Scalable Synthesis of Heptafluoroisobutyl Amide for Next-Gen Electronic Insulating Gases
The global transition towards environmentally sustainable electrical infrastructure has intensified the search for alternatives to sulfur hexafluoride (SF6), a potent greenhouse gas with a global warming potential approximately 23,900 times that of CO2. In this critical landscape, heptafluoroisobutyronitrile (C4 gas) has emerged as a leading candidate for high-voltage insulation due to its superior dielectric properties and significantly lower environmental impact. Central to the production of this advanced electronic gas is the key intermediate, heptafluoroisobutyl amide, which serves as the foundational building block for the nitrile functionality. Patent CN108440327B discloses a groundbreaking method for synthesizing this vital precursor from bis-(heptafluoroisopropyl)-ketone and ammonia, offering a pathway that addresses both the safety concerns and efficiency limitations of prior art technologies. This technical insight report analyzes the proprietary low-temperature ammonolysis process, highlighting its potential to redefine supply chain reliability for manufacturers of next-generation fluorinated electronic materials.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of fluorinated amides has been plagued by significant operational hazards and inefficient atom economy, particularly when relying on gaseous ammonia as the nitrogen source. Traditional protocols often involve bubbling ammonia gas directly into reaction vessels containing perfluorinated ketones, a process that inherently suffers from poor mass transfer and low absorption rates. Consequently, a substantial portion of the expensive ammonia reagent fails to participate in the chemical transformation and is instead vented directly into the atmosphere, creating severe environmental compliance issues and necessitating complex scrubbing systems. Furthermore, the exothermic nature of ammonolysis reactions, when uncontrolled by a liquid medium, can lead to thermal runaways that compromise product integrity and pose serious safety risks to plant personnel. These legacy methods typically report yields hovering around 67% to 74%, indicating significant material loss that drives up the cost of goods sold for downstream electronic gas producers.
The Novel Approach
The methodology outlined in patent CN108440327B represents a paradigm shift by replacing hazardous gaseous reagents with a controlled liquid-phase system using an ammonia methanol solution. By dissolving ammonia in methanol to create a 7 mol/L solution, the process ensures intimate contact between the nucleophile and the electrophilic carbonyl carbon of the bis-(heptafluoroisopropyl)-ketone, drastically improving reaction kinetics and selectivity. This liquid-liquid interaction allows for precise thermal management, keeping the reaction temperature strictly below 0°C to suppress unwanted side reactions such as over-ammonolysis or decomposition of the sensitive perfluoroalkyl chains. The result is a streamlined workflow that eliminates the need for high-pressure gas handling equipment and reduces the generation of hazardous waste streams, aligning perfectly with modern green chemistry principles. Moreover, the implementation of this optimized route has demonstrated the capability to push isolated yields significantly higher, reaching levels above 86% in pilot examples, which translates directly to enhanced profitability for industrial-scale operations.
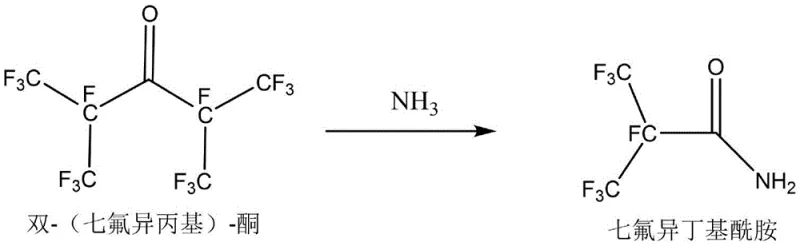
Mechanistic Insights into Low-Temperature Ammonolysis
The core chemical transformation involves the nucleophilic attack of ammonia on the highly electron-deficient carbonyl carbon of the perfluorinated ketone, facilitated by the strong electron-withdrawing nature of the surrounding heptafluoroisopropyl groups. In this specific substrate, the steric bulk and electronegativity of the fluorine atoms activate the carbonyl center towards nucleophilic addition-elimination, yet they also render the molecule susceptible to cleavage if conditions are too vigorous. The mechanism likely proceeds through a tetrahedral intermediate where the ammonia adds to the carbonyl, followed by the expulsion of a heptafluoroisopropyl anion or radical species, which is subsequently protonated to form heptafluoroisopropane as a byproduct. Maintaining the temperature gradient from -5°C to 0°C is critical because it stabilizes this transition state, preventing the degradation of the newly formed amide bond which could otherwise revert or decompose under higher thermal stress. This delicate balance ensures that the reaction stops selectively at the amide stage without progressing to further degradation products or polymeric tars that are common in uncontrolled fluorine chemistry.
Impurity control in this synthesis is achieved through a multi-stage purification strategy that leverages both physical and chemical separation techniques to ensure the high purity required for electronic grade applications. Following the reaction, the removal of the methanol solvent via rotary evaporation at moderate temperatures (30-45°C) prevents thermal shock to the crude product, which might otherwise trap solvent molecules within the crystal lattice. The subsequent addition of an adsorbent, specifically 200-400 mesh colorless silica gel or activated carbon, plays a pivotal role in scavenging trace colored impurities and residual acidic byproducts that could catalyze decomposition during storage. Finally, recrystallization from chloroform at controlled temperatures (35-40°C for dissolution followed by cooling) allows for the selective precipitation of the target heptafluoroisobutyl amide, leaving soluble impurities in the mother liquor. This rigorous purification protocol ensures that the final crystals are colorless and transparent, meeting the stringent spectral specifications required for downstream conversion into insulating gases.
How to Synthesize Heptafluoroisobutyl Amide Efficiently
The synthesis of heptafluoroisobutyl amide via this patented route requires strict adherence to temperature profiles and reagent ratios to maximize yield and safety. The process begins with the preparation of a standardized ammonia methanol solution, which is then dosed slowly into the ketone substrate to manage the exotherm effectively. Detailed standard operating procedures regarding stirring rates, addition times, and crystallization parameters are essential for reproducibility at scale. For the complete step-by-step technical guide including specific equipment setups and safety protocols, please refer to the standardized synthesis instructions provided below.
- Prepare a 7 mol/L ammonia methanol solution and slowly add it to bis-(heptafluoroisopropyl)-ketone at a molar ratio of 1.5: 1 while maintaining the temperature below 0°C.
- Maintain the reaction mixture at temperatures between -5°C and 0°C for approximately 6 hours total to ensure complete conversion to the pale yellow solution.
- Remove the solvent via rotary evaporation at 30-45°C, treat the crude product with silica gel adsorbent, and recrystallize using chloroform to obtain colorless crystals.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this novel synthesis route offers substantial strategic advantages by mitigating several critical bottlenecks associated with traditional fluorinated intermediate manufacturing. The shift from gaseous to liquid ammonia handling fundamentally alters the safety profile of the facility, removing the need for specialized high-pressure gas storage and delivery infrastructure, which in turn lowers capital expenditure and insurance premiums. By eliminating the venting of unreacted ammonia, the process not only adheres to increasingly strict environmental regulations regarding volatile organic compounds and greenhouse gases but also maximizes the utility of every kilogram of raw material purchased. This improvement in atom economy directly correlates to a reduction in the effective cost per unit of the active pharmaceutical or electronic ingredient, providing a competitive edge in price-sensitive markets. Furthermore, the simplified workup procedure involving standard rotary evaporation and filtration reduces the cycle time per batch, allowing for increased throughput without the need for additional reactor volume.
- Cost Reduction in Manufacturing: The elimination of expensive gas scrubbing systems and the reduction in raw material waste through higher conversion rates lead to significant operational cost savings. By utilizing a liquid-phase reagent system, the process avoids the losses associated with gas escape, ensuring that the stoichiometric ratio of ammonia to ketone is utilized much more efficiently than in conventional gas-liquid reactions. Additionally, the use of common solvents like methanol and chloroform, which are readily available and easily recycled, minimizes the dependency on exotic or costly specialty reagents. The overall simplification of the downstream processing, requiring only evaporation and recrystallization rather than complex distillation or chromatography, further reduces energy consumption and labor costs associated with purification.
- Enhanced Supply Chain Reliability: Sourcing heptafluoroisobutyl amide produced via this method ensures a more stable supply chain due to the reduced reliance on hazardous gas logistics which are often subject to strict transportation regulations and delays. The robustness of the liquid-phase reaction makes it less susceptible to fluctuations in ambient conditions or minor variations in reagent quality, resulting in consistent batch-to-batch performance that is crucial for long-term production planning. Manufacturers can maintain lower safety stock levels because the higher yield and predictability of the process reduce the risk of production shortfalls. This reliability is particularly valuable for customers in the electronics sector who require just-in-time delivery of high-purity precursors to maintain their own manufacturing schedules for insulating gases.
- Scalability and Environmental Compliance: The process is inherently scalable from laboratory to commercial production because it utilizes standard chemical engineering unit operations such as stirred tank reactors and rotary evaporators that are easily replicated at larger volumes. The absence of toxic gas emissions simplifies the permitting process for new production facilities and reduces the ongoing regulatory burden related to air quality monitoring and reporting. Waste generation is minimized not only by the high yield but also by the ability to recover and reuse the silica gel adsorbent and solvents, aligning with corporate sustainability goals and circular economy initiatives. This environmental stewardship enhances the brand value of the supply chain partners and future-proofs the production capability against tightening global environmental legislation.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of heptafluoroisobutyl amide based on the patented technology. These answers are derived directly from the experimental data and process descriptions found in the intellectual property documentation to ensure accuracy and relevance for industry stakeholders. Understanding these details is crucial for evaluating the feasibility of integrating this intermediate into your specific manufacturing workflows.
Q: Why is liquid ammonia preferred over gaseous ammonia in this synthesis?
A: Using a liquid ammonia methanol solution significantly enhances operational safety and raw material utilization compared to gaseous ammonia, which often suffers from poor absorption and direct atmospheric emission.
Q: What is the critical temperature range for the ammonolysis reaction?
A: The reaction must be strictly controlled at temperatures not higher than 0°C, specifically ramping from -5°C to 0°C, to prevent side reactions and ensure the formation of the desired amide structure.
Q: How is the purity of the final heptafluoroisobutyl amide ensured?
A: High purity is achieved through a combination of solvent removal under mild vacuum, adsorption treatment using 200-400 mesh silica gel to remove colored impurities, and final recrystallization in chloroform.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Heptafluoroisobutyl Amide Supplier
As the demand for eco-friendly insulating gases continues to surge, securing a dependable source of high-quality heptafluoroisobutyl amide is paramount for maintaining competitiveness in the electronic materials sector. NINGBO INNO PHARMCHEM stands ready to support your development and production needs with our extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our state-of-the-art facilities are equipped to handle complex fluorinated chemistry with the utmost safety and precision, ensuring that all deliverables meet stringent purity specifications through our rigorous QC labs. We understand the critical nature of this intermediate in the value chain and are committed to delivering consistency and quality that matches the high standards of the global semiconductor and power transmission industries.
We invite you to engage with our technical team to discuss how this optimized synthesis route can be tailored to your specific volume requirements and cost targets. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the economic benefits of switching to this greener manufacturing process. Please contact our technical procurement team today to索取 specific COA data and route feasibility assessments that will demonstrate our capability to be your long-term strategic partner in the advancement of fluorine chemistry.
