Advanced Synthesis of 4-Bromo-2-Chloro-6-Methoxypyridine for Commercial Pharmaceutical Manufacturing
Introduction to Novel Synthetic Pathways
The pharmaceutical industry constantly seeks robust and scalable routes for heterocyclic building blocks, and the synthesis of 4-bromo-2-chloro-6-methoxypyridine represents a critical advancement in this domain. As disclosed in patent CN111072554A, a groundbreaking two-step methodology has been established that transforms inexpensive starting materials into high-value intermediates with exceptional efficiency. This process begins with the selective methoxy substitution of 4-amino-2,6-dichloropyridine, followed by a refined diazotization-bromination sequence to install the bromine atom at the 4-position. The strategic innovation lies not just in the reaction steps themselves, but in the precise control of catalytic systems and solvent environments that dictate product purity and yield.
For R&D directors and process chemists, the significance of this patent extends beyond mere chemical transformation; it offers a solution to long-standing selectivity issues that have plagued previous attempts at synthesizing this scaffold. By utilizing specific iodide-based catalysts in the first step and a mixed acid system in the second, the inventors have successfully navigated the complex reactivity of the pyridine ring. This approach ensures that the final product, a versatile intermediate for drug discovery, is obtained with minimal impurities, thereby reducing the burden on downstream purification processes. The overall reaction scheme is depicted below, illustrating the seamless transition from the dichloro precursor to the target bromo-methoxy derivative.
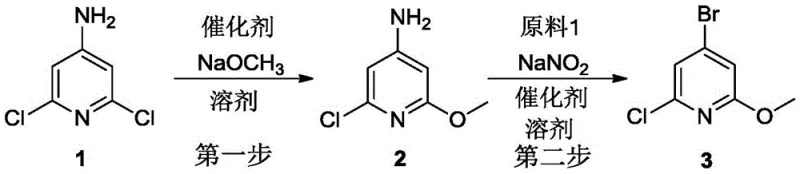
Furthermore, the commercial implications of this technology are profound for supply chain stakeholders. The reliance on commodity chemicals such as sodium methoxide, sodium nitrite, and sodium bromide ensures that raw material costs remain low and supply continuity is high. Unlike routes that depend on exotic reagents or extreme conditions that are difficult to maintain in large reactors, this method operates under relatively mild and controllable parameters. This makes it an ideal candidate for reliable pharmaceutical intermediate supplier networks aiming to secure long-term contracts for API production without the risk of batch-to-batch variability.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of methoxy-substituted pyridines has been fraught with challenges regarding regioselectivity and byproduct formation. Prior art, such as the route illustrated in literature Scheme 1, typically relies on forcing conditions to drive the substitution reaction. In these conventional methods, 4-amino-2,6-dichloropyridine is subjected to high temperature and high pressure in the presence of sodium methoxide. While this might seem straightforward, the lack of a selective catalyst often leads to over-substitution, resulting in a mixture of the desired mono-methoxy product and the unwanted 4-amino-2,6-dimethoxypyridine byproduct.
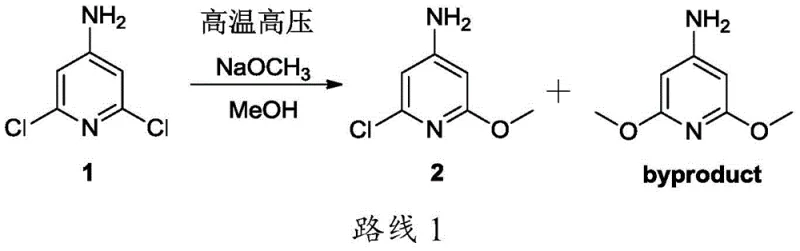
The separation of these byproducts is notoriously difficult due to their similar polarity and physicochemical properties, often requiring extensive column chromatography which is impractical for industrial scale-up. Additionally, attempting to mitigate this by reducing the amount of base or lowering the temperature often results in incomplete conversion of the starting material, leaving significant amounts of unreacted dichloro compound. This creates a purification bottleneck where the cost of isolating the pure intermediate skyrockets, rendering the process economically unviable for commercial manufacturing. Furthermore, traditional diazotization methods for the second step often utilize pure hydrobromic acid as both solvent and bromine source, generating massive volumes of acidic wastewater that pose severe environmental compliance hurdles.
The Novel Approach
The methodology presented in CN111072554A fundamentally alters the reaction landscape by introducing a catalytic system that enhances nucleophilic selectivity. Instead of relying solely on thermal energy, the process incorporates iodide salts, such as sodium iodide or tetrabutylammonium iodide, which act as potent nucleophilic catalysts. This modification allows the methoxy substitution to proceed efficiently at moderate temperatures around 100°C, effectively suppressing the formation of the dimethoxy byproduct. The result is a reaction profile where the desired 4-amino-2-chloro-6-methoxypyridine is formed with high specificity, simplifying the workup to a mere filtration or crystallization step.
In the subsequent bromination step, the novel approach replaces the environmentally hazardous pure hydrobromic acid solvent with a mixture of sulfuric acid and water. This shift is critical for green chemistry compliance. By using sodium bromide as the bromine source in the presence of a catalytic amount of cuprous bromide, the reaction minimizes the generation of free elemental bromine, which is a common cause of poly-bromination side reactions. This controlled environment ensures that the bromine atom is installed exclusively at the 4-position, preserving the integrity of the chloro and methoxy groups. The combination of these innovations results in a process that is not only higher yielding but also significantly safer and more sustainable for large-scale operations.
Mechanistic Insights into Catalytic Methoxy Substitution and Diazotization
To fully appreciate the technical superiority of this synthesis, one must delve into the mechanistic nuances of the iodide-catalyzed nucleophilic aromatic substitution. In the absence of a catalyst, the displacement of the chlorine atom at the 2- or 6-position of the pyridine ring by methoxide is driven by the electron-withdrawing nature of the ring nitrogen. However, without guidance, the reaction lacks discrimination, leading to double substitution. The introduction of iodide ions changes the kinetic pathway; iodide is a superior nucleophile compared to methoxide and can rapidly displace the chlorine to form a transient iodo-intermediate. This iodo-species is highly reactive towards methoxide, facilitating a rapid second substitution that releases the iodide back into the cycle. This catalytic loop accelerates the formation of the mono-substituted product while the specific reaction conditions prevent the second chlorine from reacting, likely due to steric or electronic deactivation after the first substitution.
Regarding the second step, the mechanism involves the classic formation of a diazonium salt followed by a Sandmeyer-type replacement. The critical innovation here is the management of the brominating species. In traditional setups, excess HBr can lead to electrophilic aromatic substitution on the activated pyridine ring, causing unwanted poly-bromination. By utilizing a sulfuric acid matrix with stoichiometric sodium bromide and catalytic CuBr, the concentration of reactive bromine species is tightly regulated. The copper catalyst facilitates the single-electron transfer necessary for the radical substitution mechanism, ensuring that the diazonium group is replaced by bromine before it can decompose or react elsewhere. This precise control is what allows the process to achieve high purity without the need for complex chromatographic purification, a key factor for maintaining cost efficiency in API intermediate manufacturing.
How to Synthesize 4-Bromo-2-Chloro-6-Methoxypyridine Efficiently
Implementing this synthesis in a pilot or production plant requires strict adherence to the optimized parameters defined in the patent examples. The process is divided into two distinct stages, each with specific temperature and molar ratio requirements to ensure maximum yield and safety. The first stage focuses on the selective substitution, while the second handles the sensitive diazotization. Operators must monitor reaction progress via HPLC or TLC to determine the exact endpoint, particularly in the diazotization step where temperature control is paramount to prevent diazonium salt decomposition. The following guide outlines the standardized operational procedure derived from the most successful experimental examples provided in the intellectual property documentation.
- Perform methoxy substitution on 4-amino-2,6-dichloropyridine using sodium methoxide and an iodide catalyst in methanol at 100°C.
- Conduct diazotization of the intermediate using sodium nitrite and sulfuric acid at low temperatures (-5 to 0°C).
- Complete bromination by adding sodium bromide and catalytic cuprous bromide, followed by neutralization and recrystallization.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthetic route offers tangible benefits that extend far beyond the laboratory bench. The primary advantage lies in the drastic simplification of the supply chain for raw materials. By utilizing commodity chemicals like sodium methoxide and sodium bromide, manufacturers can avoid the volatility associated with sourcing specialized or hazardous reagents. This stability translates directly into cost reduction in pharmaceutical intermediate manufacturing, as the reliance on expensive, custom-synthesized catalysts is eliminated. The use of tetrabutylammonium iodide, while a specialty chemical, is required only in catalytic amounts, meaning its impact on the overall bill of materials is negligible compared to the savings gained from improved yield and reduced waste disposal costs.
- Cost Reduction in Manufacturing: The elimination of high-pressure equipment requirements for the first step significantly lowers capital expenditure (CAPEX) for production facilities. Conventional methods often necessitate expensive autoclaves capable of withstanding extreme pressures, whereas this novel method operates effectively in standard heated reactors. Furthermore, the high selectivity of the reaction means that less starting material is wasted on byproducts, directly improving the atom economy. The ability to isolate the intermediate via simple filtration or crystallization rather than energy-intensive distillation or chromatography further drives down operational expenditures (OPEX), making the final product highly competitive in the global market.
- Enhanced Supply Chain Reliability: The robustness of this chemical process ensures consistent batch-to-batch quality, which is critical for maintaining uninterrupted API production schedules. Because the reaction conditions are mild and the reagents are stable, the risk of batch failure due to equipment malfunction or reagent degradation is minimized. This reliability allows supply chain planners to forecast inventory needs with greater accuracy and reduce safety stock levels. Additionally, the simplified purification process shortens the overall cycle time from raw material intake to finished goods, enabling faster response times to market demand fluctuations and reducing lead time for high-purity pharmaceutical intermediates.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, this route is vastly superior to legacy methods. The replacement of pure hydrobromic acid with a sulfuric acid/water mixture reduces the corrosivity of the waste stream and simplifies neutralization procedures. The reduction in heavy metal usage, specifically limiting cuprous bromide to catalytic quantities, eases the burden on wastewater treatment facilities and ensures compliance with increasingly stringent environmental regulations. This 'green' profile not only mitigates regulatory risk but also aligns with the sustainability goals of major pharmaceutical clients, enhancing the commercial appeal of the supplier.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity on how this method resolves specific industry pain points. Understanding these details is essential for technical teams evaluating the feasibility of integrating this route into their existing manufacturing portfolios.
Q: How does the new catalyst system improve selectivity in the first step?
A: The addition of iodide catalysts like tetrabutylammonium iodide facilitates the nucleophilic substitution, preventing the formation of the dimethoxy byproduct often seen in high-pressure literature methods.
Q: What environmental advantages does this synthesis route offer?
A: By replacing pure hydrobromic acid with a sulfuric acid/water mixture and using sodium bromide instead of stoichiometric cuprous bromide, the process significantly reduces acidic wastewater and heavy metal waste.
Q: Is this process suitable for large-scale production?
A: Yes, the patent demonstrates successful scale-up from gram to kilogram levels with high yields (up to 91.9% in Step 1 and 83.6% in Step 2) and simple purification via recrystallization.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4-Bromo-2-Chloro-6-Methoxypyridine Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from a patented laboratory method to a commercial reality requires more than just chemical knowledge; it demands engineering excellence and rigorous quality assurance. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the promising yields reported in CN111072554A can be replicated reliably on an industrial scale. We understand that for complex heterocyclic intermediates, maintaining stringent purity specifications is non-negotiable, which is why our rigorous QC labs employ advanced analytical techniques to verify every batch against the highest international standards.
We invite potential partners to engage with our technical procurement team to discuss how this optimized synthesis can benefit your specific supply chain. Whether you require a Customized Cost-Saving Analysis to compare this route against your current incumbent suppliers or need specific COA data to validate the impurity profile, we are prepared to provide comprehensive support. By leveraging our expertise in route feasibility assessments, we can help you secure a stable, cost-effective supply of this critical building block, empowering your drug development programs with the high-quality materials they deserve.
