Advanced Acenaphthene Imidazole NHC-Pd Catalysts: Technical Breakthroughs and Commercial Scalability
Advanced Acenaphthene Imidazole NHC-Pd Catalysts: Technical Breakthroughs and Commercial Scalability
The landscape of transition-metal catalysis is undergoing a significant transformation driven by the demand for more robust and efficient synthetic methodologies. A pivotal development in this domain is documented in patent CN103406148A, which introduces a novel class of acenaphthene imidazole nitrogen heterocyclic carbene (NHC) allyl palladium chloride compounds. These catalysts represent a substantial leap forward in organometallic chemistry, specifically designed to address the limitations of traditional systems in aminocarbonylation reactions. For R&D directors and process chemists, the introduction of these sterically bulky and electronically tunable ligands offers a pathway to achieve higher turnover numbers and improved substrate tolerance. The core innovation lies in the unique fusion of the acenaphthene backbone with the imidazole ring, creating a rigid, conjugated system that profoundly influences the electronic environment of the palladium center. This structural modification is not merely academic; it translates directly into practical benefits for the manufacturing of complex pharmaceutical intermediates and fine chemicals, where reliability and purity are paramount.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, palladium-catalyzed cross-coupling and carbonylation reactions have relied heavily on phosphine ligands, which, while effective, suffer from inherent instability and sensitivity. Phosphine ligands are notoriously prone to oxidation upon exposure to air, leading to the formation of phosphine oxides that deactivate the catalyst and complicate purification processes. Furthermore, the carbon-phosphorus bond is generally less stable than the carbon-metal bond found in NHC complexes, often resulting in ligand dissociation under the elevated temperatures required for challenging substrates. This instability necessitates rigorous exclusion of oxygen and moisture, increasing the operational complexity and cost of large-scale manufacturing. Additionally, conventional catalysts often require high loadings to drive reactions to completion, which not only increases the cost of precious metals but also leaves behind significant metal residues that are difficult to remove from the final active pharmaceutical ingredient (API). These factors collectively create bottlenecks in supply chains, extending lead times and introducing variability in batch-to-batch consistency.
The Novel Approach
The novel acenaphthene imidazole NHC-Pd catalysts described in the patent offer a compelling solution to these entrenched problems by leveraging the superior electronic and steric properties of N-heterocyclic carbenes. Unlike phosphines, NHC ligands form exceptionally strong sigma bonds with the palladium center, rendering the complex thermally stable and resistant to decomposition even under harsh reaction conditions. The acenaphthene moiety further enhances this stability through its extended conjugation, which acts as a powerful electron donor, increasing the electron density on the palladium atom. This electronic enrichment facilitates the oxidative addition step, which is often the rate-determining step in catalytic cycles involving aryl halides. Consequently, these catalysts can operate effectively at lower loadings, typically around 1.0 mol%, while maintaining high activity. The result is a streamlined process that reduces the reliance on expensive palladium sources and minimizes the generation of metal-contaminated waste, aligning perfectly with modern green chemistry principles and cost-reduction strategies.
Mechanistic Insights into Acenaphthene Imidazole NHC-Pd Catalysis
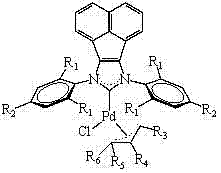
To fully appreciate the utility of these catalysts, one must delve into the mechanistic nuances that govern their reactivity. The acenaphthene imidazole ligand framework provides a unique electronic environment that distinguishes it from simpler imidazole derivatives. The fused aromatic rings of the acenaphthene group create a larger conjugated system, which significantly amplifies the σ-electron donor character of the carbene carbon. This strong donation stabilizes the low-valent palladium species generated during the catalytic cycle, preventing the formation of inactive palladium black precipitates. Moreover, the steric bulk provided by the substituents on the nitrogen atoms (R1 groups such as iso-propyl or iso-butyl) creates a protective pocket around the metal center. This steric shielding prevents bimolecular decomposition pathways and promotes the coordination of substrates in a specific orientation that favors reductive elimination. The bond length data provided in the patent, such as the Pd-C bond distance of approximately 2.048 Å, confirms the strong interaction between the ligand and the metal, which is critical for maintaining catalytic integrity throughout the reaction duration.
Impurity control is another critical aspect where the mechanistic design of these catalysts offers distinct advantages. In traditional coupling reactions, side reactions such as homocoupling of aryl halides or beta-hydride elimination can lead to complex impurity profiles that are difficult to separate. The high selectivity of the acenaphthene NHC-Pd system minimizes these off-cycle pathways by ensuring rapid and efficient turnover of the desired catalytic cycle. The stability of the catalyst also means that it does not degrade into free ligands or metal salts that could interact with sensitive functional groups on the substrate. This results in a cleaner reaction profile with fewer by-products, simplifying the downstream purification process. For procurement and quality assurance teams, this translates to a higher quality crude product, reducing the burden on purification units and lowering the overall cost of goods sold. The ability to tolerate various functional groups without deactivation further expands the scope of applicable substrates, making this technology versatile for diverse synthetic routes.
How to Synthesize Acenaphthene Imidazole NHC-Pd Catalyst Efficiently
The preparation of these high-performance catalysts is designed to be straightforward and scalable, avoiding the need for exotic reagents or extreme conditions. The synthesis involves a simple metathesis reaction between an acenaphthene benzimidazole salt hydrochlorate and an allyl palladium chloride dimer in the presence of a base. This one-pot procedure allows for the direct formation of the active catalytic species without the isolation of unstable free carbene intermediates. The use of common solvents like tetrahydrofuran and bases like potassium tert-butoxide ensures that the process can be easily transferred from the laboratory to pilot and production scales. Detailed standardized synthesis steps are provided in the guide below to assist process engineers in replicating these results with high fidelity.
- Under a nitrogen atmosphere, charge a 50 mL round-bottom flask with acenaphthene benzimidazole salt hydrochlorate (2.1 mmol), allyl palladium chloride dimer (1.0 mmol), and potassium tert-butoxide (2.5 mmol).
- Add tetrahydrofuran (24 mL) as the solvent and stir the reaction mixture at room temperature for 24 hours to ensure complete complexation.
- Remove the solvent under reduced pressure, purify the crude product via column chromatography, and vacuum dry to obtain the final yellow solid catalyst.
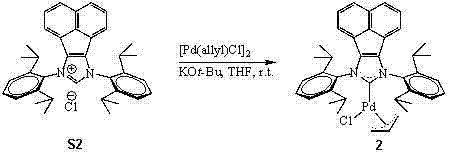
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this novel catalyst technology presents a strategic opportunity to optimize manufacturing costs and enhance supply reliability. The primary economic driver is the significant reduction in catalyst loading required to achieve high conversion rates. Traditional methods often necessitate loadings of 5 mol% or higher to overcome inefficiencies, whereas this new class of catalysts operates effectively at 1.0 mol%. This five-fold reduction in precious metal usage directly correlates to substantial raw material cost savings, especially given the volatile market price of palladium. Furthermore, the enhanced stability of the catalyst eliminates the need for specialized storage conditions such as inert atmosphere gloveboxes, reducing infrastructure costs and minimizing the risk of material spoilage during transit and warehousing. The robustness of the system also allows for the use of less purified solvents and reagents in some cases, further driving down input costs without compromising reaction performance.
- Cost Reduction in Manufacturing: The implementation of these acenaphthene imidazole catalysts leads to a drastic simplification of the production workflow. By eliminating the need for expensive phosphine ligands and reducing the quantity of palladium required, the overall bill of materials is significantly lowered. Additionally, the high selectivity of the catalyst reduces the formation of difficult-to-remove impurities, which decreases the consumption of silica gel and solvents during the purification stage. This cumulative effect results in a leaner manufacturing process with a smaller environmental footprint and lower waste disposal costs. The ability to run reactions at atmospheric pressure of CO, rather than high pressure, also reduces the capital expenditure required for specialized high-pressure reactors, making the technology accessible for a wider range of manufacturing facilities.
- Enhanced Supply Chain Reliability: Supply chain continuity is often threatened by the sensitivity of reagents and the complexity of synthesis routes. The acenaphthene imidazole precursors are synthesized from readily available starting materials, ensuring a stable and secure supply of the catalyst itself. The long shelf-life of these complexes, owing to their air and moisture stability, allows manufacturers to maintain strategic stockpiles without the fear of degradation. This resilience buffers the supply chain against disruptions caused by logistics delays or sudden spikes in demand. Moreover, the reproducibility of the catalytic performance ensures consistent batch quality, reducing the incidence of failed batches that can derail production schedules and delay deliveries to downstream customers.
- Scalability and Environmental Compliance: Scaling up chemical processes often introduces new challenges related to heat transfer and mixing, but the robust nature of these catalysts mitigates many of these risks. The reactions proceed smoothly at moderate temperatures (90°C), which are easily manageable in standard jacketed reactors, facilitating a seamless transition from gram to ton scale. From an environmental perspective, the reduction in metal waste and the use of less hazardous reagents align with increasingly stringent global regulations on chemical manufacturing. This compliance reduces the regulatory burden on the company and enhances its reputation as a sustainable manufacturer, which is a growing requirement for partnerships with major multinational corporations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this catalyst technology. These answers are derived directly from the experimental data and specifications outlined in the patent documentation, providing a reliable basis for decision-making. Understanding these details is crucial for integrating this technology into existing production lines and maximizing its potential benefits.
Q: What represents the primary advantage of acenaphthene imidazole ligands over classical imidazoles?
A: Acenaphthene imidazole ligands possess a larger conjugated system compared to classical imidazoles. This structural feature enhances their σ-electron donor character while maintaining weak π-electron acceptor properties, resulting in higher electron density on the central palladium metal and superior catalytic activity in oxidative addition steps.
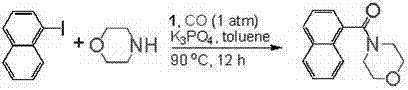
Q: What are the typical reaction conditions for aminocarbonylation using these catalysts?
A: The patented process utilizes mild conditions, typically requiring a catalyst loading of 1.0 mol%, a reaction temperature of 90°C, and carbon monoxide pressure maintained at 1 atm. The reaction proceeds efficiently in toluene with potassium phosphate as the base, achieving high yields within 12 hours.
Q: How does the stability of these NHC-Pd complexes compare to phosphine-based catalysts?
A: These N-heterocyclic carbene metal complexes exhibit exceptional stability towards air, water, and high temperatures due to the strong carbon-to-metal bond. This contrasts sharply with many phosphine-based systems that are prone to oxidation and degradation, thereby simplifying storage and handling protocols in industrial settings.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Acenaphthene Imidazole Catalyst Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced catalytic technologies like the acenaphthene imidazole NHC-Pd system. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from lab-scale discovery to full-scale manufacturing is seamless. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of catalyst or intermediate delivered meets the highest industry standards. We understand that consistency is key in pharmaceutical manufacturing, and our robust quality management systems are designed to provide the reliability your supply chain demands.
We invite you to collaborate with us to leverage these cutting-edge catalytic solutions for your next project. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific process requirements, demonstrating exactly how switching to this technology can improve your bottom line. Please contact our technical procurement team today to request specific COA data and route feasibility assessments. Let us help you optimize your synthesis routes, reduce costs, and accelerate your time to market with our premium chemical solutions.
