Advanced Rhodium-Catalyzed Synthesis of Furanone Isoquinolinones for Commercial Scale Production
Introduction to Advanced Heterocyclic Synthesis
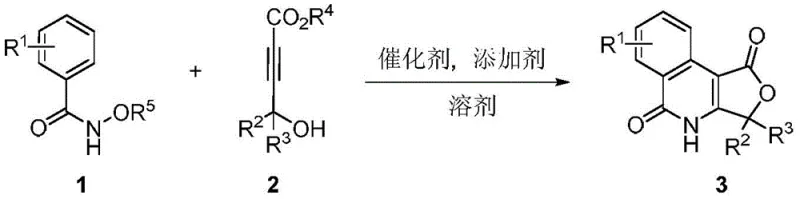
The development of efficient synthetic routes for fused nitrogen/oxygen heterocyclic compounds remains a critical priority in modern medicinal chemistry, particularly for creating scaffolds with potent biological activity. Patent CN108148070B introduces a groundbreaking methodology for synthesizing furanone isoquinolinone compounds, a class of molecules with significant potential in treating cardiovascular diseases, AIDS, and psychosis. This innovation leverages a rhodium-catalyzed one-pot tandem reaction between N-alkoxy aryl formamide compounds and 4-hydroxy-2-alkynoate compounds. By enabling the simultaneous construction of complex heterocyclic systems under mild conditions, this technology addresses longstanding challenges in organic synthesis regarding step economy and operational simplicity. For pharmaceutical manufacturers, this represents a pivotal shift towards more sustainable and cost-effective production of high-value intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of furanone isoquinolinone derivatives has been plagued by significant inefficiencies that hinder commercial viability. Traditional literature methods often rely on multi-step sequences that require the isolation of unstable intermediates, leading to cumulative yield losses and increased waste generation. Furthermore, these conventional pathways frequently necessitate harsh reaction conditions, such as extreme temperatures or the use of hazardous reagents, which pose safety risks and complicate process control. The starting materials in older methods are often difficult to obtain or require extensive pre-functionalization, driving up raw material costs and extending lead times. Additionally, poor regioselectivity in traditional cyclization reactions can result in complex mixtures of isomers, making downstream purification arduous and reducing the overall purity of the final active pharmaceutical ingredient.
The Novel Approach
In stark contrast, the novel approach detailed in the patent utilizes a direct C-H activation strategy that streamlines the entire synthetic process into a single operational unit. By employing a dichloro(pentamethylcyclopentadienyl)rhodium(III) dimer catalyst, the reaction achieves high efficiency through a one-pot tandem mechanism that directly couples readily available N-alkoxy aryl formamides with 4-hydroxy-2-alkynoates. This method operates under remarkably mild conditions, typically between 80-120 °C, which reduces energy consumption and minimizes thermal degradation of sensitive functional groups. The reaction exhibits excellent regioselectivity, ensuring that the desired furanone isoquinolinone scaffold is formed predominantly, thereby simplifying purification protocols. This technological leap not only accelerates the synthesis timeline but also enhances the overall atom economy of the process.
Mechanistic Insights into Rhodium-Catalyzed C-H Activation
The core of this synthetic breakthrough lies in the sophisticated rhodium(III) catalytic cycle that drives the C-H activation and subsequent annulation. The mechanism initiates with the coordination of the rhodium catalyst to the directing group on the N-alkoxy aryl formamide, facilitating the selective cleavage of the ortho C-H bond. This metallacycle intermediate then undergoes migratory insertion with the alkyne moiety of the 4-hydroxy-2-alkynoate, forming a new carbon-carbon bond with high precision. Following insertion, the hydroxyl group participates in an intramolecular nucleophilic attack, closing the furanone ring and releasing the final fused heterocyclic product while regenerating the active catalyst species. This elegant cascade allows for the formation of two rings and multiple bonds in a single transformation, showcasing the power of transition metal catalysis in complex molecule assembly.
Furthermore, the system demonstrates exceptional tolerance to diverse electronic and steric environments, which is crucial for generating a broad library of analogs for drug discovery. The use of additives such as potassium fluoride or cesium acetate plays a vital role in modulating the reactivity of the catalyst and stabilizing key intermediates, ensuring consistent performance across different substrate classes. Even substrates bearing electron-withdrawing groups like fluorine or chlorine, or bulky groups like tert-butyl, proceed smoothly to give the corresponding products in good yields. This robustness indicates that the catalytic cycle is resilient against potential poisoning or deactivation, a common issue in industrial catalysis. Such mechanistic reliability translates directly to process stability during scale-up operations.
How to Synthesize Furanone Isoquinolinone Efficiently
Implementing this synthesis in a laboratory or pilot plant setting requires careful attention to the stoichiometry and reaction environment to maximize yield and purity. The standard protocol involves dissolving the N-alkoxy aryl formamide and the 4-hydroxy-2-alkynoate in a solvent like ethylene glycol dimethyl ether (DME) or 1,4-dioxane. The catalyst loading is typically low, around 2.5 mol%, which is economically favorable for large-scale applications. An appropriate additive, such as potassium fluoride, is added to promote the reaction, and the mixture is heated under an inert nitrogen atmosphere to prevent oxidation of sensitive components. Detailed standardized synthesis steps follow below to ensure reproducibility and safety compliance.
- Dissolve N-alkoxy aryl formamide compounds and 4-hydroxy-2-alkynoate compounds in a suitable solvent such as ethylene glycol dimethyl ether.
- Add the dichloro(pentamethylcyclopentadienyl)rhodium(III) dimer catalyst and an additive like potassium fluoride to the reaction mixture.
- Stir and react the mixture at 80-120 °C under a nitrogen atmosphere for approximately 12 hours to obtain the target furanone isoquinolinone product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this rhodium-catalyzed methodology offers transformative benefits that extend beyond mere chemical efficiency. The consolidation of multiple synthetic steps into a single one-pot reaction drastically reduces the number of unit operations required, which directly correlates to lower capital expenditure on equipment and reduced labor costs. By eliminating the need for isolating intermediates, the process minimizes material handling and exposure, thereby enhancing workplace safety and reducing the risk of cross-contamination. The use of commercially available and inexpensive starting materials ensures a stable supply chain, mitigating the risks associated with sourcing exotic or custom-synthesized precursors. Moreover, the mild reaction conditions allow for the use of standard stainless steel reactors rather than specialized high-pressure or high-temperature vessels, further lowering the barrier to entry for manufacturing.
- Cost Reduction in Manufacturing: The elimination of intermediate isolation steps and the high atom economy of the tandem reaction significantly lower the overall cost of goods sold. By avoiding the use of stoichiometric amounts of hazardous reagents often found in traditional methods, waste disposal costs are substantially reduced. The high regioselectivity minimizes the formation of by-products, which means less solvent and energy are required for purification processes like chromatography or recrystallization. Additionally, the low catalyst loading combined with the potential for catalyst recovery contributes to a more economical use of precious metals, optimizing the financial footprint of the production process.
- Enhanced Supply Chain Reliability: The reliance on simple, commodity-grade chemicals such as N-methoxybenzamides and alkynoates ensures that raw material availability is not a bottleneck for production schedules. These precursors are widely produced by multiple suppliers globally, reducing dependency on single-source vendors and enhancing negotiation leverage. The robustness of the reaction conditions means that production is less susceptible to delays caused by equipment maintenance or stringent environmental controls required for harsher chemistries. This reliability allows for more accurate forecasting and inventory management, ensuring consistent delivery of high-purity pharmaceutical intermediates to downstream clients.
- Scalability and Environmental Compliance: The process is inherently designed for scalability, as demonstrated by its successful execution in standard reaction vessels without the need for specialized pressure equipment. The reduction in solvent usage and waste generation aligns with green chemistry principles, facilitating easier compliance with increasingly strict environmental regulations. The ability to run the reaction under air or nitrogen with simple workup procedures simplifies the technology transfer from lab to plant. This ease of scale-up ensures that commercial quantities can be produced rapidly to meet market demand without compromising on quality or safety standards.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and technical specifications outlined in the patent documentation. Understanding these details is essential for R&D teams evaluating the feasibility of integrating this route into their existing manufacturing pipelines. The information provided here serves as a foundational guide for assessing the compatibility of this method with specific project requirements.
Q: What are the primary advantages of this Rhodium-catalyzed method over traditional synthesis?
A: This method utilizes a one-pot tandem reaction that simultaneously constructs hexabasic nitrogen and pentabasic oxygen heterocycles, significantly simplifying the process compared to multi-step traditional methods which often require harsh conditions and difficult-to-obtain raw materials.
Q: Is this synthesis method suitable for large-scale industrial production?
A: Yes, the patent explicitly states the method is suitable for industrial production due to its simple operation, mild reaction conditions (80-120 °C), wide substrate applicability, and excellent regioselectivity, which facilitates easier purification and scale-up.
Q: What types of substrates are compatible with this catalytic system?
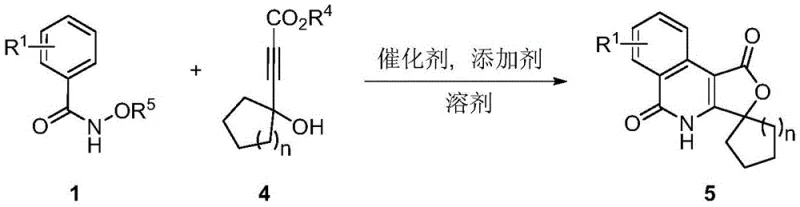
A: The system demonstrates broad substrate tolerance, accepting N-alkoxy aryl formamides with various substituents (hydrogen, halogens, alkyl, methoxy, phenyl) and 4-hydroxy-2-alkynoates with different alkyl or phenyl groups, including cyclic variants.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Furanone Isoquinolinone Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic value of advanced synthetic methodologies like the rhodium-catalyzed route described in CN108148070B for developing next-generation therapeutics. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from benchtop discovery to full-scale manufacturing is seamless and efficient. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of furanone isoquinolinone intermediates meets the highest industry standards. Our commitment to technical excellence allows us to navigate the complexities of C-H activation chemistry, delivering consistent quality even for challenging molecular architectures.
We invite you to collaborate with us to leverage this cutting-edge technology for your specific drug development programs. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your volume requirements. We are prepared to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our optimized processes can accelerate your timelines and reduce your overall development costs. Let us be your partner in turning innovative chemical concepts into commercial reality.
