Advanced Synthesis of 2-Bromo-4-nitrophenylacetonitrile for Commercial Antibiotic Manufacturing
Advanced Synthesis of 2-Bromo-4-nitrophenylacetonitrile for Commercial Antibiotic Manufacturing
The pharmaceutical industry constantly seeks robust and efficient synthetic routes for critical antibiotic intermediates, and the methodology disclosed in patent CN101402589A represents a significant advancement in this domain. This specific technical disclosure outlines a streamlined two-step synthesis for 2-bromo-4-nitrophenylacetonitrile, a pivotal building block in the construction of various therapeutic agents. By utilizing phenylacetonitrile as a readily available starting material, the process circumvents many of the logistical and safety hurdles associated with traditional halogenation methods. For R&D directors and procurement specialists alike, understanding the nuances of this pathway is essential for securing a reliable pharmaceutical intermediate supplier capable of delivering high-purity materials consistently. The strategic implementation of this chemistry not only optimizes yield but also aligns with modern green chemistry principles by reducing hazardous waste streams.
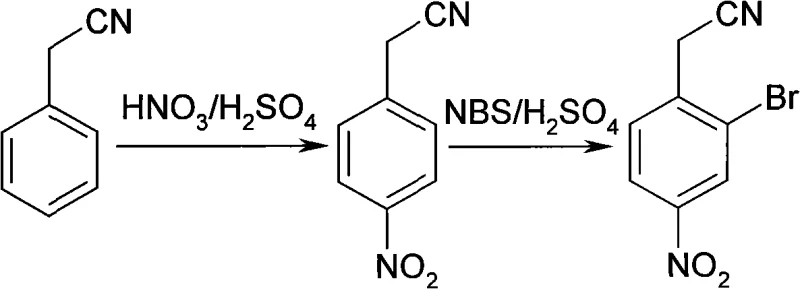
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of halogenated nitro-aromatic compounds has been plagued by issues related to regioselectivity and the handling of dangerous reagents. Conventional approaches often rely on direct bromination using elemental bromine, which poses severe safety risks due to its volatility, corrosivity, and toxicity, necessitating expensive containment infrastructure. Furthermore, traditional electrophilic aromatic substitution on activated rings frequently results in mixtures of ortho-, meta-, and para-isomers, complicating the purification process and driving up the cost of goods sold. In many legacy processes, the order of functional group installation is suboptimal, leading to deactivation of the ring that makes subsequent substitutions difficult or requiring harsh conditions that degrade product quality. These inefficiencies create bottlenecks in cost reduction in API manufacturing, as extensive chromatographic purification or recrystallization steps are often required to meet stringent pharmacopeial standards.
The Novel Approach
The innovative route presented in the patent data fundamentally shifts the paradigm by reversing the typical order of operations and employing N-bromosuccinimide (NBS) as a superior brominating agent. By first establishing the nitro group at the para-position through controlled mixed-acid nitration, the substrate is pre-organized for the subsequent selective bromination. The use of NBS in concentrated sulfuric acid generates the active brominating species in situ under mild conditions, avoiding the exothermic runaway risks associated with liquid bromine. This novel approach ensures that the bromine atom is installed specifically at the ortho-position relative to the acetonitrile group, leveraging the directing effects of the existing substituents. Consequently, this method delivers a cleaner reaction profile with fewer by-products, directly addressing the need for high-purity OLED material or pharmaceutical precursors where trace impurities can be detrimental to biological activity or material performance.
Mechanistic Insights into Electrophilic Aromatic Substitution and Regiocontrol
The success of this synthesis hinges on a deep understanding of electronic effects and steric constraints during electrophilic aromatic substitution. In the first step, phenylacetonitrile undergoes nitration where the -CH2CN group acts as a moderate deactivator but an ortho/para director. However, due to the steric bulk of the methylene nitrile group, the electrophilic nitronium ion preferentially attacks the para-position, yielding p-nitrophenylacetonitrile with high selectivity. This intermediate is crucial because the introduction of the strongly electron-withdrawing nitro group significantly deactivates the aromatic ring, preventing over-nitration and setting the stage for the next transformation. The precise control of temperature between -20°C and 30°C during this phase is vital to suppress side reactions and ensure the integrity of the nitrile functionality, which is sensitive to hydrolysis under acidic conditions.
In the second mechanistic phase, the bromination of p-nitrophenylacetonitrile is driven by the generation of an electrophilic bromine species from NBS in the highly acidic medium. Although the nitro group is meta-directing, the -CH2CN group retains its ortho/para directing influence. Since the para-position is already occupied by the nitro group, the incoming bromine electrophile is directed to the ortho-position relative to the nitrile group. The concentrated sulfuric acid serves not only as a solvent but also as a catalyst that protonates the NBS, facilitating the release of the reactive bromine species. This specific mechanistic pathway minimizes the formation of dibromo impurities or isomers where bromine might attack the meta-position, thereby simplifying the impurity profile. For technical teams, this level of mechanistic control translates to a more predictable process that is easier to validate and scale.
How to Synthesize 2-Bromo-4-nitrophenylacetonitrile Efficiently
Executing this synthesis requires careful attention to thermal management and reagent addition rates to maximize the overall yield, which the patent reports can reach up to 65.1% over two steps. The process begins with the preparation of the mixed acid, followed by the controlled addition of phenylacetonitrile to maintain the reaction temperature within the specified window, ensuring optimal conversion to the nitro-intermediate. Following isolation of the nitro-compound, the bromination step involves dissolving the solid in concentrated sulfuric acid and adding NBS in batches to manage the exotherm effectively.
- Nitration: React phenylacetonitrile with mixed acid (concentrated sulfuric and nitric acid) at controlled temperatures between -20°C and 30°C to form p-nitrophenylacetonitrile.
- Bromination: Dissolve the nitrated intermediate in concentrated sulfuric acid and add N-bromosuccinimide (NBS) gradually while maintaining temperature control to achieve selective ortho-bromination.
- Isolation: Precipitate the final product by pouring the reaction mixture into ice water, followed by filtration, washing, and drying to obtain high-purity 2-bromo-4-nitrophenylacetonitrile.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this synthetic route offers substantial benefits that resonate deeply with procurement managers and supply chain heads focused on stability and efficiency. The elimination of elemental bromine removes a major regulatory and safety burden, reducing the need for specialized storage facilities and hazardous waste disposal protocols. This simplification of the safety profile allows for more flexible manufacturing scheduling and reduces the risk of production stoppages due to safety audits or incidents. Moreover, the reliance on commodity chemicals like sulfuric acid and nitric acid ensures that raw material costs remain stable and predictable, shielding the supply chain from the volatility often seen with specialized organometallic reagents. The robustness of the isolation procedure, which relies on simple precipitation in ice water, further enhances throughput by minimizing solvent usage and drying times.
- Cost Reduction in Manufacturing: The economic viability of this process is underpinned by the high atom economy of the NBS bromination and the avoidance of expensive transition metal catalysts. By achieving high selectivity without the need for precious metals, the process eliminates the costly and time-consuming step of heavy metal scavenging, which is a standard requirement in many catalytic cross-coupling reactions. Additionally, the ability to recover product from the filtrate by neutralization, as described in the patent improvements, ensures that material loss is minimized, effectively lowering the cost per kilogram of the final API intermediate. This holistic approach to cost management makes the technology highly attractive for large-scale production environments.
- Enhanced Supply Chain Reliability: The use of widely available starting materials such as phenylacetonitrile ensures that the supply chain is not vulnerable to single-source bottlenecks. Unlike complex chiral building blocks or custom-synthesized reagents that may have long lead times, the inputs for this process are standard industrial chemicals with established global supply networks. This accessibility translates to shorter lead times for high-purity pharmaceutical intermediates, allowing manufacturers to respond more agilely to market demand fluctuations. The simplicity of the workup also means that production cycles are shorter, enabling faster turnover and improved inventory management for downstream customers.
- Scalability and Environmental Compliance: Scaling this process from laboratory to commercial production is straightforward due to the absence of sensitive reagents that require inert atmospheres or cryogenic conditions beyond standard cooling. The aqueous workup generates waste streams that are easier to treat compared to organic solvent-heavy processes, aligning with increasingly strict environmental regulations. The potential to neutralize the acidic filtrate to recover additional product not only boosts yield but also reduces the volume of acidic waste requiring neutralization, contributing to a lower environmental footprint. This compliance readiness is a critical factor for multinational corporations seeking sustainable partners for their chemical supply needs.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of this specific chemical intermediate. These insights are derived directly from the experimental data and claims within the patent documentation to provide accurate guidance for potential partners. Understanding these details is crucial for assessing the feasibility of integrating this material into your existing development pipelines.
Q: What are the key advantages of using NBS over elemental bromine in this synthesis?
A: Using N-bromosuccinimide (NBS) instead of elemental bromine significantly enhances operational safety by eliminating the handling of volatile and corrosive liquid bromine. Furthermore, NBS offers superior regioselectivity in concentrated sulfuric acid, minimizing the formation of poly-brominated impurities and simplifying the downstream purification process.
Q: How does the nitration step influence the final product purity?
A: The initial nitration of phenylacetonitrile is highly para-selective due to steric hindrance and electronic effects, yielding p-nitrophenylacetonitrile as the primary intermediate. This high regioselectivity in the first step ensures that the subsequent bromination occurs at the desired ortho-position relative to the nitrile group, drastically reducing isomeric impurities.
Q: Is this synthesis method scalable for industrial production?
A: Yes, the method described in patent CN101402589A is designed for scalability. It utilizes common industrial reagents like concentrated sulfuric acid and employs simple isolation techniques such as ice-water precipitation and filtration, which are easily adaptable from laboratory bench scale to multi-ton commercial manufacturing without requiring complex equipment.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Bromo-4-nitrophenylacetonitrile Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the successful development of new antibiotic therapies. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet your volume requirements without compromising on quality. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch of 2-bromo-4-nitrophenylacetonitrile meets the exacting standards required for pharmaceutical applications. Our commitment to technical excellence allows us to navigate the complexities of chemical manufacturing with precision and reliability.
We invite you to collaborate with us to optimize your supply chain and reduce your overall manufacturing costs. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific project needs. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our advanced synthesis capabilities can support your long-term strategic goals. Let us be your partner in bringing innovative medicines to market faster and more efficiently.
