Advanced Green Synthesis of Sulfamoylcarboxylic Acid Derivatives for Pharmaceutical Applications
Introduction to Advanced Sulfamoylcarboxylic Acid Synthesis
The pharmaceutical industry continuously seeks robust and scalable synthetic routes for complex intermediates, particularly those serving as precursors for potent therapeutic agents like NPY Y5 receptor antagonists. Patent CN101146768B, published in early 2012, introduces a groundbreaking methodology for the preparation of sulfamoylcarboxylic acid derivatives, addressing critical bottlenecks in traditional synthesis. This technology represents a significant leap forward in process chemistry, shifting away from hazardous chlorinated solvents towards a greener, toluene-based system enhanced by specific additives. For R&D directors and procurement specialists, understanding this shift is vital, as it directly impacts the purity profile, cost structure, and supply chain reliability of high-purity pharmaceutical intermediates. The innovation lies not just in the chemical transformation but in the engineering of the reaction medium to maximize efficiency while minimizing environmental footprint.
At the core of this invention is the realization that solubility and reactivity can be decoupled through the strategic use of co-solvents or additives. Traditional methods often struggled with the heterogeneity of reactants, leading to inconsistent batch-to-batch performance. By introducing controlled amounts of water or lower alcohols into a toluene matrix, the inventors achieved a stable reaction environment that supports high-yield coupling and subsequent oxidation steps. This approach eliminates the need for tedious purification techniques like column chromatography, which are notoriously difficult to scale. Consequently, this patent provides a blueprint for the commercial scale-up of complex pharmaceutical intermediates, offering a pathway to reduce lead time for high-purity compounds while adhering to stringent global environmental regulations regarding solvent use and waste disposal.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior art methods, such as those described in WO01/37826 and WO2003/076374, relied heavily on dichloromethane (DCM) or ethyl acetate as primary solvents. The use of DCM presents severe challenges in modern manufacturing due to its classification as a hazardous air pollutant and the increasing regulatory pressure to eliminate chlorinated solvents from pharmaceutical processes. Furthermore, these legacy routes often necessitated the isolation of intermediates via column chromatography to achieve acceptable purity levels. Chromatography is a major bottleneck in industrial chemistry; it is labor-intensive, consumes vast quantities of silica gel and solvents, and significantly drives up the cost of goods sold (COGS). Additionally, previous methods reported overall yields below 70%, largely due to losses during isomer conversion and purification steps. The reliance on tetrahydrofuran (THF) in some variations also introduced safety concerns regarding peroxide formation and solvent recovery, complicating the operational safety profile for large-scale production facilities.
The Novel Approach
The novel approach detailed in CN101146768B fundamentally reengineers the solvent system by utilizing toluene, a commodity chemical with a well-established supply chain and favorable safety profile. The breakthrough is the addition of specific additives, such as water, methanol, or isopropanol, which surprisingly enhance the reaction kinetics despite the low solubility of the starting amine in toluene. This method allows for a continuous, telescoped process where the coupling product can be oxidized directly without isolation, drastically simplifying the workflow. As illustrated in the reaction scheme below, the process flows seamlessly from the initial amine coupling to the final sulfone formation.
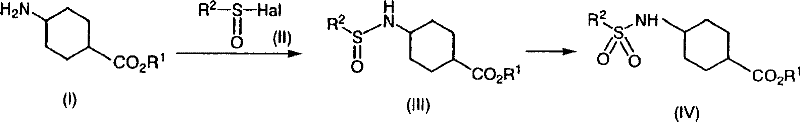
This streamlined workflow results in yields approaching 90-95%, a substantial improvement over the sub-70% yields of conventional methods. By avoiding chromatography and utilizing a single primary solvent system, the process significantly reduces waste generation and solvent consumption. For a reliable pharmaceutical intermediate supplier, adopting this methodology translates to a more competitive pricing structure and a more resilient supply chain, as it removes dependency on restricted solvents and complex purification infrastructure. The ability to run the reaction at mild temperatures (0°C to 50°C) further enhances energy efficiency and operational safety, making it an ideal candidate for cost reduction in API manufacturing.
Mechanistic Insights into Additive-Promoted Coupling and Oxidation
The mechanistic success of this synthesis hinges on the interplay between the sulfinyl chloride electrophile and the cyclohexylamine nucleophile within a biphasic or semi-heterogeneous toluene system. In the absence of additives, the reaction suffers from poor mass transfer and potential hydrolysis of the sensitive sulfinyl chloride by trace moisture. However, the deliberate addition of stoichiometric amounts of water or alcohol appears to create a micro-environment that facilitates proton transfer and stabilizes the transition state. The additive likely acts to solvate the amine hydrochloride salt, increasing its effective concentration at the reaction interface without dissolving it completely, thereby maintaining a controlled release of the free amine for coupling. This prevents the exothermic runaway often seen in sulfonylation reactions and minimizes side reactions such as over-sulfonylation or ester hydrolysis during the coupling phase.
Following the coupling, the oxidation of the sulfinyl group to the sulfonyl group is achieved using hydrogen peroxide, often catalyzed by tungstate species. This step is critical for establishing the final pharmacophore required for NPY Y5 receptor antagonism. The patent highlights that performing hydrolysis of the ester group prior to oxidation (Method B) can be advantageous for certain substrates to prevent acid-catalyzed decomposition. The scheme below details this alternative pathway where the ester is hydrolyzed to the acid before the final oxidation step, showcasing the versatility of the platform.
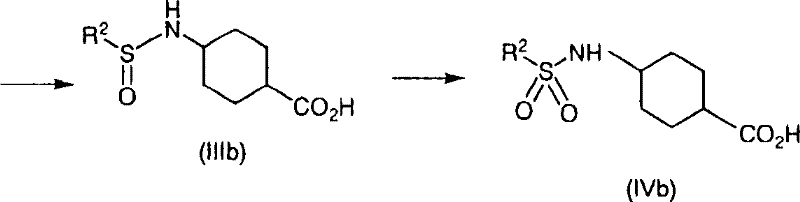
Impurity control is inherently built into this mechanism. By avoiding strong acidic conditions during the intermediate stages and utilizing mild oxidative conditions, the formation of degradation byproducts is minimized. The crystallization of the final product directly from the reaction mixture upon pH adjustment serves as a powerful purification step, effectively rejecting soluble impurities and unreacted starting materials. This "crystallization-driven purity" is a hallmark of robust process design, ensuring that the final high-purity sulfonamide intermediates meet strict specifications without the need for recrystallization or chromatographic polishing. For R&D teams, this implies a cleaner impurity profile, simplifying the regulatory filing process and reducing the risk of genotoxic impurity carryover.
How to Synthesize Sulfamoylcarboxylic Acid Derivatives Efficiently
Implementing this synthesis requires precise control over reaction parameters, particularly temperature and additive ratios. The process begins with the suspension of the amine salt in toluene, followed by the controlled addition of the base and the sulfinyl chloride reagent. The choice of additive—whether water for cost efficiency or isopropanol for solubility tuning—depends on the specific substrate properties. Once the coupling is complete, the reaction mixture can be directly subjected to oxidative conditions or hydrolysis depending on the desired final functional group. The detailed standardized synthesis steps for this high-efficiency route are outlined in the guide below.
- React 4-aminocyclohexanecarboxylate with tert-butylsulfinyl chloride in toluene using water or alcohol as an additive.
- Optionally hydrolyze the ester intermediate to the carboxylic acid form using aqueous base before oxidation.
- Oxidize the sulfinyl group to the sulfonyl group using hydrogen peroxide and a tungsten catalyst to yield the final product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented methodology offers transformative economic and logistical benefits. The primary driver for cost optimization is the elimination of dichloromethane and the associated waste treatment costs. Chlorinated solvents require specialized incineration or recovery systems, which represent a significant operational expense. By switching to toluene, a solvent with a mature recycling infrastructure and lower regulatory burden, manufacturers can achieve substantial cost savings in waste management. Furthermore, the drastic increase in yield from roughly 50-70% to over 90% means that less raw material is required to produce the same amount of active intermediate. This material efficiency directly lowers the variable cost per kilogram, enhancing margin potential in a competitive market.
- Cost Reduction in Manufacturing: The removal of column chromatography is perhaps the most significant financial advantage. Chromatography is not only expensive in terms of consumables like silica gel but also limits throughput due to long cycle times. By replacing this with a crystallization-based isolation, the process throughput is vastly increased, and capital expenditure on purification equipment is reduced. Additionally, the use of commodity reagents like hydrogen peroxide and sodium tungstate instead of exotic oxidants further drives down the raw material costs. These factors combine to create a leaner, more cost-effective manufacturing process that is resilient to raw material price fluctuations.
- Enhanced Supply Chain Reliability: Reliance on restricted solvents like DCM poses a supply risk, as availability can be impacted by environmental policy changes. Toluene and simple alcohols are produced in massive volumes globally, ensuring a stable and secure supply chain. The robustness of the reaction against batch-to-batch variability in starting material crystallinity also ensures consistent delivery schedules. Suppliers utilizing this method can guarantee on-time delivery without the risk of production delays caused by failed purification steps or solvent shortages, providing peace of mind to downstream API manufacturers.
- Scalability and Environmental Compliance: The process is inherently scalable, having been designed with industrial application in mind. The ability to telescope multiple steps without isolation reduces the number of unit operations, minimizing hold-up volume and processing time. From an environmental perspective, the reduction in solvent usage and the elimination of hazardous chlorinated waste align with Green Chemistry principles. This compliance facilitates easier permitting for new manufacturing lines and enhances the corporate sustainability profile, which is increasingly important for partnerships with major multinational pharmaceutical companies focused on ESG goals.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and comparative examples provided in the patent documentation, ensuring accuracy and relevance for technical decision-makers. Understanding these nuances is crucial for evaluating the feasibility of integrating this route into existing production portfolios.
Q: Why is toluene preferred over dichloromethane in this synthesis?
A: Toluene is preferred because dichloromethane is environmentally restricted and requires complex waste treatment. The patented method demonstrates that toluene, when combined with specific additives like water or alcohols, achieves superior yields without the regulatory and disposal burdens associated with chlorinated solvents.
Q: What is the role of additives like water or isopropanol in the reaction?
A: Additives such as water or isopropanol act as phase-transfer promoters or solubility enhancers. They stabilize the reaction interface between the solid amine salt and the organic solvent, preventing the decomposition of the sensitive sulfinyl chloride reagent and ensuring consistent high yields across different batches.
Q: Can this process be scaled for commercial API production?
A: Yes, the process is designed for industrial scalability. It eliminates the need for column chromatography, allows for continuous operation without isolating intermediates, and uses commodity chemicals, making it highly suitable for large-scale commercial manufacturing of pharmaceutical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Sulfamoylcarboxylic Acid Derivative Supplier
The technological advancements described in CN101146768B underscore the complexity and precision required to manufacture high-value pharmaceutical intermediates. At NINGBO INNO PHARMCHEM, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory processes are successfully translated into robust industrial realities. Our commitment to quality is backed by stringent purity specifications and rigorous QC labs equipped with state-of-the-art analytical instrumentation. We understand that the consistency of intermediates like sulfamoylcarboxylic acid derivatives is paramount for the efficacy and safety of the final drug product, and our processes are validated to deliver this consistency reliably.
We invite you to collaborate with us to leverage these advanced synthetic routes for your pipeline. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements. By partnering with us, you gain access to optimized manufacturing protocols that reduce lead time for high-purity intermediates while ensuring supply continuity. Please contact us to request specific COA data and route feasibility assessments for your upcoming projects, and let us demonstrate how our technical expertise can drive value for your organization.
