Advanced Nickel-Catalyzed Synthesis of Chiral Alpha-Amino Boronic Acids for Commercial Scale
The pharmaceutical industry continuously seeks robust methodologies for constructing chiral building blocks, particularly those containing boron functionalities which serve as critical warheads in protease inhibitors. Patent CN114716466A introduces a groundbreaking nickel-catalyzed asymmetric hydroamidation strategy that fundamentally shifts the paradigm for synthesizing chiral alpha-amino boronic acids and their esters. This technology addresses long-standing challenges in regioselectivity and enantioselectivity while utilizing inexpensive nickel salts instead of precious metals. The significance of this advancement is underscored by the wide array of bioactive molecules that rely on this structural motif, including blockbuster drugs such as Bortezomib and Vaborbactam. By enabling a direct, one-step construction of the chiral center adjacent to the boron atom, this method offers a streamlined pathway that is highly attractive for industrial adoption. 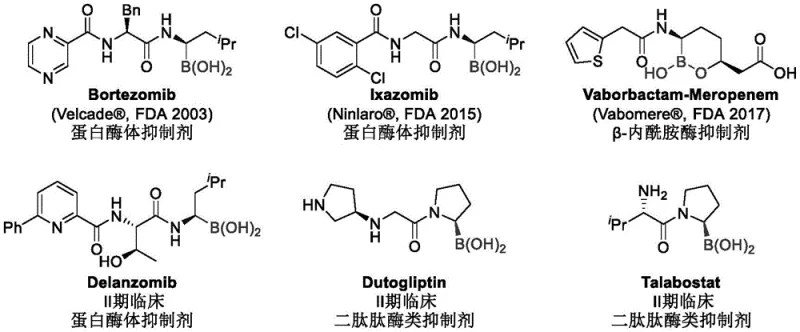
The commercial viability of any synthetic route is often determined by its ability to outperform conventional methods in terms of efficiency and cost. Traditional approaches to chiral alpha-amino boronic acids frequently rely on chiral auxiliary groups, which necessitate additional synthetic steps for installation and removal, thereby reducing overall atom economy and increasing waste generation. Furthermore, existing transition metal-catalyzed methods often suffer from the requirement of expensive catalysts, difficult ligand synthesis, and harsh reaction conditions that complicate scale-up. In contrast, the novel approach disclosed in the patent utilizes a nickel-hydrogen catalytic system that operates under mild conditions, typically between 0°C and 50°C. This drastic reduction in thermal stress not only enhances safety but also preserves the integrity of sensitive functional groups present in complex substrates. 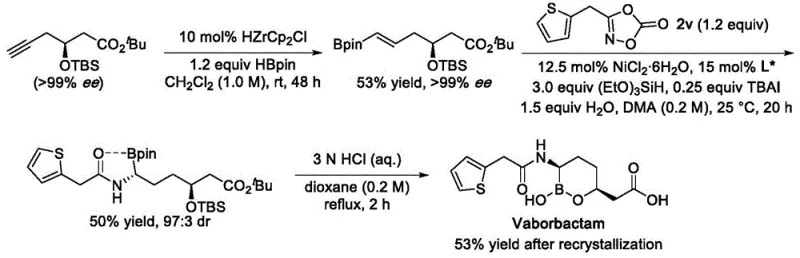
Understanding the mechanistic underpinnings of this transformation is crucial for R&D teams aiming to optimize the process for specific substrates. The reaction proceeds through the generation of a high-activity nickel-hydride species, which undergoes migratory insertion into the alkenyl boronic acid ester. This step is meticulously controlled by a chiral amino alcohol ligand, ensuring that the subsequent coupling with the electrophilic dioxazolone reagent occurs with exceptional stereocontrol. The result is a product with excellent regioselectivity, where the amide group is installed exclusively at the alpha position relative to the boron atom. This precision minimizes the formation of regioisomers, simplifying downstream purification and significantly improving the purity profile of the final active pharmaceutical ingredient. 
For process chemists looking to implement this technology, the operational simplicity is a major advantage. The synthesis involves dissolving the nickel catalyst, chiral ligand, hydrogen source, and additives in a dry organic solvent under an inert atmosphere. Once the alkenyl boronic acid and the dioxazolone amidation reagent are added, the mixture is sealed and allowed to react at ambient or slightly elevated temperatures. The detailed standardized synthesis steps see the guide below.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, the adoption of this nickel-catalyzed methodology presents substantial strategic benefits. The shift away from noble metal catalysts to abundant nickel salts directly translates to a reduction in raw material volatility and cost exposure. Additionally, the simplified workflow reduces the number of unit operations required, which inherently lowers the risk of batch failure and improves overall throughput. This efficiency is critical for maintaining consistent supply lines in a market where demand for chiral intermediates is steadily increasing. The robustness of the reaction conditions also means that manufacturing can be scaled with greater confidence, ensuring that production timelines are met without compromising on quality standards.
- Cost Reduction in Manufacturing: The elimination of expensive chiral auxiliary groups and noble metal catalysts removes significant cost drivers from the bill of materials. By avoiding the need for additional steps to install and remove auxiliaries, the process reduces labor, energy, and solvent consumption. This streamlined approach leads to substantial cost savings in the overall manufacturing budget, allowing for more competitive pricing strategies in the global market without sacrificing margin.
- Enhanced Supply Chain Reliability: The reliance on readily available starting materials such as simple alkenyl boronic acids and dioxazolones mitigates the risk of supply disruptions associated with specialized reagents. The mild reaction conditions reduce the dependency on complex cryogenic equipment or high-pressure reactors, making the process adaptable to a wider range of manufacturing facilities. This flexibility ensures a more resilient supply chain capable of responding quickly to fluctuations in market demand.
- Scalability and Environmental Compliance: The high atom utilization efficiency and reduced waste generation align perfectly with modern green chemistry principles and environmental regulations. The simplified purification process, driven by high regioselectivity, minimizes the volume of solvent waste requiring treatment. This not only lowers disposal costs but also facilitates smoother regulatory approvals for commercial production, supporting sustainable long-term growth.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding this synthesis technology. These insights are derived directly from the patent specifications and are intended to clarify the practical implications for potential partners. Understanding these details is essential for evaluating the feasibility of integrating this route into your existing production portfolio.
Q: What are the advantages of nickel-catalyzed hydroamidation over traditional methods?
A: This method eliminates the need for expensive chiral auxiliary groups and harsh reaction conditions, offering higher atom utilization efficiency and simpler operation compared to traditional transition metal catalysis.
Q: Can this synthesis method be applied to complex drug molecules like Vaborbactam?
A: Yes, the patent explicitly demonstrates the successful synthesis of Vaborbactam intermediates with high enantioselectivity, proving its utility for bioactive molecule production.
Q: What represents the key cost-saving factor in this manufacturing process?
A: The use of readily available raw materials and the elimination of noble metal catalysts significantly reduce material costs, while the one-step nature minimizes processing time and waste.
- Prepare the reaction mixture by dissolving metallic nickel catalyst, chiral ligand, hydrogen source, and additive in dry organic solvent under inert gas.
- Add alkenyl boronic acid ester and amidation reagent (dioxazolone) to the mixture and seal the reaction vessel.
- React at mild temperatures (0-50°C), then concentrate and purify via column chromatography to obtain the target chiral compound.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral Alpha-Amino Boronic Acid Supplier
NINGBO INNO PHARMCHEM stands at the forefront of translating advanced academic research into commercial reality. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative methods like this nickel-catalyzed hydroamidation are successfully implemented at an industrial level. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch meets the exacting standards required by top-tier pharmaceutical companies. Our commitment to technical excellence ensures that the theoretical benefits of this patent are fully realized in the final product delivered to your facility.
We invite you to engage with our technical procurement team to discuss how this technology can optimize your specific supply chain requirements. By requesting a Customized Cost-Saving Analysis, you can gain a clear understanding of the economic impact of switching to this more efficient route. We encourage you to contact us today to索取 specific COA data and route feasibility assessments tailored to your project needs, ensuring a partnership built on transparency and mutual success.
