Revolutionizing Benzimidazolo Oxathiazine Synthesis via Green Electrochemical Technology for Commercial Scale
The pharmaceutical and fine chemical industries are constantly seeking sustainable methodologies to construct complex heterocyclic scaffolds efficiently. Patent CN115894526A introduces a groundbreaking electrochemical synthesis strategy for producing benzimidazolo[1,5-c][1,2,3]oxathiazine derivatives, a class of polycyclic heterocyclic compounds with significant pharmacological potential. This technology leverages direct current electrolysis to drive an oxidative decarboxylation coupling-cyclization reaction between benzo[e][1,2,3]oxathiazine-2,2-dioxide and N-arylglycine compounds. By utilizing electrons as a traceless oxidant, this method circumvents the need for hazardous chemical oxidants, offering a greener alternative that aligns with modern environmental compliance standards. The process operates under mild conditions, typically at room temperature, and utilizes ethanol as a green solvent, demonstrating high atom economy and exceptional functional group compatibility. For R&D teams and procurement specialists, this represents a pivotal shift towards more cost-effective and scalable manufacturing processes for high-value pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of benzimidazolo[1,5-c][1,2,3]oxathiazine derivatives has relied on methodologies that pose significant economic and environmental challenges. Early approaches, such as those reported by Laha et al., utilized N,N-bis(trimethylsilylmethyl)alkylamine and required two equivalents of silver fluoride (AgF) as a stoichiometric oxidant. As illustrated in the prior art comparison below, this reliance on precious metal oxidants drastically inflates raw material costs and generates substantial heavy metal waste, complicating downstream purification and waste management. Furthermore, more recent photocatalytic methods employing perovskite CsPbBr3, eosin Y, or potassium-modified carbon nitrides have emerged, yet these introduce their own set of limitations including the use of toxic lead-containing catalysts and the requirement for specialized light irradiation equipment. These conventional routes often suffer from limited substrate scope, moderate reaction efficiency, and the logistical burden of handling sensitive or hazardous reagents, making them less ideal for large-scale industrial application.
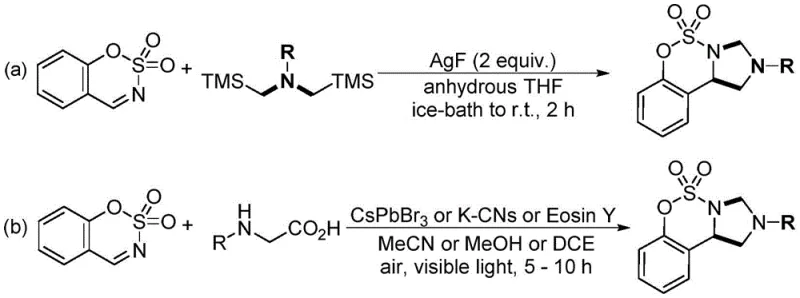
The Novel Approach
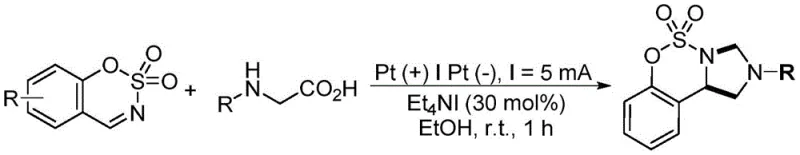
In stark contrast to these traditional pathways, the electrochemical method disclosed in CN115894526A offers a streamlined and robust solution. By employing an undivided electrochemical cell with platinum electrodes, the reaction harnesses electrical energy to drive the transformation directly, eliminating the need for external chemical oxidants entirely. The general reaction scheme demonstrates the direct coupling of benzo[e][1,2,3]oxathiazine-2,2-dioxide with N-arylglycine in the presence of a simple electrolyte salt like tetraethylammonium iodide. This approach not only simplifies the reaction setup but also significantly enhances the safety profile by removing toxic oxidants and heavy metal catalysts from the process. The use of ethanol as the solvent further underscores the green chemistry credentials of this method, providing a medium that is both effective for substrate dissolution and environmentally benign. This novel electrochemical strategy ensures high yields, often exceeding 90% under optimized conditions, while maintaining excellent selectivity, thereby reducing the formation of difficult-to-remove impurities and lowering the overall cost of goods sold for manufacturers.
Mechanistic Insights into Electrochemical Oxidative Decarboxylation Coupling
Understanding the underlying mechanism is crucial for R&D directors aiming to optimize this process for specific analogues. The reaction proceeds through a sophisticated radical pathway initiated at the electrode surfaces. At the cathode, ethanol molecules accept electrons to generate ethoxide anions and hydrogen gas. These ethoxide anions then deprotonate the N-arylglycine substrate to form an anionic intermediate. Subsequently, at the anode surface, this anion undergoes single-electron oxidation to generate an oxygen-centered radical, which rapidly decarboxylates to form a key carbon-centered aminomethyl radical. This highly reactive radical species selectively attacks the C4 position of the benzo[e][1,2,3]oxathiazine-2,2-dioxide ring system. The resulting nitrogen-centered radical intermediate then engages in a second radical coupling event or cyclization sequence, ultimately leading to the formation of the fused benzimidazolo[1,5-c][1,2,3]oxathiazine core. This mechanistic pathway highlights the precision of electrochemical control, where the potential applied dictates the generation of reactive species, minimizing side reactions common in thermal or chemical oxidation methods.
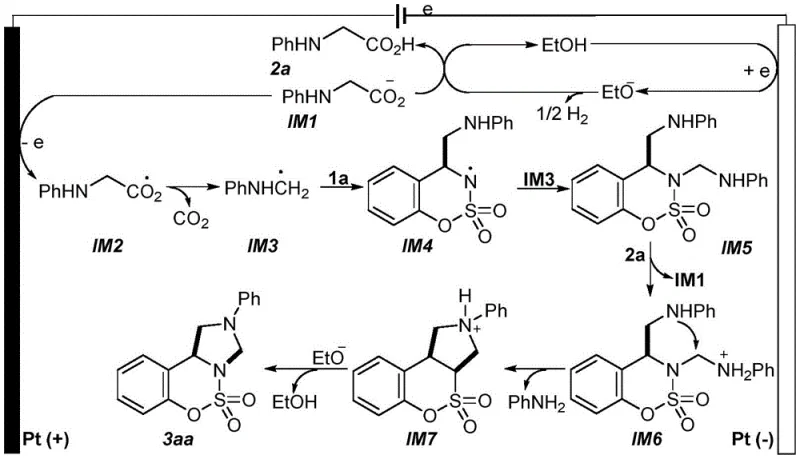
From an impurity control perspective, this mechanism offers distinct advantages. The use of electrons as the oxidant means there are no residual chemical oxidizing agents to quench or remove, which is a common source of impurities in traditional syntheses. The radical intermediates are generated in situ and consumed rapidly in the coupling steps, reducing the likelihood of homocoupling byproducts that often plague free-radical reactions. Furthermore, the mild reaction conditions (room temperature) prevent thermal degradation of sensitive functional groups on the aromatic rings, such as esters or halogens, ensuring a cleaner crude reaction profile. The selectivity of the radical attack on the specific C4 position of the oxathiazine ring ensures regioselective formation of the desired isomer, simplifying the purification process. For quality control teams, this translates to a more predictable impurity profile and higher consistency between batches, which is critical for meeting the stringent specifications required for pharmaceutical intermediates.
How to Synthesize Benzimidazolo[1,5-c][1,2,3]oxathiazine Derivatives Efficiently
Implementing this electrochemical protocol requires careful attention to electrode materials and electrolyte concentration to maximize efficiency. The standard procedure involves dissolving the benzo[e][1,2,3]oxathiazine-2,2-dioxide and N-arylglycine substrates in ethanol, followed by the addition of tetraethylammonium iodide as the supporting electrolyte. Platinum sheets serve as the optimal choice for both anode and cathode to ensure stable current flow and minimize electrode degradation. The reaction is conducted under a constant current of 5 mA at room temperature, typically reaching completion within one hour. This operational simplicity allows for easy scale-up from milligram discovery scales to kilogram production without significant re-engineering of the reactor setup. The detailed standardized synthesis steps for replicating this high-yield transformation are outlined in the guide below.
- Prepare the electrolyte solution by mixing benzo[e][1,2,3]oxathiazine-2,2-dioxide, N-arylglycine compound, and tetraethylammonium iodide (30 mol%) in ethanol solvent within an undivided electrochemical cell.
- Insert platinum sheet electrodes as both the anode and cathode into the reaction mixture, ensuring proper spacing and connection to a constant current power supply.
- Apply a direct current of 5 mA at room temperature for approximately 1 hour, monitoring progress via TLC, followed by extraction and column chromatography purification.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this electrochemical methodology presents compelling economic and logistical benefits. The elimination of expensive stoichiometric oxidants like silver fluoride and toxic photocatalysts directly addresses the volatility of raw material pricing and supply risks associated with precious metals. By substituting these costly reagents with electricity and inexpensive electrolyte salts, the variable cost per kilogram of the final product is substantially reduced. Additionally, the simplified workup procedure, which avoids complex heavy metal scavenging steps, shortens the overall production cycle time. This efficiency gain allows manufacturing facilities to increase throughput without expanding physical footprint, effectively enhancing supply chain resilience and responsiveness to market demand fluctuations.
- Cost Reduction in Manufacturing: The primary driver for cost optimization in this process is the replacement of high-value chemical oxidants with electrical energy. Traditional methods relying on silver salts incur significant material costs that scale linearly with production volume, whereas electricity costs remain relatively stable and low. Furthermore, the use of ethanol as a solvent eliminates the need for expensive, specialized organic solvents and reduces the cost of solvent recovery and disposal. The absence of heavy metal catalysts also removes the financial burden of wastewater treatment and regulatory compliance associated with toxic metal discharge, leading to substantial long-term operational savings.
- Enhanced Supply Chain Reliability: Sourcing high-purity silver fluoride or specialized photocatalysts can be subject to geopolitical supply constraints and long lead times. In contrast, the reagents required for this electrochemical method—N-arylglycines, oxathiazine precursors, and quaternary ammonium salts—are commodity chemicals available from multiple global suppliers. This diversification of the supply base mitigates the risk of production stoppages due to single-source dependency. Moreover, the robustness of the reaction conditions ensures consistent output even with minor variations in raw material quality, providing a more reliable supply stream for downstream API synthesis.
- Scalability and Environmental Compliance: Electrochemical reactors are inherently scalable, allowing for a straightforward transition from laboratory benchtop cells to industrial flow electrolysis systems. The modular nature of electrochemical cells means capacity can be increased by adding more cell units rather than building larger vessels, offering flexible capacity expansion. From an environmental standpoint, the process generates minimal hazardous waste, primarily consisting of benign organic byproducts and hydrogen gas. This aligns with increasingly strict global environmental regulations, reducing the risk of fines and facilitating smoother permitting processes for new manufacturing lines, thereby securing long-term operational continuity.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and scope of this electrochemical synthesis technology. These insights are derived directly from the experimental data and mechanistic studies presented in the patent documentation, providing clarity on reaction parameters and substrate compatibility. Understanding these details is essential for process chemists evaluating the feasibility of adopting this route for their specific project needs.
Q: What are the primary advantages of this electrochemical method over traditional silver fluoride oxidation?
A: The electrochemical method eliminates the need for expensive and stoichiometric chemical oxidants like silver fluoride (AgF), replacing them with electrons as a traceless oxidant. This significantly reduces raw material costs and simplifies the post-reaction workup by avoiding heavy metal waste disposal.
Q: Is this synthesis method compatible with diverse functional groups on the substrate?
A: Yes, the patent data demonstrates excellent functional group tolerance. The reaction successfully proceeds with substrates containing alkyl, alkoxy, halogen (fluoro, chloro, bromo, iodo), trifluoromethyl, and ester groups, yielding products with high purity and consistent yields ranging from 73% to 94%.
Q: What role does the electrolyte salt play in this reaction mechanism?
A: The electrolyte salt, preferably tetraethylammonium iodide, is essential for conducting electrons within the solution. Experimental data indicates that without the electrolyte, the electrochemical reaction does not occur. It facilitates the generation of reactive radical intermediates necessary for the oxidative decarboxylation coupling.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Benzimidazolo[1,5-c][1,2,3]oxathiazine Derivative Supplier
As the demand for complex heterocyclic intermediates grows, partnering with an experienced CDMO is vital for successful commercialization. NINGBO INNO PHARMCHEM possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from lab to plant is seamless. Our technical team is well-versed in electrochemical technologies and maintains stringent purity specifications through our rigorous QC labs, guaranteeing that every batch meets the highest industry standards. We understand the critical nature of supply chain stability and are committed to delivering high-purity intermediates with consistent quality.
We invite you to discuss how this innovative electrochemical route can optimize your manufacturing costs and timelines. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and comprehensive route feasibility assessments to support your R&D and production goals, ensuring a competitive edge in the global pharmaceutical market.
