Advanced Synthesis of Dobutamine Hydrochloride Intermediate: A Safer, Scalable Commercial Route
Advanced Synthesis of Dobutamine Hydrochloride Intermediate: A Safer, Scalable Commercial Route
The cardiovascular pharmaceutical market continues to demand robust supply chains for critical anti-shock agents, with dobutamine hydrochloride remaining a cornerstone therapy for heart failure and cardiogenic shock. Central to the efficient manufacturing of this life-saving drug is the production of its key precursor, N-(3,4-dimethoxyphenethyl)-4-(4-methoxyphenyl)butan-2-amine hydrochloride, often referred to as Compound V. A significant technological breakthrough in this domain is detailed in patent CN111807975A, which outlines a novel preparation method that fundamentally restructures the synthetic pathway. This innovation addresses long-standing industry pain points regarding safety, cost, and impurity control by replacing hazardous high-pressure hydrogenation and toxic solvents with a milder, chemoselective reduction strategy. For global procurement and R&D teams, understanding this shift is critical for securing a reliable dobutamine intermediate supplier capable of meeting stringent regulatory standards while optimizing manufacturing economics.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Compound V has been plagued by significant operational hazards and regulatory complexities inherent to older patented technologies, such as those described in US3987200. Traditional routes predominantly relied on high-pressure catalytic hydrogenation using palladium on carbon (Pd/C), a process that necessitates specialized, expensive high-pressure reactor vessels and introduces severe safety risks related to hydrogen gas handling on an industrial scale. Furthermore, alternative methods reported by manufacturers like the Shanghai No. 2 Pharmaceutical Factory utilized p-toluenesulfonic acid as a catalyst in conjunction with highly toxic solvents like benzene and diethyl ether. The use of p-toluenesulfonic acid poses a critical quality risk, as it can react with alcohol solvents to generate genotoxic p-toluenesulfonate esters, creating a formidable hurdle for regulatory approval in major markets. Additionally, the reliance on benzene, a known carcinogen, and diethyl ether, which has a low flash point and high volatility, complicates waste treatment and increases the overall environmental footprint of the manufacturing process.
The Novel Approach
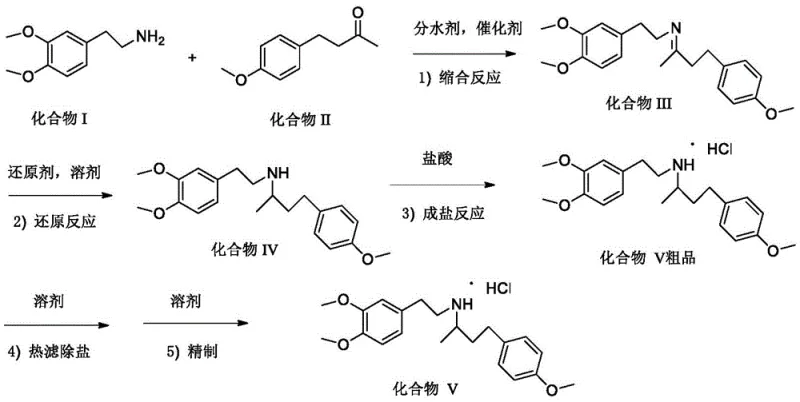
In stark contrast to these legacy methods, the process disclosed in CN111807975A introduces a paradigm shift by utilizing acetic acid as a benign catalyst and cyclohexane as an azeotropic water-removing agent for the initial condensation step. This modification not only eliminates the risk of genotoxic sulfonate ester formation but also replaces carcinogenic benzene with cyclohexane, which offers a safer operating profile and easier solvent recovery due to its favorable azeotropic properties with water. The subsequent reduction step abandons high-pressure hydrogenation in favor of potassium borohydride (KBH4) in anhydrous ethanol, a reagent system that operates under atmospheric pressure and moderate temperatures (60-70°C). This transition drastically simplifies the equipment requirements, allowing standard glass-lined or stainless steel reactors to be used instead of specialized high-pressure autoclaves. The final purification strategy employs isopropanol for hot filtration and recrystallization, effectively removing inorganic salts generated during the reaction without the need for hazardous ether extractions, thereby streamlining the workflow for cost reduction in pharmaceutical intermediates manufacturing.
Mechanistic Insights into Acid-Catalyzed Condensation and Borohydride Reduction
The core chemical innovation lies in the precise control of the reductive amination sequence, beginning with the condensation of 3,4-dimethoxyphenethylamine (Compound I) and 4-(4-methoxyphenyl)-2-butanone (Compound II). In this step, acetic acid acts as a mild proton donor, facilitating the nucleophilic attack of the amine on the ketone carbonyl group to form an imine intermediate (Compound III). The use of cyclohexane is mechanistically crucial here; it forms a low-boiling azeotrope with water (boiling point 80-85°C), allowing for the continuous removal of the water byproduct via a Dean-Stark trap or similar apparatus. This drives the equilibrium towards the imine product according to Le Chatelier's principle, ensuring high conversion rates without the need for excessive reagent loading. The molar ratio is optimized to approximately 1:1.05 (amine to ketone), balancing cost efficiency with reaction completeness, while the catalyst loading is kept minimal (0.01:1 molar ratio) to prevent unnecessary salt formation that could complicate downstream processing.
Following condensation, the mechanism shifts to hydride transfer reduction. Unlike catalytic hydrogenation which relies on surface adsorption and dissociation of hydrogen gas on a metal lattice, the use of potassium borohydride provides a direct source of nucleophilic hydride ions (H-) in the alcoholic solvent. The borohydride anion attacks the electrophilic carbon of the imine bond, reducing it to the secondary amine (Compound IV). This chemical reduction is highly chemoselective and proceeds smoothly at 60-70°C, avoiding the over-reduction or side reactions often seen with more aggressive reducing agents. The subsequent salification with hydrochloric acid converts the free base into the stable hydrochloride salt (Compound V). Crucially, the purification mechanism leverages the differential solubility of the organic product versus inorganic byproducts (such as potassium chloride and borate salts) in isopropanol. By dissolving the crude product in refluxing isopropanol and performing a hot filtration, insoluble inorganic salts are physically removed before the product crystallizes upon cooling, resulting in a high-purity dobutamine intermediate with purity levels exceeding 99.9% as demonstrated in the patent examples.
How to Synthesize Dobutamine Hydrochloride Intermediate Efficiently
The implementation of this synthesis route requires careful attention to stoichiometry and thermal management to maximize yield and purity. The process is divided into distinct operational units: condensation with azeotropic dehydration, atmospheric chemical reduction, salt formation, and a dual-stage recrystallization protocol. While the general chemistry is straightforward, the specific parameters regarding solvent volumes, temperature ramps, and filtration timing are critical for reproducibility on a multi-ton scale. Operators must ensure complete water removal during the condensation phase to prevent hydrolysis of the imine, and strictly control the addition rate of hydrochloric acid during salification to manage exotherms and crystal morphology. For a detailed breakdown of the specific operational parameters, reagent quantities, and temperature profiles required to execute this synthesis successfully, please refer to the standardized technical guide below.
- Condensation Reaction: React 3,4-dimethoxyphenethylamine with 4-(4-methoxyphenyl)-2-butanone using acetic acid catalyst and cyclohexane for azeotropic water removal.
- Reduction and Salification: Reduce the condensation product with potassium borohydride in anhydrous ethanol, followed by hydrochloric acid salification to form the crude salt.
- Purification: Perform hot filtration in isopropanol to remove inorganic salts, followed by recrystallization to obtain high-purity Compound V.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic sourcing perspective, the adoption of the methodology described in CN111807975A offers profound advantages that extend beyond simple chemical yield. By transitioning away from high-pressure hydrogenation and toxic solvents, manufacturers can significantly de-risk their supply chains and reduce the total cost of ownership for the production facility. The elimination of palladium catalysts removes a major variable cost component, as precious metal prices are volatile and the recovery of spent catalyst adds processing steps. Furthermore, the avoidance of genotoxic reagents like p-toluenesulfonic acid simplifies the regulatory filing process, reducing the time and expense associated with impurity qualification and validation studies. This streamlined approach allows for faster technology transfer and more reliable supply continuity, which is essential for maintaining the commercial scale-up of complex pharmaceutical intermediates in a regulated environment.
- Cost Reduction in Manufacturing: The replacement of expensive palladium on carbon catalysts with commodity chemicals like acetic acid and potassium borohydride results in substantial raw material cost savings. Additionally, the shift from high-pressure autoclaves to standard atmospheric reactors reduces capital expenditure (CAPEX) on equipment and lowers maintenance costs associated with high-pressure safety systems. The use of recyclable solvents like cyclohexane and isopropanol further enhances economic efficiency by minimizing waste disposal fees and solvent purchase volumes.
- Enhanced Supply Chain Reliability: The reagents utilized in this novel process, including 3,4-dimethoxyphenethylamine and 4-(4-methoxyphenyl)-2-butanone, are readily available bulk chemicals with stable supply chains, unlike specialized catalysts that may have limited suppliers. The robustness of the atmospheric pressure process means that production is less susceptible to interruptions caused by equipment failure or safety shutdowns, ensuring consistent delivery schedules. This reliability is critical for downstream API manufacturers who require just-in-time delivery to meet their own production targets without holding excessive inventory buffers.
- Scalability and Environmental Compliance: The process is inherently greener, avoiding the use of benzene and ether, which are subject to increasingly strict environmental regulations and emission limits. The simplified workup procedure, which relies on hot filtration rather than complex liquid-liquid extractions, reduces solvent consumption and wastewater generation. This environmental compatibility facilitates easier permitting for new production lines and aligns with the sustainability goals of modern pharmaceutical companies, making it an ideal candidate for reducing lead time for high-purity pharmaceutical intermediates while maintaining a low environmental footprint.
Frequently Asked Questions (FAQ)
To assist technical decision-makers in evaluating the feasibility of this synthesis route for their specific applications, we have compiled answers to common inquiries regarding safety, purity, and scalability. These insights are derived directly from the experimental data and beneficial effects reported in the patent literature, providing a factual basis for assessing the technology's potential impact on your manufacturing operations. Understanding these nuances is essential for conducting a thorough risk-benefit analysis before committing to a new supplier or process change.
Q: How does this new method improve safety compared to traditional Pd/C hydrogenation?
A: The novel process eliminates the need for high-pressure catalytic hydrogenation using expensive palladium on carbon (Pd/C). Instead, it utilizes atmospheric pressure reduction with potassium borohydride, significantly reducing explosion risks and equipment costs associated with high-pressure reactors.
Q: What measures are taken to control genotoxic impurities in this synthesis?
A: Unlike prior art methods that used p-toluenesulfonic acid (which can form genotoxic tosylate esters), this method employs acetic acid as the catalyst. Additionally, the purification strategy uses isopropanol hot filtration to effectively remove inorganic salts and potential organic impurities, ensuring high product purity.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the process is designed for scalability. It avoids hazardous solvents like benzene and ether, using safer alternatives like cyclohexane and isopropanol. The mild reaction conditions (60-70°C) and atmospheric pressure operations make it highly adaptable for commercial scale-up of complex pharmaceutical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Dobutamine Hydrochloride Intermediate Supplier
The synthesis route detailed in CN111807975A represents a significant advancement in the manufacturing of cardiovascular drug intermediates, offering a blend of safety, efficiency, and regulatory compliance that is difficult to match with legacy technologies. At NINGBO INNO PHARMCHEM, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this patent are fully realized in practical, large-scale operations. Our state-of-the-art facilities are equipped to handle the specific requirements of this process, including azeotropic distillation units and precise temperature-controlled reactors, while our rigorous QC labs enforce stringent purity specifications to guarantee that every batch meets the highest international standards for pharmaceutical intermediates.
We invite procurement leaders and R&D directors to engage with us for a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. Our technical team is ready to provide specific COA data from pilot runs and comprehensive route feasibility assessments to demonstrate how switching to this optimized synthesis path can enhance your supply chain resilience. By partnering with us, you gain access to a supply source that prioritizes both chemical excellence and commercial viability, ensuring a steady flow of high-quality materials for your critical drug formulations.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
