Revolutionizing Oxazolidinone Intermediate Production via Aqueous Cyclization Technology
Introduction to Advanced Green Synthesis Technologies
The pharmaceutical industry is currently undergoing a significant paradigm shift towards greener, more sustainable manufacturing processes, driven by both regulatory pressure and the economic necessity of reducing waste. In this context, the technological breakthroughs detailed in patent CN111808040B represent a critical advancement for the production of 2-oxo-oxazolidine-4-carboxylic acid compounds, which serve as vital intermediates in the synthesis of antibacterial agents targeting gram-negative bacilli. This patent introduces a novel synthetic route that fundamentally alters the reaction medium, moving away from hazardous organic solvents to a benign aqueous system. By leveraging S,S'-dimethyl dithiocarbonate as a key cyclization reagent in the presence of water, the process not only mitigates environmental impact but also dramatically enhances operational safety and product throughput. For R&D directors and procurement specialists alike, understanding the nuances of this technology is essential for evaluating potential supply chain partners who can deliver high-purity pharmaceutical intermediates with consistent reliability and reduced ecological footprints.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 2-oxo-oxazolidine-4-carboxylic acid derivatives has been plagued by significant inefficiencies and safety hazards inherent to traditional organic synthesis protocols. Prior art methods, such as those disclosed in patent WO2017189866A1, typically rely on the use of bis(trichloromethyl) carbonate or methyl chloroformate as cyclization agents, necessitating the use of volatile and toxic organic solvents like dichloromethane, dioxane, or acetonitrile. These solvents pose severe health risks, including irritation to the skin, eyes, and respiratory systems, requiring expensive containment and ventilation systems to ensure worker safety. Furthermore, the chemical efficiency of these legacy processes is notoriously poor; for example, the route utilizing bis(trichloromethyl) carbonate in a dioxane solution often results in abysmal product yields, sometimes as low as 8.5%, which is economically unsustainable for large-scale manufacturing.
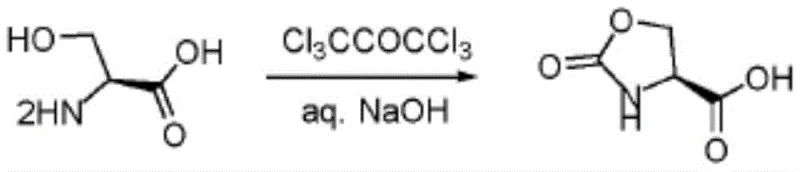
Similarly, other established methods described in documents like US20080267916A1 attempt to mitigate some issues but still suffer from suboptimal performance metrics. These processes often involve complex workup procedures, such as freeze-drying and multiple solvent exchanges between acetonitrile and aqueous phases, which increase energy consumption and processing time. Even with these elaborate steps, the calculated product yields rarely exceed 62%, leaving a substantial portion of valuable starting materials wasted as byproducts. The reliance on halogenated solvents also creates a significant burden for waste disposal and environmental compliance, adding hidden costs to the final product price that erode profit margins for downstream API manufacturers.
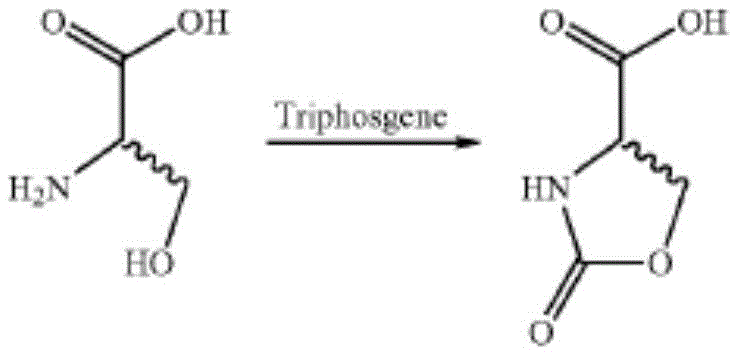
The Novel Approach
In stark contrast to these inefficient legacy methods, the novel approach outlined in CN111808040B utilizes a streamlined, two-step protocol that prioritizes both atom economy and environmental stewardship. The core innovation lies in the substitution of dangerous phosgene equivalents with S,S'-dimethyl dithiocarbonate, reacting directly with serine methyl ester hydrochloride derivatives in a purely aqueous medium. This shift eliminates the need for flammable and toxic organic solvents during the critical cyclization step, thereby simplifying the reactor setup and reducing the risk of fire or explosion. The reaction conditions are remarkably mild, typically proceeding at temperatures between 5°C and 35°C, which allows for precise thermal control without the need for extreme heating or cooling infrastructure. This accessibility of reaction conditions translates directly into lower capital expenditure requirements for manufacturing facilities, making it an attractive option for contract development and manufacturing organizations (CDMOs) looking to optimize their asset utilization.
Mechanistic Insights into Aqueous Cyclization and Hydrolysis
The mechanistic pathway of this synthesis is elegantly simple yet highly effective, relying on the nucleophilic attack of the amino group on the thiocarbonyl carbon of the S,S'-dimethyl dithiocarbonate. Under inert gas protection, the serine derivative undergoes an intramolecular cyclization to form the stable oxazolidinone ring structure, releasing methanethiol byproducts which are easily managed in the aqueous phase. This cyclization is highly chemoselective, minimizing the formation of polymeric byproducts or side-reactions that often plague reactions in organic solvents. The subsequent hydrolysis step is equally robust, where the intermediate ester is cleaved under strongly alkaline conditions (pH 12-14) using common bases like sodium hydroxide or potassium hydroxide. This base-mediated hydrolysis ensures complete conversion to the free carboxylic acid without racemization, preserving the stereochemical integrity of the chiral centers which is paramount for biological activity in the final drug substance.
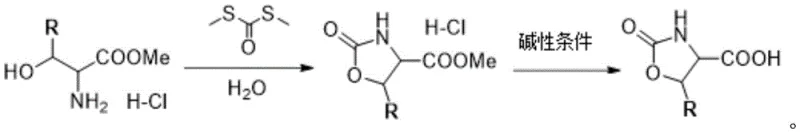
From an impurity control perspective, the aqueous nature of the reaction provides a unique advantage in purification. Many organic impurities that might co-elute in organic solvent systems are either insoluble in water or can be easily separated via pH-dependent extraction. The patent details a workup procedure involving extraction with dichloromethane or chloroform only after the initial cyclization, allowing for the removal of unreacted starting materials and non-polar byproducts before the final hydrolysis. This strategic sequencing of unit operations ensures that the final crystallization or chromatography steps yield a product of exceptional purity, often exceeding the stringent specifications required for GMP-grade pharmaceutical intermediates. The ability to consistently produce multi-configuration isomers, including specific enantiomers like (S)-2-oxooxazolidine-4-carboxylic acid, demonstrates the versatility of this mechanism in supporting diverse medicinal chemistry campaigns.
How to Synthesize 2-Oxo-Oxazolidine-4-Carboxylic Acid Efficiently
Implementing this synthesis route requires careful attention to the stoichiometry of the thiocarbonate reagent and the precise control of pH during the hydrolysis phase. The process begins with the dissolution of the amino acid ester hydrochloride in water, followed by the slow addition of S,S'-dimethyl dithiocarbonate to manage the exotherm and gas evolution. Detailed standardized operating procedures for temperature ramping and phase separation are critical to maximizing the reported yields of over 86%. For a comprehensive guide on the specific molar ratios, stirring rates, and purification techniques validated in the patent examples, please refer to the structured synthesis guide below.
- React serine methyl ester hydrochloride with S,S'-dimethyl dithiocarbonate in water under inert gas protection at 5-35°C to form the cyclic intermediate.
- Adjust the pH of the reaction mixture to 12-14 using an alkaline substance like sodium hydroxide to hydrolyze the ester group.
- Purify the final product through solvent removal and chromatography to obtain high-purity 2-oxo-oxazolidine-4-carboxylic acid derivatives.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this aqueous synthesis technology offers compelling economic and logistical benefits that extend far beyond simple yield improvements. The elimination of bulk organic solvents like dioxane and dichloromethane drastically reduces the raw material costs associated with solvent purchase, recovery, and disposal. Furthermore, the simplified workflow reduces the number of unit operations required, shortening the overall batch cycle time and increasing the throughput capacity of existing manufacturing suites. This efficiency gain allows suppliers to offer more competitive pricing structures while maintaining healthy margins, providing a distinct advantage in cost-sensitive markets such as generic antibiotic production.
- Cost Reduction in Manufacturing: The transition to water as the primary reaction solvent removes the significant expense associated with purchasing, storing, and recycling large volumes of hazardous organic chemicals. Additionally, the high reaction yield minimizes the waste of expensive chiral starting materials like serine derivatives, ensuring that a greater proportion of input costs are converted into saleable product. The reduction in hazardous waste generation also lowers the fees paid for environmental compliance and waste treatment, contributing to a leaner and more cost-effective production model.
- Enhanced Supply Chain Reliability: By utilizing readily available and stable reagents such as S,S'-dimethyl dithiocarbonate and common inorganic bases, the supply chain becomes less vulnerable to the fluctuations and shortages often seen with specialized or highly regulated reagents like phosgene equivalents. The robustness of the reaction conditions means that production is less likely to be interrupted by equipment failures related to solvent handling or extreme temperature requirements, ensuring a steady and predictable flow of intermediates to downstream API manufacturers.
- Scalability and Environmental Compliance: The inherent safety of the aqueous process facilitates easier scale-up from laboratory to commercial tonnage, as the risks of thermal runaway or solvent fires are significantly mitigated. This scalability is crucial for meeting the demands of large-volume contracts without the need for extensive retrofitting of production facilities. Moreover, the green chemistry credentials of this method align perfectly with the sustainability goals of major pharmaceutical companies, helping them reduce their Scope 3 emissions and meet corporate social responsibility targets.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and claims presented in the patent documentation, providing a factual basis for decision-making.
Q: What are the primary safety advantages of this new synthesis method compared to traditional routes?
A: Unlike conventional methods that rely on hazardous organic solvents like dichloromethane and toxic reagents such as bis(trichloromethyl) carbonate, this patented process utilizes water as the primary solvent. This eliminates the risks associated with volatile organic compounds (VOCs) and significantly reduces operator exposure to skin and respiratory irritants, ensuring a much safer production environment.
Q: How does the yield of this aqueous method compare to prior art techniques?
A: The patented aqueous synthesis method achieves product yields exceeding 86%, which represents a substantial improvement over previous techniques. For instance, older methods described in patents like WO2017189866A1 reported yields as low as 8.5%, while others struggled to reach 62%, making this new approach far more efficient for commercial scale-up.
Q: Can this method produce specific stereoisomers required for chiral drug synthesis?
A: Yes, the method is highly versatile and supports the synthesis of multi-configuration compounds. By selecting specific starting materials such as L-serine, D-serine, or various substituted amino-hydroxy esters, manufacturers can precisely produce distinct stereoisomers like (S)-2-oxooxazolidine-4-carboxylic acid or various 5-methyl derivatives, catering to diverse medicinal chemistry needs.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Oxo-Oxazolidine-4-Carboxylic Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize that the successful commercialization of complex pharmaceutical intermediates requires more than just a patent; it demands deep process engineering expertise and a commitment to quality. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from lab-scale discovery to industrial manufacturing is seamless. Our state-of-the-art facilities are equipped to handle the specific requirements of aqueous-phase organic synthesis, and our rigorous QC labs enforce stringent purity specifications to guarantee that every batch meets the exacting standards of the global pharmaceutical industry.
We invite you to collaborate with us to leverage this advanced synthesis technology for your next project. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating how this green route can optimize your bill of materials. Please contact our technical procurement team today to request specific COA data for our oxazolidinone intermediates and to discuss route feasibility assessments for your target molecules.
