generation during both synthesis and purification stages, aligning with increasingly stringent regulatory requirements for sustainable manufacturing practices without requiring significant capital investment.
Frequently Asked Questions (FAQ)
The following questions address critical technical considerations based on patent specifications and experimental validation data from multiple synthesis examples documented in CN108148021B. These responses provide essential insights for technical evaluation teams assessing implementation feasibility within existing manufacturing frameworks while addressing common concerns regarding process robustness and quality assurance protocols.
Q: How does this methodology improve upon conventional multi-step synthesis routes?
A: The patented one-step cyclization and carbene insertion process eliminates complex intermediate isolation procedures required in traditional approaches. By directly converting readily available starting materials into target heterocycles under mild conditions (80–140°C), it significantly reduces operational complexity while enhancing stereoselectivity and functional group tolerance compared to conventional methods requiring harsh reaction environments.
Q: What specific advantages does copper catalysis provide for pharmaceutical intermediate production?
A: Copper-based catalytic systems enable precise control over carbene insertion into olefin C=C bonds under nitrogen atmosphere, achieving high yields without transition metal contamination risks. The methodology maintains stringent purity specifications through simplified purification protocols while accommodating diverse substrate combinations essential for pharmaceutical development pipelines.
Q: Can this process be reliably scaled for commercial pharmaceutical manufacturing?
A: The robust reaction design demonstrates consistent performance across multiple solvent systems and catalyst concentrations with documented scalability from laboratory to pilot scale. The use of commercially available reagents and straightforward workup procedures ensures seamless transition to industrial production while maintaining critical quality attributes required for regulatory compliance.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Imine Furan Derivative Supplier
Our patented technology represents a significant advancement in heterocyclic compound manufacturing that directly addresses critical challenges in producing high-purity pharmaceutical intermediates at commercial scale. NINGBO INNO PHARMCHEM brings extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production while maintaining stringent purity specifications through rigorous QC labs equipped with advanced analytical capabilities. Our technical team has successfully implemented similar complex synthetic routes across multiple therapeutic areas, ensuring seamless technology transfer from laboratory development to full-scale manufacturing without compromising quality or regulatory compliance requirements.
We invite you to initiate a Customized Cost-Saving Analysis tailored to your specific production needs by contacting our technical procurement team today. Request detailed COA data and route feasibility assessments demonstrating how our patented methodology can optimize your supply chain while delivering consistent high-purity imine furan derivatives meeting your exact specifications.
