Transforming Pharmaceutical Intermediate Manufacturing: Nickel-Catalyzed 1,1-Aryl Boronation for Scalable Commercial Production
The Chinese patent CN111471065A introduces a groundbreaking nickel-catalyzed methodology for synthesizing 1,1-aryl boron compounds from terminal olefins, representing a significant advancement in organoboron chemistry with direct applications in pharmaceutical manufacturing. This innovative process addresses longstanding challenges in creating complex molecular architectures required for active pharmaceutical ingredients through its exceptional regioselectivity and enantioselectivity. The method employs readily available nickel catalysts paired with specifically designed ligands to facilitate the transformation of non-activated terminal olefins into valuable boron-containing intermediates under mild reaction conditions. Unlike conventional approaches that often require pre-functionalized substrates or harsh reaction environments, this patented technique operates efficiently at moderate temperatures (30-80°C) with excellent functional group tolerance across diverse substrate classes. The resulting boron compounds serve as versatile synthetic building blocks that can be readily converted into benzyl alcohol derivatives through straightforward oxidation protocols, significantly expanding their utility in complex molecule synthesis for pharmaceutical applications.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional methods for synthesizing aryl boron compounds have been severely constrained by limited substrate scope, poor stereoselectivity, and demanding reaction conditions that hinder their practical implementation in commercial pharmaceutical manufacturing settings. Previous approaches typically required activated olefin substrates or specialized pre-functionalized starting materials that significantly increased raw material costs and complexity of supply chains. Many existing protocols suffered from low regioselectivity when applied to terminal olefins, often producing undesired regioisomers that complicated purification processes and reduced overall yields of target intermediates. The harsh reaction conditions employed in conventional methods frequently necessitated specialized equipment and extensive safety protocols, creating significant barriers to scale-up from laboratory to commercial production volumes. Furthermore, traditional catalytic systems often exhibited poor functional group compatibility, limiting their applicability to complex pharmaceutical intermediates that contain sensitive moieties essential for final drug molecule construction.
The Novel Approach
The patented nickel-catalyzed methodology overcomes these limitations through a carefully engineered catalytic system featuring specifically designed ligands that enable precise control over both regioselectivity and stereoselectivity during the boronation process. This innovative approach successfully converts non-activated terminal olefins directly into valuable 1,1-aryl boron compounds with excellent selectivity under remarkably mild conditions (30-80°C), eliminating the need for pre-functionalized substrates that characterize conventional approaches. The process demonstrates exceptional functional group tolerance across a wide range of substrate classes, including those containing esters, ethers, halogens, and heterocyclic moieties commonly found in pharmaceutical intermediates. By employing commercially available nickel catalysts paired with specifically designed ligands and standard organic solvents, this method achieves high yields while maintaining operational simplicity that facilitates seamless scale-up from laboratory to commercial production environments. The resulting boron-containing intermediates possess structural features that enable straightforward conversion into diverse benzyl alcohol derivatives through simple oxidation protocols.
Mechanistic Insights into Nickel-Catalyzed 1,1-Aryl Boronation
The catalytic cycle begins with oxidative addition of the aryl halide to the nickel(0) species generated in situ from the nickel(II) precursor and reducing agent. This key step forms an aryl-nickel(II) intermediate that subsequently undergoes migratory insertion with the terminal olefin substrate, positioning the nickel center at the terminal carbon position. The unique ligand architecture plays a critical role in controlling both regioselectivity and stereoselectivity by creating a well-defined chiral environment around the nickel center that directs the approach of substrates and stabilizes key transition states. Following migratory insertion, transmetalation with bisboronic acid pinacol ester delivers the boron functionality while regenerating the active nickel species for subsequent catalytic cycles. The specific ligand design prevents undesired β-hydride elimination pathways that would lead to reduced selectivity, ensuring high fidelity in product formation through precise spatial control of substrate orientation within the catalytic pocket.
Impurity control is achieved through multiple mechanisms inherent in this catalytic system, beginning with the ligand's ability to suppress competing reaction pathways that would generate regioisomeric byproducts. The carefully optimized reaction conditions prevent over-boronation or protodeboronation side reactions that commonly plague traditional methods, while the mild temperature profile minimizes thermal decomposition pathways that could introduce impurities. The use of specific solvent systems enhances solubility control during reaction progression, preventing aggregation phenomena that might otherwise lead to heterogeneous reaction conditions and inconsistent product quality. Column chromatography purification following standard workup procedures effectively separates any minor impurities from the target product, yielding materials with purity levels suitable for direct use in subsequent pharmaceutical synthesis steps without requiring additional purification stages.
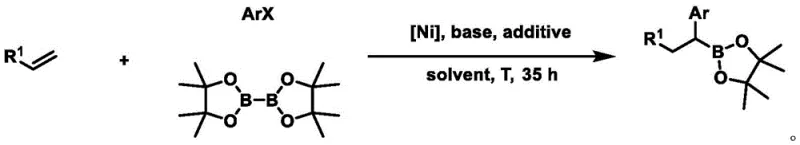
How to Synthesize 1,1-Aryl Boron Compounds Efficiently
This patented methodology represents a significant advancement in organoboron chemistry with direct applications in pharmaceutical intermediate manufacturing. The process enables efficient conversion of readily available starting materials into high-value boron-containing intermediates through a carefully optimized catalytic system that delivers superior selectivity and yield compared to conventional approaches. Detailed standardized synthesis steps are provided below to facilitate seamless implementation in commercial manufacturing environments.
- Prepare reaction mixture under inert atmosphere by dissolving nickel catalyst (5 mol%), specific ligand (5 mol%), base (1.5 equiv), and bisboronic acid pinacol ester in dry organic solvent
- Add terminal olefin (1 equiv) and aryl halide (1.5 equiv) to the reaction mixture, then seal and remove from inert environment
- Conduct reaction at controlled temperature (30-80°C) for specified duration (typically 35 hours), followed by solvent removal and column chromatography purification
Commercial Advantages for Procurement and Supply Chain Teams
This innovative manufacturing process delivers substantial value across procurement and supply chain operations by addressing critical pain points associated with traditional methods of producing complex organoboron intermediates for pharmaceutical applications. The methodology eliminates multiple processing steps while improving overall yield and purity profiles, creating significant operational efficiencies that translate directly into enhanced supply chain reliability and cost-effectiveness.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts required by alternative methods significantly reduces raw material costs while simplifying catalyst recovery protocols. The use of commercially available nickel precursors instead of precious metal catalysts creates substantial cost savings without compromising product quality or selectivity. The streamlined reaction sequence reduces solvent consumption and eliminates specialized equipment requirements that typically increase capital expenditure in traditional manufacturing setups.
- Enhanced Supply Chain Reliability: The process utilizes widely available starting materials with established global supply chains, reducing vulnerability to single-source dependencies that often disrupt traditional manufacturing operations. The robust nature of the reaction tolerates minor variations in raw material quality while maintaining consistent product specifications, providing greater flexibility in supplier selection without compromising final product quality requirements.
- Scalability and Environmental Compliance: The methodology demonstrates exceptional scalability from laboratory to commercial production volumes without requiring significant process re-engineering or specialized equipment modifications. The simplified waste stream profile resulting from reduced solvent usage and elimination of toxic byproducts significantly lowers environmental impact while simplifying waste treatment protocols required for regulatory compliance in global manufacturing facilities.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial concerns regarding implementation of this patented methodology in pharmaceutical intermediate manufacturing operations.
Q: How does this nickel-catalyzed method improve upon conventional approaches for synthesizing aryl boron compounds?
A: The patented method overcomes limitations of traditional approaches by providing excellent regioselectivity and enantioselectivity while using readily available starting materials. Unlike previous methods that required specific activated substrates, this process efficiently converts non-activated terminal olefins into valuable 1,1-aryl boron compounds with high purity.
Q: What makes this boronation process particularly suitable for pharmaceutical intermediate production?
A: The method delivers superior control over stereochemistry with excellent enantioselectivity, producing high-purity intermediates critical for pharmaceutical applications. The ability to convert these boron compounds into diverse benzyl alcohol derivatives through simple oxidation provides significant flexibility in API synthesis pathways.
Q: How does this process address supply chain challenges in pharmaceutical manufacturing?
A: The process utilizes commercially available catalysts and reagents with straightforward reaction conditions that scale efficiently from laboratory to commercial production. The simplified purification process through standard column chromatography eliminates complex separation steps that typically create bottlenecks in pharmaceutical intermediate supply chains.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1,1-Aryl Boron Compounds Supplier
Our company brings extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production of complex organoboron intermediates while maintaining stringent purity specifications required by global pharmaceutical clients. With state-of-the-art facilities featuring rigorous QC labs equipped for comprehensive analytical testing including NMR spectroscopy and HRMS verification, we ensure consistent product quality across all production scales through meticulously validated processes developed by our expert technical team.
Leverage our expertise through a Customized Cost-Saving Analysis tailored to your specific manufacturing requirements by contacting our technical procurement team today to request detailed COA data and route feasibility assessments for your next-generation pharmaceutical intermediate needs.
