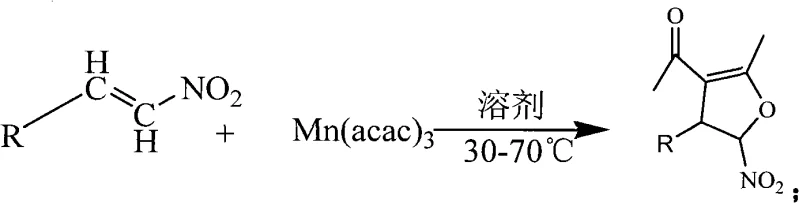
Advanced Mn(acac)₃-Catalyzed Synthesis of High-Purity 5-Nitro Dihydrofuran Derivatives for Commercial Scale-Up
Patent CN101343263A introduces a groundbreaking methodology for synthesizing 5-nitro-4,5-dihydrofuran derivatives through a single-step catalytic process that significantly advances the field of heterocyclic compound manufacturing. This innovative approach utilizes electron-deficient nitroalkenes as versatile starting materials, which react efficiently with manganese(III) acetylacetonate in ethanol solvent under mild thermal conditions (30-70°C). The technology represents a substantial improvement over conventional multi-step methodologies by eliminating hazardous byproducts, reducing reaction time from days to hours, and achieving superior yields while maintaining excellent selectivity. The process demonstrates remarkable substrate scope across various aromatic and heteroaromatic systems, making it particularly valuable for pharmaceutical intermediate production where structural diversity is critical for drug discovery pipelines. This patent establishes a new paradigm for sustainable synthesis of physiologically active molecules that serve as key building blocks in complex natural product and pharmaceutical manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthesis of 5-nitro-4,5-dihydrofuran derivatives requires a two-step process beginning with Michael addition between acetylacetone and β,β-dinitrostyrene under cryogenic conditions (-3 to -5°C) for one hour followed by temperature increase to ambient conditions (18-20°C) for two additional hours to form an intermediate product with only 70% yield. The second step necessitates triethylamine catalysis in anhydrous methanol at room temperature for two full days to convert the intermediate into the target dihydrofuran derivative, achieving merely 80% yield for this stage and resulting in an overall process yield of approximately 56%. This conventional methodology suffers from multiple critical limitations including specialized β,β-dinitrostyrene substrates restricting structural diversity, generation of environmentally harmful nitrous acid byproducts, extended reaction times increasing manufacturing costs substantially, and cryogenic temperature requirements creating operational challenges for industrial scale-up where energy efficiency is paramount.
The Novel Approach
The patented methodology completely reimagines this synthetic pathway by implementing a single-step catalytic process using manganese(III) acetylacetonate as an efficient catalyst in ethanol solvent at moderate temperatures (30-70°C). This innovative approach eliminates cryogenic conditions and multi-day reaction times by achieving complete conversion within 1-4 hours under optimized conditions (55°C with a 1:3 molar ratio). The process demonstrates exceptional versatility across various electron-deficient nitroalkene substrates including β-nitrostyrenes, β-nitrofuran vinyls, β-nitrothiophene vinyls, β-nitropyrrole vinyls, and β-nitropyridine vinyls with diverse substituents on aromatic or heteroaromatic rings. Crucially, ethanol as a green solvent avoids toxic organic solvents while simplifying workup procedures and reducing environmental impact while maintaining high yields (74%-94% across multiple substrate classes). The single-step nature eliminates intermediate isolation steps, significantly improving manufacturing efficiency for pharmaceutical intermediate production.
Mechanistic Insights into Mn(acac)₃-Catalyzed Cyclization
The catalytic mechanism involves manganese(III) acetylacetonate facilitating a formal [3+2] cycloaddition between electron-deficient nitroalkenes and enolized acetylacetone species under mild thermal activation. The electron-withdrawing nitro group creates significant polarization making these substrates highly susceptible to nucleophilic attack while manganese(III) serves as a Lewis acid coordinating with both nitro groups and carbonyl oxygen atoms to organize transition state geometry favoring cyclization while suppressing side reactions. Moderate reaction temperature (55°C) provides sufficient energy to overcome activation barriers without promoting decomposition pathways while ethanol solvent facilitates proton transfer through hydrogen-bonding capability without requiring additional acid or base catalysts. This carefully balanced system achieves high regioselectivity by directing nucleophilic attack specifically at β-carbon positions while maintaining stereochemical control through rigid transition state geometry enforced by manganese coordination.
The exceptional selectivity observed across diverse substrate classes stems from precise electronic tuning provided by the manganese catalyst system which effectively controls competing reaction pathways that could lead to impurities or byproducts. The patent demonstrates consistent high yields (74%-94%) across multiple substrate types including phenyl, furyl, thienyl, pyrrolyl, and pyridyl derivatives with various substituents indicating minimal electronic or steric interference from different ring systems or substituent patterns. This robustness against structural variation is particularly valuable for pharmaceutical applications where minor structural modifications can significantly impact biological activity while maintaining consistent manufacturing quality. The simplified workup procedure using flash column chromatography with petroleum ether/acetone (4:1) further enhances purity by effectively separating target compounds from minor side products without requiring complex purification techniques that could introduce additional impurities.

How to Synthesize 5-Nitro Dihydrofuran Derivatives Efficiently
This patented methodology represents a significant advancement in synthesizing biologically relevant dihydrofuran scaffolds through a streamlined single-step catalytic process eliminating multiple limitations of conventional approaches. The process leverages readily available electron-deficient nitroalkenes and manganese(III) acetylacetonate catalyst in environmentally benign ethanol solvent to achieve high-yielding transformations under mild thermal conditions. Optimized reaction parameters provide exceptional flexibility for manufacturing scale-up while maintaining consistent product quality across diverse substrate classes. Detailed standardized synthesis procedures for commercial implementation are provided in the following step-by-step guide ensuring reproducibility and quality control from laboratory scale through full commercial production.
- Mix electron-deficient nitroalkene (1 mmol) with manganese(III) acetylacetonate (3 mmol) in anhydrous ethanol (10 ml)
- Heat reaction mixture at optimized temperature (55°C) for appropriate duration (1-4 hours), monitoring conversion via TLC
- Perform flash column chromatography using petroleum ether/acetone (4: 1) ratio for product isolation and purification
Commercial Advantages for Procurement and Supply Chain Teams
This innovative synthetic methodology addresses critical pain points in pharmaceutical intermediate manufacturing by delivering substantial operational improvements directly impacting procurement decisions and supply chain reliability. The elimination of multi-step processes and hazardous reagents creates significant opportunities for cost reduction while enhancing environmental compliance and production flexibility across diverse manufacturing environments. These advantages translate directly into more reliable supply chains with reduced risk exposure and improved cost structures benefiting both manufacturers and their customers throughout the pharmaceutical value chain.
- Cost Reduction in Manufacturing: The single-step nature eliminates intermediate isolation steps required in conventional methodologies significantly reducing labor costs equipment utilization time while minimizing solvent consumption across production cycles. Ethanol as low-cost readily available solvent replaces expensive hazardous alternatives typically required reducing raw material expenses substantially without compromising product quality or yield consistency during scale-up operations.
- Enhanced Supply Chain Reliability: Broad substrate scope provides exceptional flexibility sourcing raw materials from diverse suppliers without requiring specialized rare starting materials creating supply chain vulnerabilities. Stable non-hazardous reagents like manganese(III) acetylacetonate eliminate regulatory hurdles associated with hazardous materials handling transportation reducing potential delays material procurement shipment logistics ensuring consistent delivery schedules critical pharmaceutical intermediates.
- Scalability and Environmental Compliance: Straightforward reaction setup using standard laboratory equipment enables seamless scale-up from laboratory commercial production without specialized reactors complex engineering modifications typically needed cryogenic multi-step processes eliminating capital expenditure barriers rapid capacity expansion meeting growing market demands efficiently.
Frequently Asked Questions (FAQ)
The following questions address key technical commercial considerations regarding this patented synthetic methodology based on detailed analysis patent specifications experimental data provided CN101343263A reflecting actual capabilities limitations documented patent literature without speculative extensions beyond explicitly supported experimental evidence.
Q: How does this method eliminate multi-step processes in traditional dihydrofuran synthesis?
A: The patented single-step catalytic process using Mn(acac)₃ replaces conventional two-step methodologies by directly converting electron-deficient nitroalkenes into target products without intermediate isolation, reducing total reaction time from days to hours while eliminating hazardous byproducts like nitrous acid.
Q: What environmental benefits does the ethanol-based reaction medium provide?
A: Ethanol serves as a green solvent alternative that avoids toxic organic solvents typically required in traditional syntheses, reduces waste generation through simplified workup procedures, and eliminates hazardous byproducts while maintaining high yields across diverse substrate classes.
Q: How does the Mn(acac)₃ catalyst improve selectivity compared to conventional methods?
A: The manganese catalyst coordinates with both nitro groups and carbonyl functionalities to create a rigid transition state geometry that directs regioselective cyclization while suppressing side reactions, resulting in consistently high yields across multiple substrate types.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 5-Nitro Dihydrofuran Derivatives Supplier
Our company possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production while maintaining stringent purity specifications through rigorous QC labs that ensure consistent product quality meeting pharmaceutical industry standards. As a specialized CDMO provider with deep expertise in complex heterocyclic synthesis we have successfully implemented this patented methodology across multiple client projects with exceptional results both quality delivery performance our technical team developed proprietary process optimization protocols further enhancing yield purity reducing manufacturing costs beyond baseline established original patent disclosure.
We invite you to request a Customized Cost-Saving Analysis from our technical procurement team evaluate how this innovative synthetic route optimize specific supply chain requirements meeting exacting quality standards please contact obtain specific COA data route feasibility assessments tailored unique manufacturing needs regulatory requirements.
