Advanced Metal-Free Synthesis of C2-Phosphono Indole Compounds for Commercial Scale-Up
The pharmaceutical and fine chemical industries are constantly seeking more efficient and sustainable pathways to access complex heterocyclic scaffolds, particularly those containing phosphorus motifs which are critical for biological activity. Patent CN110256493B introduces a groundbreaking methodology for the preparation of C2-phosphono indole compounds, addressing significant bottlenecks in current synthetic strategies. This technology leverages a Brønsted acid-catalyzed coupling between 3-(3-indolyl)-3-hydroxy-2-oxindoles and H-phosphine oxides, offering a robust alternative to traditional transition metal-mediated processes. By operating under air atmosphere and generating water as the sole byproduct, this invention aligns perfectly with modern green chemistry principles while delivering high-purity intermediates essential for drug discovery programs. The strategic elimination of heavy metal catalysts not only simplifies the purification workflow but also mitigates regulatory concerns regarding metal residues in active pharmaceutical ingredients.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
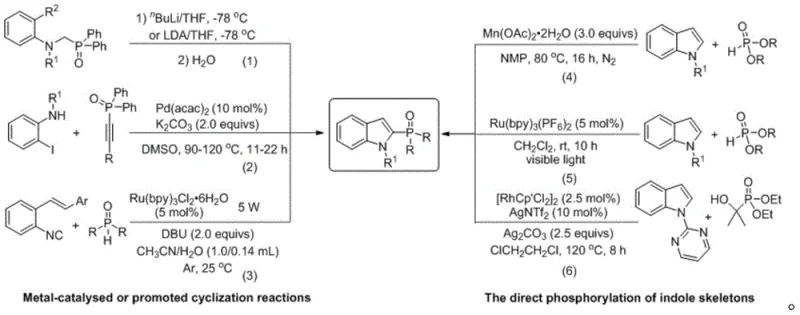
Historically, the construction of C2-phosphono indole skeletons has relied heavily on metal-catalyzed cyclization reactions or direct phosphorylation using expensive photosensitizers and transition metals. As illustrated in the comparative analysis of existing technologies, conventional routes often necessitate the use of palladium, ruthenium, or manganese salts, which drive up production costs and introduce complex downstream processing requirements. These traditional methods frequently demand harsh reaction conditions, ranging from cryogenic temperatures of -78°C to elevated heating up to 120°C, alongside the need for specialized ligands and inert gas protection. Furthermore, the reliance on stoichiometric amounts of metal salts often leads to significant metal contamination in the final product, necessitating additional scavenging steps that reduce overall yield and increase waste generation. Such limitations hinder the scalability and economic viability of producing these valuable intermediates for commercial applications.
The Novel Approach
In stark contrast to the cumbersome legacy techniques, the novel approach disclosed in CN110256493B utilizes a straightforward acid-catalyzed condensation that proceeds efficiently under mild thermal conditions. This method employs readily available Brønsted acids such as trifluoromethanesulfonic acid, camphorsulfonic acid, or p-toluenesulfonic acid to activate the hydroxyl group of the oxindole precursor, facilitating a dehydration event that generates a highly reactive electrophilic species. The subsequent nucleophilic attack by the H-phosphine oxide occurs smoothly without the need for external oxidants or light sources. This paradigm shift allows the reaction to be conducted in common organic solvents like dichloromethane, acetonitrile, or toluene at temperatures ranging from 0°C to 100°C. The operational simplicity, combined with the avoidance of precious metals, represents a substantial advancement in the synthesis of phosphorus-containing heterocycles, offering a cleaner and more cost-effective route for industrial manufacturing.
Mechanistic Insights into Acid-Catalyzed Dehydration and Addition
The core of this technological breakthrough lies in the unique reaction mechanism that bypasses the need for metal coordination complexes. Under acidic conditions, the tertiary alcohol moiety of the 3-(3-indolyl)-3-hydroxy-2-oxindole substrate undergoes protonation followed by the loss of a water molecule. This dehydration step generates a stabilized carbocation intermediate, which can resonate to form a vinylinium species, significantly enhancing its electrophilicity at the C2 position of the indole ring. The H-phosphine oxide, acting as a nucleophile, then engages in a 1,2-addition pathway to this activated intermediate. This mechanistic pathway is distinct from radical-based or oxidative coupling mechanisms seen in photocatalytic methods, providing greater control over regioselectivity and minimizing side reactions. The mild acidity ensures that sensitive functional groups on the aromatic rings remain intact, preserving the structural integrity required for downstream biological testing.
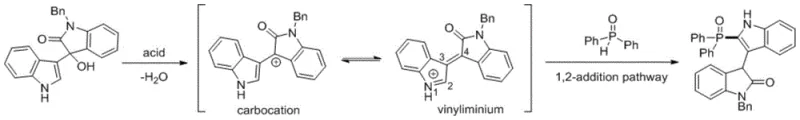
From an impurity control perspective, this mechanism offers distinct advantages by limiting the formation of metal-complexed byproducts or homocoupling products often associated with oxidative conditions. The reaction profile is clean, with the primary side product being water, which can be easily removed during the workup phase. The use of specific acids allows for fine-tuning the reaction rate; stronger acids like HOTf can accelerate the process at lower temperatures, while weaker organic acids like CSA provide a gentler activation suitable for more sensitive substrates. This tunability is crucial for process chemists aiming to optimize yield and purity profiles across a diverse library of analogues. Understanding this mechanistic nuance enables the precise selection of reaction parameters to maximize efficiency while maintaining the high quality standards demanded by the pharmaceutical sector.
How to Synthesize C2-Phosphono Indole Efficiently
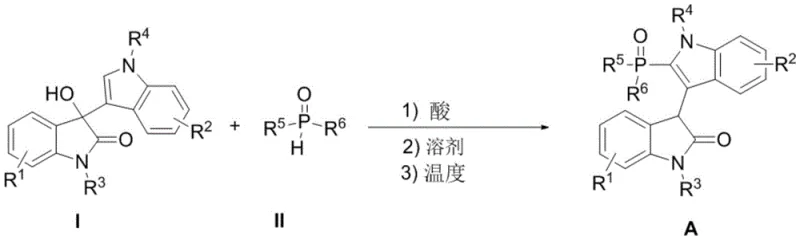
The practical implementation of this synthesis involves a simple one-pot procedure where the oxindole derivative and the phosphine oxide are combined in a solvent system with a catalytic amount of acid. The general reaction scheme demonstrates the versatility of this approach across various substitution patterns on both the indole and oxindole cores. Reaction monitoring is typically achieved via thin-layer chromatography (TLC), and upon completion, the product can be isolated through standard silica gel column chromatography. This streamlined workflow reduces the number of unit operations compared to multi-step metal-catalyzed sequences, directly translating to labor savings and reduced solvent consumption. The detailed standardized synthesis steps for specific derivatives are outlined in the guide below.
- Mix compound I (3-(3-indolyl)-3-hydroxy-2-oxindole derivative) and compound II (H-phosphine oxide) in a suitable organic solvent.
- Add a catalytic amount of Brønsted acid such as trifluoromethanesulfonic acid, camphorsulfonic acid, or p-toluenesulfonic acid.
- Stir the reaction mixture at temperatures between 0°C and 100°C until completion, then purify via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this metal-free synthesis protocol presents a compelling value proposition centered on cost stability and operational reliability. By eliminating the dependency on volatile precious metal markets for catalysts like palladium or ruthenium, manufacturers can achieve significant cost reduction in pharmaceutical intermediate manufacturing. The removal of metal scavenging resins and the associated filtration steps further simplifies the production line, reducing both capital expenditure on equipment and operational expenditure on consumables. Additionally, the use of commodity chemicals as catalysts ensures a stable supply chain that is less susceptible to geopolitical disruptions often affecting rare earth or precious metal supplies.
- Cost Reduction in Manufacturing: The exclusion of expensive transition metal catalysts and specialized ligands drastically lowers the raw material bill of materials. Furthermore, the simplified workup procedure, which avoids complex metal removal protocols, reduces solvent usage and waste disposal costs. This lean manufacturing approach enhances the overall profit margin for high-volume production runs without compromising product quality.
- Enhanced Supply Chain Reliability: Since the key reagents including the acid catalysts and phosphine oxides are commercially available in bulk quantities, the risk of supply bottlenecks is minimized. The robustness of the reaction under air atmosphere removes the need for specialized inert gas infrastructure, allowing for production in standard facilities. This flexibility ensures consistent delivery schedules and reduces lead time for high-purity pharmaceutical intermediates.
- Scalability and Environmental Compliance: The reaction generates water as the only byproduct, resulting in high atom economy and a significantly reduced environmental footprint. This green chemistry profile facilitates easier regulatory approval and compliance with increasingly stringent environmental regulations. The mild reaction conditions and simple purification methods make the process highly scalable from laboratory benchtop to multi-ton commercial production.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this acid-catalyzed phosphorylation technology. These insights are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing clarity on the method's versatility and operational parameters. Understanding these details helps stakeholders evaluate the feasibility of integrating this route into their existing manufacturing pipelines.
Q: What are the advantages of this acid-catalyzed method over traditional metal-catalyzed routes?
A: This method eliminates the need for expensive transition metal catalysts like palladium or ruthenium, thereby avoiding metal residue issues in the final product and significantly reducing raw material costs while operating under milder conditions.
Q: What is the atom economy of this phosphorylation reaction?
A: The reaction exhibits high atom economy because the only byproduct generated during the transformation is water, making the process environmentally friendly and simplifying waste treatment protocols.
Q: Can this method tolerate various functional groups on the indole scaffold?
A: Yes, the protocol demonstrates high compatibility with diverse functional groups including halogens, nitro groups, and alkyl substituents on both the oxindole and indole rings, allowing for broad substrate scope.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable C2-Phosphono Indole Supplier
As the global demand for specialized heterocyclic building blocks continues to rise, NINGBO INNO PHARMCHEM stands ready to support your development needs with our advanced synthesis capabilities. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet your volume requirements whether for clinical trials or full-scale market launch. Our commitment to quality is underpinned by stringent purity specifications and rigorous QC labs that verify every batch against the highest industry standards, guaranteeing the consistency and reliability of our C2-phosphono indole intermediates.
We invite you to collaborate with us to leverage this innovative metal-free technology for your next project. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific volume needs. We are prepared to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our optimized processes can enhance your supply chain efficiency and reduce overall project costs.
