Optimizing Azilsartan Production: A Technical Analysis of Novel Synthetic Routes for Commercial Scale-Up
The pharmaceutical landscape for antihypertensive agents continues to evolve, driven by the need for more efficient and cost-effective manufacturing processes for Angiotensin II receptor blockers. Patent CN103880830A introduces a significant technological breakthrough in the synthesis of Azilsartan, addressing critical bottlenecks found in prior art methodologies. This novel approach fundamentally restructures the synthetic pathway to bypass the formation of inevitable impurities that have historically plagued industrial production. By shifting from a linear sequence that exposes sensitive functional groups to harsh conditions, to a convergent strategy that assembles robust fragments, the patent outlines a route capable of delivering high-purity active pharmaceutical ingredients (APIs). For R&D directors and procurement specialists, understanding this shift is vital, as it represents a transition from a process with a reported total recovery of only 23% to one that ensures purity levels exceeding 99.5% via HPLC analysis. This report dissects the chemical rationale behind this improvement and evaluates its impact on commercial viability and supply chain stability.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthesis routes for Azilsartan, exemplified by the methodology disclosed in U.S. Pat 5583141, rely on a sequence that introduces significant chemical instability early in the process. In these conventional pathways, the synthesis begins with a benzimidazole derivative containing a 2-ethoxy group, which is intended to remain intact throughout the subsequent transformations. However, the chemical reality is that this ethoxy moiety is highly susceptible to de-ethylation under the reaction conditions required for ring closure and functionalization. This instability triggers a cascade of side reactions, generating a complex mixture of structural analogs and degradation products known as Impurity 1 through Impurity 4. The presence of these structurally similar byproducts creates a severe downstream burden, necessitating elaborate and multi-step purification protocols to isolate the desired API. Consequently, the cumulative effect of these side reactions and the extensive refining required results in a dismal overall yield, often hovering around 23%, which renders the process economically unviable for large-scale commercial manufacturing.
The Novel Approach
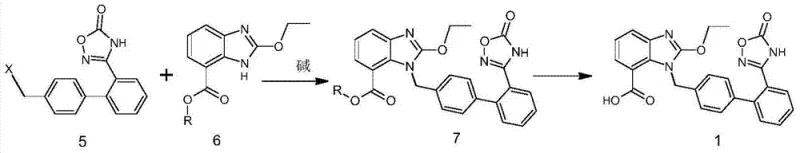
In stark contrast, the methodology presented in CN103880830A adopts a divergent synthetic logic that effectively isolates the reactive centers until the final assembly stages. Instead of building the oxadiazole ring onto an already complex and sensitive benzimidazole scaffold, this novel route constructs the oxadiazole-containing biphenyl fragment independently. This key intermediate, characterized by a halogenated methyl group, is chemically robust and free from the labile ethoxy groups that cause issues in the traditional route. The core innovation lies in the late-stage coupling of this stable fragment with the benzimidazole derivative. By postponing the formation of the critical bond until the molecular architecture is largely complete, the process avoids exposing sensitive functionalities to incompatible reagents. This strategic rearrangement of the synthetic tree not only simplifies the reaction profile but also inherently suppresses the formation of the notorious de-ethylated impurities, thereby streamlining the isolation process and significantly boosting the final throughput.
Mechanistic Insights into Nucleophilic Substitution and Cyclization
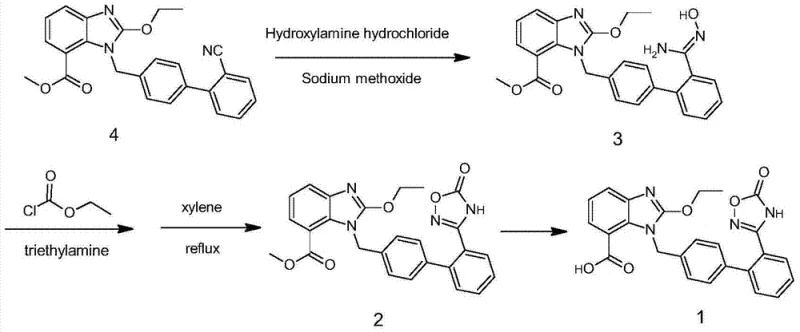
The success of this novel synthesis hinges on the precise control of nucleophilic substitution mechanisms during the coupling phase. The reaction involves the interaction between a halogenated intermediate (Compound 5, where X is Cl or Br) and a benzimidazole carboxylate (Compound 6). Mechanistically, this is a classic SN2-type displacement where the nitrogen atom of the benzimidazole ring acts as a nucleophile, attacking the benzylic carbon of the biphenyl fragment. The choice of base is critical here; bases such as potassium carbonate or triethylamine are employed to deprotonate the benzimidazole nitrogen, enhancing its nucleophilicity without inducing elimination reactions or hydrolyzing the ester moiety prematurely. The reaction is typically conducted in polar aprotic or moderately polar solvents like toluene, THF, or ethyl acetate at temperatures ranging from 20°C to 60°C. These mild conditions are sufficient to drive the coupling to completion while maintaining the integrity of the ester group, which is essential for the subsequent hydrolysis step. The stoichiometry is carefully balanced, often using a slight excess of the benzimidazole component to ensure full conversion of the valuable halogenated intermediate.
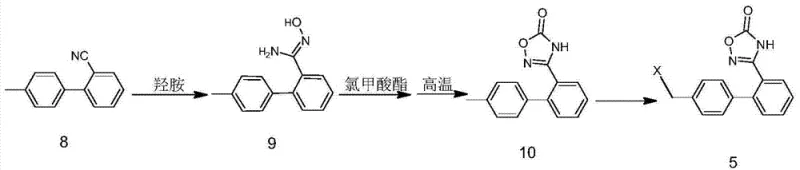
Furthermore, the preparation of the key halogenated intermediate (Compound 5) involves a high-temperature cyclization that is uniquely managed to prevent decomposition. The precursor amidoxime (Compound 9) reacts with chloroformates at elevated temperatures (100°C to 180°C) to form the oxadiazolone ring (Compound 10). This step requires solvents with high boiling points, such as DMSO or DMF, to sustain the thermal energy needed for cyclization. Following this, a radical halogenation using agents like N-bromosuccinimide (NBS) or N-chlorosuccinimide (NCS) in the presence of initiators like AIBN introduces the reactive handle for the final coupling. This sequence ensures that the oxadiazole ring is fully formed and stabilized before it ever encounters the benzimidazole system, effectively decoupling the two complex heterocyclic formations and allowing each to proceed under its own optimal conditions without mutual interference.
How to Synthesize Azilsartan Efficiently
The implementation of this synthetic route requires careful attention to solvent selection and temperature control to maximize the benefits of the convergent strategy. The process is designed to be operationally simple, avoiding the need for cryogenic conditions or exotic catalysts that often complicate technology transfer. By utilizing commercially available starting materials and standard reagents, the barrier to entry for manufacturing is significantly lowered. The following guide outlines the standardized operational framework derived from the patent examples, providing a clear roadmap for process chemists aiming to replicate these high-yield results in a pilot or production setting.
- Couple the halogenated intermediate (Compound 5) with the benzimidazole ester (Compound 6) using a base like potassium carbonate or triethylamine in solvents such as toluene or THF at 20-60°C.
- Isolate the coupled ester intermediate (Compound 7) through standard workup procedures like washing and recrystallization.
- Perform hydrolysis on Compound 7 using aqueous alkali (LiOH, NaOH, or KOH) in methanol, followed by acidification to precipitate high-purity Azilsartan.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this novel synthesis route translates directly into tangible operational efficiencies and risk mitigation. The primary advantage lies in the drastic simplification of the purification train. By eliminating the generation of de-ethylated impurities, the need for complex chromatographic separations or multiple recrystallization steps is removed. This reduction in downstream processing not only shortens the batch cycle time but also decreases the consumption of solvents and consumables, leading to substantial cost savings in raw material utilization. Furthermore, the robustness of the intermediates means that the process is less sensitive to minor fluctuations in reaction parameters, enhancing the reliability of supply and reducing the rate of batch failures. This stability is crucial for maintaining continuous production schedules and meeting the rigorous quality standards required by global regulatory bodies.
- Cost Reduction in Manufacturing: The economic impact of this process is driven by the elimination of yield-killing side reactions. In the conventional route, the loss of material to impurities necessitates processing larger volumes of starting material to obtain the same amount of API, inflating the cost of goods sold. By contrast, the novel route's high selectivity ensures that a much larger proportion of the input mass is converted into the desired product. Additionally, the avoidance of expensive and hazardous reagents often required for impurity scrubbing further lowers the operational expenditure. The simplified workup procedure reduces labor hours and utility costs associated with extended heating, cooling, and filtration cycles, creating a leaner and more cost-effective manufacturing profile.
- Enhanced Supply Chain Reliability: Supply continuity is often threatened by processes that rely on unstable intermediates with short shelf lives. The intermediates generated in this new pathway, particularly the halogenated oxadiazole derivative, are chemically stable and can be stored or transported with minimal degradation. This stability allows for greater flexibility in production planning, enabling manufacturers to build inventory buffers of key intermediates without the risk of spoilage. Moreover, the use of common, commodity-grade solvents and reagents reduces dependency on specialized supply chains that might be prone to disruption. This resilience ensures that production can be sustained even during periods of market volatility, securing the supply of this critical antihypertensive ingredient.
- Scalability and Environmental Compliance: Scaling a chemical process often amplifies safety and environmental challenges, but this route is inherently designed for industrial expansion. The reaction conditions are moderate, avoiding extreme pressures or temperatures that require specialized reactor vessels. The reduction in solvent usage and waste generation aligns with green chemistry principles, minimizing the environmental footprint of the manufacturing site. Fewer purification steps mean less hazardous waste discharge, simplifying compliance with increasingly stringent environmental regulations. This scalability ensures that the process can be seamlessly transferred from kilogram-scale development to multi-ton commercial production without the need for fundamental re-engineering, facilitating rapid market entry.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and validation of this synthesis method. They are derived from the specific experimental data and comparative analysis provided in the patent documentation, offering clarity on the practical aspects of adopting this technology.
Q: Why does the conventional synthesis of Azilsartan suffer from low yields?
A: Conventional methods, such as those described in US Pat 5583141, involve reacting a benzimidazole derivative containing an unstable ethoxy group. During the reaction conditions, this group is prone to de-ethylation, generating a series of difficult-to-remove impurities that drastically reduce the overall yield to approximately 23%.
Q: How does the novel route in CN103880830A improve purity?
A: The novel route utilizes a convergent strategy where the oxadiazole ring is pre-formed on the biphenyl fragment before coupling. This avoids the harsh conditions on the sensitive benzimidazole core, eliminating the formation of de-ethylated impurities and ensuring final product purity greater than 99.5% by HPLC.
Q: What are the preferred reaction conditions for the key coupling step?
A: The coupling between the halogenated intermediate and the benzimidazole derivative is optimally performed at mild temperatures between 20°C and 60°C. Preferred solvents include toluene, tetrahydrofuran (THF), or ethyl acetate, using bases such as potassium carbonate or triethylamine to drive the nucleophilic substitution efficiently.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Azilsartan Supplier
The technical advantages outlined in patent CN103880830A demonstrate a clear path toward more efficient Azilsartan production, yet translating laboratory success to commercial reality requires experienced partnership. NINGBO INNO PHARMCHEM stands ready to leverage this advanced chemistry, bringing extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our facility is equipped with rigorous QC labs and adheres to stringent purity specifications, ensuring that every batch of Azilsartan or its intermediates meets the highest international standards. We understand the complexities of managing convergent syntheses and are prepared to optimize every unit operation to maximize yield and minimize cost for our partners.
We invite pharmaceutical companies and generic manufacturers to engage with us for a Customized Cost-Saving Analysis tailored to your specific volume requirements. Our technical procurement team is available to provide specific COA data and route feasibility assessments to help you evaluate the potential of this novel synthesis for your supply chain. By collaborating with us, you gain access to a robust supply of high-quality intermediates and APIs, secured by a manufacturing process designed for long-term stability and efficiency.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
