Advanced Lewis Acid Catalyzed Synthesis of Polysubstituted 4H-Pyran Intermediates for Commercial Scale
Advanced Lewis Acid Catalyzed Synthesis of Polysubstituted 4H-Pyran Intermediates for Commercial Scale
The landscape of organic synthesis for heterocyclic compounds is constantly evolving, driven by the relentless demand for more efficient, cost-effective, and environmentally benign manufacturing processes. A significant breakthrough in this domain is detailed in patent CN114181182B, which discloses a novel synthesis method for polysubstituted 4H-pyran compounds. These heterocyclic scaffolds are not merely academic curiosities; they serve as critical building blocks in the construction of bioactive natural products, pharmaceutical intermediates, and advanced functional materials such as organic light-emitting diodes (OLEDs) and dyes. The traditional reliance on harsh reaction conditions and precious metal catalysts has long been a bottleneck for the industrial scalability of these valuable molecules. This new methodology addresses these challenges head-on by utilizing accessible Lewis acid catalysts to facilitate a [4+2] cycloaddition between β,γ-unsaturated-α-ketoesters and alkynes, offering a robust pathway for the reliable pharmaceutical intermediate supplier seeking to optimize their production lines.
For R&D directors and process chemists, the implications of this technology are profound, particularly regarding purity profiles and impurity control. The ability to synthesize these complex cores under mild conditions minimizes thermal degradation and side reactions that often plague high-temperature processes. Furthermore, the avoidance of transition metals like palladium or gold removes a significant regulatory hurdle, as residual heavy metals in active pharmaceutical ingredients (APIs) are subject to strict limits. By shifting to earth-abundant Lewis acids, manufacturers can streamline their purification workflows, ensuring that the final high-purity OLED material or drug candidate meets stringent quality specifications without the need for extensive metal scavenging steps. This patent represents a pivotal shift towards greener, more sustainable chemistry that aligns perfectly with modern corporate sustainability goals.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the 4H-pyran ring system has relied heavily on the [4+2] cycloaddition of α,β-unsaturated carbonyl compounds with alkynes. However, the existing literature and established industrial practices reveal significant drawbacks that hinder widespread adoption. Primarily, conventional methods frequently necessitate the use of pre-activated substrates, which adds extra synthetic steps, increases waste generation, and drives up the overall cost of goods sold (COGS). Moreover, many reported protocols depend on noble metal catalysis, involving expensive elements such as gold or palladium complexes. These catalysts are not only costly to procure but also pose severe challenges in terms of removal from the final product, often requiring specialized resin treatments or multiple recrystallizations that erode overall yield. Additionally, the reaction conditions for these traditional routes are often苛刻 (harsh), requiring elevated temperatures or inert atmospheres that are difficult to maintain consistently on a multi-ton scale, thereby introducing variability and risk into the supply chain.
The Novel Approach
In stark contrast, the methodology described in CN114181182B offers a streamlined and economically superior alternative. By employing readily available β,γ-unsaturated-α-ketoesters directly, the process eliminates the need for substrate pre-activation, effectively shortening the synthetic route and reducing raw material consumption. The core innovation lies in the use of Lewis acids such as Hafnium Chloride (HfCl4), Indium Chloride (InCl3), or Indium Trifluoromethanesulfonate (In(OTf)3) to activate the reaction partners. This catalytic system operates efficiently at mild temperatures, often ranging from 0°C to 60°C, and completes within a practical timeframe of 0.5 to 6 hours. This dramatic reduction in energy input and reaction severity translates directly into cost reduction in pharmaceutical intermediates manufacturing. The simplicity of the workup, typically involving standard column chromatography or crystallization, further enhances the operational efficiency, making this approach highly attractive for commercial scale-up of complex polymer additives or fine chemicals.
Mechanistic Insights into Lewis Acid-Catalyzed [4+2] Cycloaddition
Understanding the mechanistic underpinnings of this transformation is crucial for process optimization and troubleshooting. The reaction proceeds through a Lewis acid-mediated activation of the carbonyl group within the β,γ-unsaturated-α-ketoester. The Lewis acid coordinates with the oxygen atom of the carbonyl moiety, increasing the electrophilicity of the adjacent carbon centers and facilitating the nucleophilic attack by the alkyne. This interaction lowers the activation energy barrier for the subsequent [4+2] cycloaddition, allowing the reaction to proceed smoothly under mild thermal conditions. The specific choice of Lewis acid can fine-tune the reactivity; for instance, harder Lewis acids may preferentially coordinate with harder oxygen donors, while softer metals might interact differently with the alkyne pi-system. This tunability allows chemists to adapt the protocol to a wide variety of electronic environments presented by different substituents on the starting materials.

From an impurity control perspective, the mildness of this catalytic cycle is a distinct advantage. High-temperature reactions often promote polymerization of alkynes or decomposition of sensitive ester functionalities, leading to complex impurity profiles that are difficult to separate. By maintaining the reaction temperature below 60°C, the formation of these thermal byproducts is significantly suppressed. Furthermore, the high regioselectivity observed in many of the examples suggests that the Lewis acid effectively directs the orientation of the cycloaddition, minimizing the formation of regioisomers. This level of control is essential for producing high-purity electronic chemical intermediates where even trace isomers can detrimentally affect the performance of the final device. The robustness of the mechanism across different substrates ensures consistent quality, a key metric for any supply chain负责人 (head).
How to Synthesize Polysubstituted 4H-Pyran Efficiently
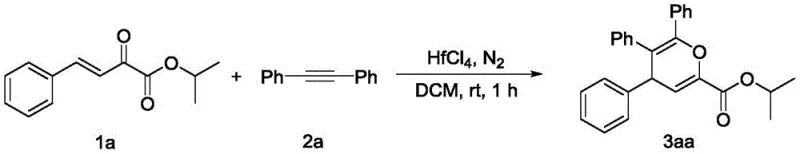
The practical implementation of this synthesis is straightforward and designed for reproducibility in both laboratory and pilot plant settings. The general procedure involves charging a reactor with the ketoester and alkyne substrates under an inert nitrogen atmosphere to prevent moisture interference, followed by the addition of the Lewis acid catalyst and a suitable organic solvent such as dichloromethane or 1,2-dichloroethane. The reaction progress is monitored until completion, after which standard workup procedures yield the desired product. To illustrate the feasibility and specific conditions, consider the synthesis of compound 3aa, a representative polysubstituted 4H-pyran derivative.
- Under nitrogen protection, sequentially add β,γ-unsaturated-α-ketoester, alkyne, Lewis acid catalyst, and solvent into the reactor.
- Stir the reaction mixture at a controlled temperature ranging from 0°C to 60°C until the reaction is complete, typically within 0.5 to 6 hours.
- Concentrate the reaction liquid using a rotary evaporator and purify the crude product via column chromatography using petroleum ether and ethyl acetate.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain strategists, the adoption of this synthesis method offers tangible benefits that extend beyond mere chemical elegance. The primary driver for value creation here is the substantial reduction in catalyst costs. By replacing precious metals with abundant Lewis acids, the direct material cost of the catalytic system is drastically lowered. This shift also mitigates supply risk, as the market for indium or hafnium salts is generally more stable and less prone to geopolitical volatility compared to platinum group metals. Furthermore, the simplified downstream processing reduces the consumption of solvents and auxiliary materials required for metal scavenging, contributing to a leaner and more cost-effective manufacturing operation. These factors combined create a compelling business case for integrating this technology into existing production portfolios.
- Cost Reduction in Manufacturing: The elimination of expensive noble metal catalysts represents a direct saving on bill of materials. Additionally, the mild reaction conditions reduce energy consumption for heating and cooling, while the simplified purification process lowers labor and solvent costs associated with extensive chromatography or metal removal steps. The overall process efficiency is enhanced, leading to a more competitive cost structure for the final intermediates.
- Enhanced Supply Chain Reliability: The use of commercially available and stable starting materials ensures a consistent supply of raw inputs. The robustness of the reaction conditions means that production schedules are less likely to be disrupted by equipment limitations or safety concerns associated with high-pressure or high-temperature operations. This reliability is critical for maintaining continuous supply to downstream customers in the pharmaceutical and electronics sectors.
- Scalability and Environmental Compliance: The protocol is inherently scalable, having been demonstrated to work effectively with various substrate loadings. The reduced generation of heavy metal waste aligns with increasingly stringent environmental regulations, simplifying waste disposal and reducing the environmental footprint of the manufacturing site. This compliance advantage future-proofs the production process against tightening global standards.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and technical specifications provided in the patent documentation, ensuring accuracy and relevance for potential partners. Understanding these details is essential for evaluating the fit of this technology within your specific manufacturing context.
Q: What are the advantages of using Lewis acids over noble metal catalysts for 4H-pyran synthesis?
A: The use of Lewis acids such as Hafnium Chloride or Indium Trifluoromethanesulfonate eliminates the need for expensive noble metals like palladium or gold, significantly reducing raw material costs and simplifying the removal of heavy metal residues from the final pharmaceutical intermediate.
Q: What is the substrate scope for this synthesis method?
A: The method demonstrates broad applicability, accommodating various R groups including saturated alkyl, substituted alkyl, aryl, naphthyl, and thienyl groups, allowing for the diverse functionalization required in complex drug molecule construction.
Q: What are the typical reaction conditions required for this cyclization?
A: The reaction proceeds under mild conditions, typically at room temperature or slightly elevated temperatures up to 60°C, with reaction times ranging from 0.5 to 6 hours, making it highly energy-efficient and suitable for large-scale production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Polysubstituted 4H-Pyran Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this Lewis acid-catalyzed synthesis for the production of high-value heterocyclic intermediates. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from lab bench to full-scale manufacturing is seamless and efficient. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of polysubstituted 4H-pyran delivered meets the highest industry standards for pharmaceutical and electronic applications.
We invite you to collaborate with us to leverage this advanced chemistry for your next project. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality needs. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us help you optimize your supply chain with this innovative synthesis method.
