Advanced Cobalt-Catalyzed Synthesis of Alpha-Phosphorylated Alpha-Amino Acid Esters for Pharma
Introduction to Green Phosphorylation Technology
The landscape of organophosphorus chemistry is undergoing a significant transformation driven by the urgent need for sustainable and atom-economical synthetic methodologies. Patent CN108948077B introduces a groundbreaking approach to synthesizing alpha-phosphorylated alpha-amino acid ester compounds, a class of molecules pivotal in the development of bioactive pharmaceutical ingredients and agrochemical agents. This technology leverages a transition metal-catalyzed cross-dehydrogenative coupling (CDC) strategy, which fundamentally alters the traditional paradigm of carbon-phosphorus bond construction. By utilizing molecular oxygen from the air as the terminal oxidant, this method bypasses the necessity for stoichiometric amounts of hazardous chemical oxidants, thereby aligning perfectly with the principles of green chemistry. The core innovation lies in the direct activation of inert carbon-hydrogen bonds in N-arylglycine esters, enabling a streamlined one-step synthesis that maximizes atom utilization while minimizing environmental footprint.
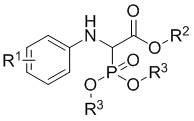
The structural versatility of the resulting alpha-phosphorylated alpha-amino acid esters is defined by the general formula (I), where diverse substituents can be introduced to tune the physicochemical properties for specific biological applications. As illustrated in the general structure below, the molecule features a central alpha-carbon bonded to an amine, an ester group, and a phosphonate moiety, creating a chiral center that is often critical for biological activity. The ability to systematically vary the R1 group on the aromatic ring, along with the R2 and R3 alkyl or benzyl groups on the ester and phosphite functionalities, provides medicinal chemists with a powerful toolkit for structure-activity relationship (SAR) studies. This flexibility ensures that the technology is not limited to a single compound but serves as a robust platform for generating libraries of high-value intermediates for drug discovery programs.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of carbon-phosphorus bonds in amino acid derivatives has relied heavily on classical substitution reactions that require pre-functionalized substrates. Traditional routes typically involve the halogenation of the alpha-position of amino acids or the use of activated leaving groups, which necessitates multiple synthetic steps prior to the actual phosphorylation event. These multi-step sequences not only increase the overall production time and cost but also generate substantial quantities of chemical waste, including halogenated byproducts and salt residues from neutralization steps. Furthermore, many conventional methods employ strong bases or highly reactive phosphorus halides, which pose significant safety risks and require stringent handling protocols in a manufacturing environment. The reliance on precious metal catalysts in some modern variants further exacerbates the cost burden and introduces challenges related to residual metal contamination in the final pharmaceutical product, necessitating expensive purification processes to meet regulatory standards.
The Novel Approach
In stark contrast, the methodology disclosed in CN108948077B represents a paradigm shift by enabling the direct coupling of unfunctionalized N-arylglycine esters with phosphites through a catalytic oxidative process. This novel approach eliminates the need for pre-activation of the substrate, effectively collapsing what was once a multi-step sequence into a single, efficient transformation. The reaction proceeds under mild thermal conditions, typically around 80°C, using inexpensive and earth-abundant cobalt salts as the catalyst, which drastically reduces the raw material costs compared to palladium or rhodium-based systems. The use of air as the oxidant is perhaps the most significant operational advantage, as it removes the need for storing and handling dangerous peroxides or hypervalent iodine reagents. This simplification of the reaction setup not only enhances process safety but also facilitates easier scale-up, as the engineering requirements for gas-liquid mass transfer with air are far less complex than those for handling hazardous liquid oxidants.

Mechanistic Insights into Cobalt-Catalyzed Cross-Dehydrogenative Coupling
The mechanistic underpinning of this transformation involves a sophisticated interplay between the cobalt catalyst and the molecular oxygen present in the air. The cycle likely initiates with the coordination of the cobalt species to the nitrogen atom of the N-arylglycine ester, which activates the adjacent alpha-carbon-hydrogen bond towards abstraction. Under the influence of the oxidant, a single-electron transfer process generates a reactive radical intermediate at the alpha-position. This carbon-centered radical is then trapped by the nucleophilic phosphite species, forming the new carbon-phosphorus bond. The regeneration of the active cobalt catalyst is achieved through the reduction of molecular oxygen, which serves as the terminal electron acceptor, producing water as the only benign byproduct. This catalytic cycle is highly efficient, operating with low catalyst loading (typically 10 mol%), and demonstrates remarkable tolerance to various functional groups, ensuring that sensitive moieties on the aromatic ring or the ester side chains remain intact throughout the reaction.
From an impurity control perspective, the mildness of the reaction conditions plays a crucial role in maintaining high product purity. Unlike harsh acidic or basic conditions that might promote racemization or hydrolysis of the ester groups, this neutral to slightly acidic environment preserves the stereochemical integrity of the amino acid backbone, although the reaction itself produces a racemic mixture unless chiral catalysts are employed. The selectivity of the C-H activation is directed by the adjacent amine group, minimizing the formation of regioisomers that could complicate downstream purification. Furthermore, the use of acetonitrile as the preferred solvent ensures good solubility for both organic substrates and the ionic cobalt catalyst, facilitating homogeneous catalysis which generally leads to cleaner reaction profiles. The combination of high chemical selectivity and the absence of aggressive reagents results in a crude product profile that is amenable to straightforward purification via column chromatography or crystallization, yielding materials with purity levels suitable for subsequent pharmaceutical processing.
How to Synthesize Alpha-Phosphorylated Alpha-Amino Acid Esters Efficiently
The practical execution of this synthesis protocol is designed for operational simplicity, making it accessible for both laboratory research and pilot plant operations. The process begins by combining the N-arylglycine ester substrate and the phosphite coupling partner in a dry reaction vessel equipped with magnetic stirring. A cobalt salt, preferably cobalt perchlorate hexahydrate due to its superior solubility and activity, is added to the mixture along with acetonitrile as the reaction medium. The system is then heated to 80°C under an open air atmosphere or with an air balloon, allowing the oxidative coupling to proceed over a period of approximately 12 hours. Reaction progress is monitored via thin-layer chromatography (TLC) to ensure complete consumption of the starting material. Upon completion, the solvent is removed under reduced pressure, and the resulting residue is subjected to standard purification techniques to isolate the target alpha-phosphorylated product in high yield.
- Mix N-arylglycine ester and phosphite compound in an organic solvent such as acetonitrile with a cobalt catalyst like cobalt perchlorate hexahydrate.
- Heat the reaction mixture to approximately 80°C under an air atmosphere for about 12 hours to facilitate the oxidative coupling.
- Upon completion, remove the solvent via rotary evaporation and purify the crude residue using column chromatography to isolate the pure product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this cobalt-catalyzed technology offers compelling economic and logistical benefits that directly impact the bottom line. The elimination of pre-functionalized starting materials means that manufacturers can source cheaper, commodity-grade N-arylglycine esters rather than paying a premium for halogenated or activated derivatives. This shift in raw material sourcing significantly reduces the cost of goods sold (COGS) and mitigates the risk of supply disruptions associated with specialized reagents. Additionally, the replacement of expensive precious metal catalysts with abundant cobalt salts results in substantial cost savings on catalyst procurement and recovery. The simplified workup procedure, which avoids complex quenching steps required for reactive oxidants, translates into reduced labor hours and lower utility consumption during the manufacturing process, further enhancing the overall cost efficiency of the production campaign.
- Cost Reduction in Manufacturing: The economic model of this synthesis is heavily favored by the use of air as a free and inexhaustible oxidant, removing the recurring cost of purchasing chemical oxidants like TBHP or DTBP. Moreover, the high atom economy of the cross-dehydrogenative coupling ensures that a larger proportion of the input mass is converted into the desired product, minimizing waste disposal costs which are a significant factor in fine chemical manufacturing. The ability to run the reaction at moderate temperatures (80°C) also reduces energy consumption compared to high-temperature reflux conditions often required in traditional methods. These cumulative factors create a leaner manufacturing process that delivers significant cost reductions without compromising on the quality or yield of the final active pharmaceutical ingredient intermediate.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the reliance on widely available and stable starting materials. N-arylglycine esters and dialkyl phosphites are bulk chemicals produced by numerous suppliers globally, reducing the dependency on single-source vendors for exotic reagents. The robustness of the reaction conditions, which tolerate moisture and oxygen to a certain extent (since air is the oxidant), makes the process less sensitive to minor variations in raw material quality or environmental conditions during storage and transport. This inherent stability ensures consistent production schedules and reduces the likelihood of batch failures due to reagent degradation, thereby securing a steady flow of intermediates to downstream formulation units and preventing costly production delays.
- Scalability and Environmental Compliance: Scaling this process from gram to tonnage scale is facilitated by the use of standard reactor materials and the absence of highly corrosive or explosive reagents. The benign nature of the byproducts (primarily water) simplifies effluent treatment and helps facilities maintain compliance with increasingly stringent environmental regulations regarding volatile organic compounds (VOCs) and hazardous waste. The high yields observed across a broad range of substrates indicate that the process is robust enough to handle the variability inherent in large-scale operations. This scalability ensures that the technology can meet the growing demand for alpha-phosphorylated amino acid derivatives in the pharmaceutical and agrochemical sectors, providing a reliable long-term supply solution for strategic partners.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and scope defined in the patent literature, providing a clear understanding of the method's capabilities and limitations. Understanding these details is crucial for R&D teams evaluating the feasibility of integrating this route into their existing process pipelines and for procurement teams assessing the long-term viability of the supply chain.
Q: What are the primary advantages of this cobalt-catalyzed method over traditional phosphorylation?
A: This method eliminates the need for pre-functionalized substrates and hazardous oxidants, utilizing air as a green terminal oxidant which significantly simplifies the workflow and reduces waste generation compared to traditional halogenation-based routes.
Q: Is this synthesis method suitable for large-scale industrial production?
A: Yes, the process employs commercially available cobalt salts and mild reaction conditions (80°C), avoiding expensive precious metals and extreme pressures, making it highly scalable and economically viable for commercial manufacturing.
Q: What types of substituents are tolerated in this cross-dehydrogenative coupling reaction?
A: The reaction demonstrates broad substrate scope, tolerating various electron-donating and electron-withdrawing groups on the aryl ring, as well as diverse alkyl and benzyl groups on both the ester and phosphite moieties.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Alpha-Phosphorylated Alpha-Amino Acid Ester Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this cobalt-catalyzed cross-dehydrogenative coupling technology in accelerating the development of next-generation therapeutics. As a leading CDMO partner, we possess the technical expertise and infrastructure to translate this innovative academic research into robust, commercial-scale manufacturing processes. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project can seamlessly transition from clinical trial material to market supply. We are committed to delivering high-purity alpha-phosphorylated alpha-amino acid esters that meet stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation to verify identity and assay.
We invite you to collaborate with us to leverage this cost-effective and environmentally friendly synthesis route for your specific drug development programs. Our technical sales team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements, demonstrating how this method can optimize your budget. Please contact our technical procurement team today to request specific COA data for our catalog compounds or to discuss route feasibility assessments for your custom synthesis projects. Let us be your strategic partner in bringing high-quality phosphorus-containing intermediates to the global market efficiently and sustainably.
