Scalable Production of 2,3,5-Tribenzyloxy-D-ribose Acid-1,4-lactone via Green Oxidation
Scalable Production of 2,3,5-Tribenzyloxy-D-ribose Acid-1,4-lactone via Green Oxidation
The pharmaceutical industry is constantly seeking robust, scalable, and environmentally sustainable pathways for the synthesis of critical antiviral intermediates. Patent CN111253348A introduces a groundbreaking methodology for the preparation of 2,3,5-tribenzyloxy-D-ribose acid-1,4-lactone, a pivotal building block in the synthesis of Remdesivir and related nucleoside analogs. This invention fundamentally re-engineers the oxidation step of the synthetic route, transitioning away from hazardous, waste-intensive protocols toward a catalytic system utilizing sodium bromide and TEMPO with sodium hypochlorite as the terminal oxidant. For R&D directors and procurement strategists, this shift represents not merely a chemical optimization but a strategic supply chain enhancement, offering a reliable pharmaceutical intermediates supplier pathway that mitigates regulatory risks associated with volatile sulfur compounds and heavy metal waste. The technical depth of this patent lies in its ability to maintain high stereochemical integrity while drastically simplifying downstream purification, a balance that is often elusive in complex carbohydrate chemistry.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the oxidation of protected ribose derivatives to their corresponding lactones has relied heavily on activated DMSO protocols, such as the Swern oxidation or variations using acetic anhydride, as well as chromium-based reagents like PCC. As illustrated in the prior art reaction scheme, these conventional methods present severe bottlenecks for commercial manufacturing. The use of DMSO and acetic anhydride inevitably generates dimethyl sulfide, a compound with an intensely unpleasant odor that requires specialized scrubbing systems and complicates facility operations. Furthermore, these reactions often produce high concentrations of acidic wastewater, necessitating costly neutralization and treatment processes before discharge. From a quality perspective, the harsh conditions can lead to the formation of stubborn impurities, specifically the ethyl ester derivative (Impurity 6) and the ring-opened hydrolysis product (Impurity 7), which are difficult to remove without extensive chromatographic purification.
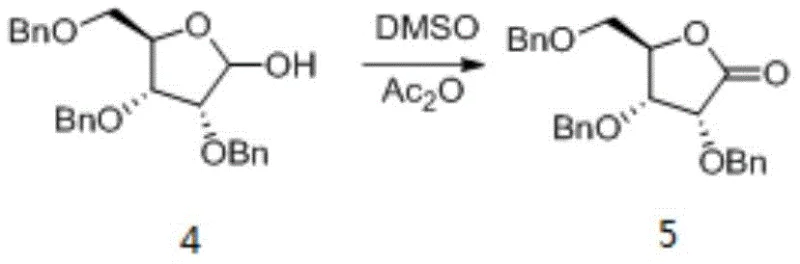
The Novel Approach
The novel approach disclosed in the patent circumvents these issues by employing a biphasic oxidation system driven by the TEMPO/NaBr catalytic cycle. This method operates under significantly milder conditions, typically between 0°C and 5°C, which preserves the acid-sensitive lactone ring and prevents the hydrolysis that leads to ring-opening impurities. By replacing stoichiometric oxidants with catalytic amounts of TEMPO and inexpensive sodium hypochlorite, the process eliminates the generation of dimethyl sulfide entirely. Moreover, the workup procedure is revolutionized; instead of relying on silica gel chromatography, which is impractical for multi-kilogram batches due to solvent consumption and throughput limitations, the new method utilizes a precise crystallization protocol. This shift from chromatography to crystallization is a hallmark of a mature, industrial-grade process, directly addressing the needs of a reliable pharmaceutical intermediates supplier aiming for high throughput and reduced operational expenditure.
Mechanistic Insights into TEMPO-Mediated Catalytic Oxidation
The core of this technological advancement lies in the efficient regeneration of the active oxoammonium species via the sodium bromide co-catalyst. In this mechanism, TEMPO acts as the primary oxidant for the alcohol substrate, converting the hydroxyl group at the C1 position into the carbonyl functionality required for lactonization. The reduced hydroxylamine form of TEMPO is subsequently re-oxidized by hypochlorous acid, which is generated in situ from sodium hypochlorite. The presence of sodium bromide accelerates this cycle by forming reactive bromine species that facilitate the turnover of the catalyst. This catalytic loop ensures that the oxidation potential remains controlled and selective, targeting the primary alcohol without over-oxidizing or degrading the sensitive benzyl protecting groups on the ribose scaffold. The choice of dichloromethane as the solvent is critical, as it provides the necessary solubility for the organic substrate while allowing for effective phase transfer with the aqueous hypochlorite solution.

Impurity control is intrinsically linked to the mildness of this mechanistic pathway. Traditional acidic or high-temperature oxidation methods often promote the hydrolysis of the lactone ring, leading to the open-chain acid form (Impurity 7), or facilitate esterification if acetic anhydride is present (Impurity 6). The TEMPO-mediated system operates at near-neutral pH levels during the reaction phase, significantly reducing the kinetic drive for these side reactions. Furthermore, the subsequent purification via crystallization leverages the solubility differences between the target lactone and any trace unreacted starting material or minor byproducts. By optimizing the solvent mixture of methyl tert-butyl ether and n-hexane, the process achieves a high degree of purity without the need for preparative HPLC or flash chromatography, ensuring that the final product meets the stringent purity specifications required for GMP manufacturing of antiviral APIs.
How to Synthesize 2,3,5-Tribenzyloxy-D-ribose Acid-1,4-lactone Efficiently
The implementation of this synthesis route requires careful attention to temperature control and reagent addition rates to maximize yield and minimize side reactions. The patent details a robust protocol where the catalyst loading and oxidant addition are finely tuned to ensure complete conversion of Intermediate 4 while maintaining the structural integrity of the molecule. The following guide outlines the standardized operational parameters derived from the patent examples, designed to assist process chemists in replicating this high-efficiency transformation in a pilot or production setting. For detailed step-by-step instructions including exact masses and timing, please refer to the structured guide below.
- Dissolve intermediate 4 in dichloromethane and add catalytic amounts of sodium bromide and TEMPO under stirring.
- Maintain temperature at 0-5°C and slowly add 10% aqueous sodium hypochlorite solution dropwise to initiate oxidation.
- After reaction completion, separate layers, wash organic phase, concentrate, and induce crystallization using methyl tert-butyl ether and n-hexane.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this TEMPO-catalyzed process offers substantial strategic benefits beyond mere chemical elegance. The elimination of silica gel chromatography is perhaps the most significant cost driver, as chromatographic purification is notoriously solvent-intensive and labor-heavy, often becoming the bottleneck in scaling up fine chemical production. By switching to a crystallization-based purification, the process drastically reduces solvent consumption and waste disposal costs, directly contributing to cost reduction in pharmaceutical intermediates manufacturing. Additionally, the removal of malodorous byproducts like dimethyl sulfide simplifies facility compliance with environmental regulations, reducing the need for specialized abatement equipment and lowering the overall operational overhead associated with safety and environmental health protocols.
- Cost Reduction in Manufacturing: The replacement of expensive stoichiometric oxidants such as Dess-Martin periodinane or IBX with commodity chemicals like sodium hypochlorite and sodium bromide results in a dramatic decrease in raw material costs. Furthermore, the avoidance of acetic anhydride eliminates the formation of ethyl ester impurities, thereby increasing the overall yield of the desired product and reducing the loss of valuable starting materials. The simplified workup procedure also reduces labor hours and utility consumption, as there is no need for the extensive solvent evaporation and column packing required in traditional methods, leading to substantial cost savings across the production lifecycle.
- Enhanced Supply Chain Reliability: The reagents utilized in this novel protocol, including sodium hypochlorite, sodium bromide, and TEMPO, are globally available commodity chemicals with stable supply chains, unlike specialized oxidants that may face availability fluctuations. This reliability ensures consistent production schedules and reduces the risk of delays caused by raw material shortages. The robustness of the reaction conditions also means that the process is less sensitive to minor variations in reagent quality, providing a more forgiving and stable manufacturing window that supports continuous supply to downstream API manufacturers without interruption.
- Scalability and Environmental Compliance: The design of this process is inherently scalable, moving away from batch-limited chromatography to continuous-friendly crystallization techniques. This facilitates the commercial scale-up of complex pharmaceutical intermediates from kilogram to metric-ton scales without the need for proportional increases in purification infrastructure. Environmentally, the process aligns with green chemistry principles by minimizing hazardous waste generation and avoiding the use of heavy metals like chromium, thus simplifying waste treatment and ensuring long-term sustainability and compliance with increasingly strict global environmental standards.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this oxidation technology. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation, offering clarity on why this method represents a superior choice for modern pharmaceutical synthesis. Understanding these nuances is essential for technical teams evaluating process transfers or looking to optimize existing supply chains for nucleoside analog production.
Q: Why is the TEMPO/NaBr catalytic system superior to traditional DMSO-based oxidation?
A: The TEMPO/NaBr system operates under mild conditions (0-5°C) avoiding the generation of malodorous dimethyl sulfide and high-concentration acidic wastewater associated with DMSO/Ac2O methods, while eliminating the need for silica gel chromatography.
Q: How does this process control specific impurities like ethyl ester derivative 6?
A: By avoiding acetic anhydride and harsh acidic conditions, the new method prevents the formation of ethyl ester impurity 6 and ring-opening impurity 7, ensuring higher purity suitable for downstream API synthesis.
Q: Is this method suitable for large-scale commercial manufacturing?
A: Yes, the replacement of column chromatography with a simple crystallization step using methyl tert-butyl ether and n-hexane significantly enhances scalability and reduces solvent waste, making it ideal for metric-ton production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2,3,5-Tribenzyloxy-D-ribose Acid-1,4-lactone Supplier
At NINGBO INNO PHARMCHEM, we recognize that the efficiency of your downstream API synthesis depends heavily on the quality and consistency of your starting materials. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the advanced TEMPO-catalyzed oxidation process described in CN111253348A can be seamlessly translated from the laboratory to full-scale manufacturing. Our rigorous QC labs and stringent purity specifications guarantee that every batch of 2,3,5-tribenzyloxy-D-ribose acid-1,4-lactone meets the highest industry standards, free from the critical impurities that often plague conventional synthesis routes.
We invite you to collaborate with our technical procurement team to explore how this optimized route can enhance your project economics. By requesting a Customized Cost-Saving Analysis, you can gain a detailed understanding of the potential reductions in waste disposal and raw material expenses specific to your volume requirements. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, ensuring that your supply chain is built on a foundation of chemical excellence and operational reliability.
