Advanced Vacuum-Reflux Synthesis of 2,3-Dihydroxy-6-Chloroquinoxaline for Commercial Scale-Up
The global demand for high-efficiency herbicides continues to drive innovation in the synthesis of critical agrochemical intermediates. Patent CN111454223B introduces a transformative synthetic method for 2,3-dihydroxy-6-chloroquinoxaline, a pivotal precursor in the production of Quizalofop-p-ethyl. This technical breakthrough addresses long-standing challenges in purity and yield that have historically plagued the manufacturing of this key heterocyclic compound. By shifting from traditional high-temperature rearrangement pathways to a controlled vacuum-reflux condensation strategy, the patent outlines a process that significantly mitigates side reactions and tar formation. For R&D directors and procurement specialists, understanding this shift is crucial, as it represents a move towards more sustainable and economically viable production methodologies. The ability to produce a standard substance with qualified content not only ensures the quality of the final herbicide but also streamlines the regulatory compliance process for exported agricultural chemicals.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of quinoxaline derivatives has relied heavily on the thermal rearrangement of precursors such as p-chloro-o-nitro-n-acetanilide in strong alkaline solutions. This conventional pathway is fraught with significant chemical inefficiencies, primarily due to the generation of persistent impurities. As illustrated in the reaction mechanisms below, the rearrangement process inherently produces approximately 5-10% of 2,3-dihydroxy-6-chloroquinoxaline as an unavoidable byproduct. These impurities possess chemical properties strikingly similar to the desired intermediate, making their separation via standard purification techniques exceptionally difficult and costly. Furthermore, alternative literature methods involving the reaction of 4-chlorophthaline with diethyl oxalate under atmospheric reflux suffer from severe thermal degradation. The high temperatures required for atmospheric reflux often lead to the formation of intractable tars and uncontrolled side reactions, drastically reducing the overall yield and complicating the downstream processing workflow.
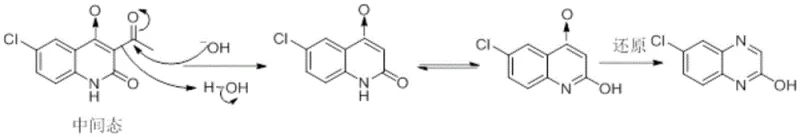
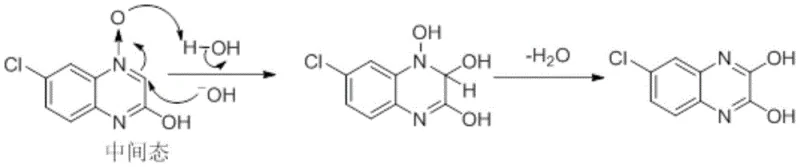
The Novel Approach
In stark contrast to these legacy methods, the novel approach detailed in the patent utilizes a direct condensation reaction under negative pressure. This method employs 4-chloro-o-phenylenediamine and diethyl oxalate as the primary starting materials, reacting them in the presence of a catalytic base. The core innovation lies in the application of vacuum rectification during the reflux stage. By maintaining the reaction system at a negative pressure ranging from -0.03 to -0.08 MPa, the process effectively lowers the boiling point of the reaction mixture. This thermal moderation prevents the thermal decomposition of sensitive intermediates and suppresses the polymerization reactions that lead to tar formation. Moreover, the continuous removal of the ethanol byproduct through rectification serves a dual purpose: it prevents the reverse reaction and thermodynamically drives the equilibrium towards the formation of the desired quinoxaline ring, thereby accelerating the reaction rate and enhancing the final product quality.
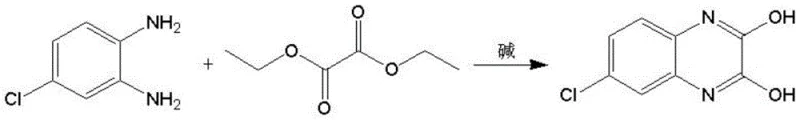
Mechanistic Insights into Base-Catalyzed Vacuum Condensation
The mechanistic superiority of this synthesis lies in the synergistic effect of base activation and Le Chatelier's principle applied through vacuum distillation. When a base such as sodium ethoxide or trioctylamine is introduced into the reaction vessel, it acts as a potent activator for the amine groups on the 4-chloro-o-phenylenediamine substrate. This activation significantly lowers the activation energy required for the nucleophilic attack on the carbonyl carbons of the diethyl oxalate. Consequently, the cyclization can proceed efficiently at lower temperatures compared to non-catalyzed thermal methods. The reduced thermal load is critical for preserving the structural integrity of the chloro-substituted aromatic ring, preventing dehalogenation or other thermal degradations that often occur under vigorous atmospheric reflux conditions. This precise control over reaction kinetics ensures that the formation of the quinoxaline core is both rapid and selective.
Furthermore, the management of the reaction equilibrium is masterfully handled through the physical removal of the ethanol byproduct. In a closed or atmospheric system, the accumulation of ethanol would eventually establish an equilibrium that limits the conversion of reactants to products. However, by integrating a decompression rectification device, the system continuously strips ethanol from the reaction liquor as it forms. This constant removal shifts the chemical equilibrium decisively to the right, ensuring near-complete consumption of the expensive diamine starting material. This mechanistic feature not only boosts the theoretical yield but also simplifies the post-reaction workup, as the reaction mixture contains fewer unreacted starting materials and side products, facilitating a cleaner crystallization process.
How to Synthesize 2,3-Dihydroxy-6-Chloroquinoxaline Efficiently
The operational protocol for this synthesis is designed for robustness and scalability, making it highly suitable for industrial adoption. The process begins with the precise charging of stoichiometric amounts of 4-chloro-o-phenylenediamine and diethyl oxalate, along with the selected base catalyst, into a reactor equipped for vacuum distillation. The system is then sealed and subjected to a controlled negative pressure before heating is initiated. This sequence is vital to prevent oxidative degradation and to ensure immediate removal of volatiles upon reaching the reflux temperature. Following the reaction period, typically lasting between 2 to 5 hours depending on the specific vacuum level and base used, the mixture undergoes a rigorous purification sequence involving hot filtration and low-temperature crystallization. The detailed standardized synthesis steps for replicating this high-yield process are outlined in the guide below.
- Charge 4-chloro-o-phenylenediamine, diethyl oxalate, and a base (sodium ethoxide or trioctylamine) into a reactor equipped with a vacuum rectification column.
- Apply negative pressure (-0.03 to -0.08 MPa) and heat to reflux, continuously distilling off the ethanol byproduct to drive the reaction forward.
- After reaction completion, decolorize with activated carbon, filter hot, and crystallize the filtrate at 0-5°C to obtain the pure product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this vacuum-reflux technology offers substantial strategic advantages beyond mere chemical efficiency. The primary benefit is the drastic simplification of the purification train. Traditional methods often require extensive chromatographic separations or multiple recrystallization steps to remove the stubborn 5-10% impurity profile associated with rearrangement routes. By virtually eliminating these impurities at the source through better reaction control, the new method reduces the consumption of solvents, activated carbon, and energy required for purification. This streamlined workflow translates directly into a significant reduction in manufacturing costs, allowing for more competitive pricing of the final agrochemical intermediate without sacrificing margin.
- Cost Reduction in Manufacturing: The elimination of high-temperature tar formation means that reactor downtime for cleaning is significantly minimized, and the yield of usable product per batch is maximized. By avoiding the need for complex separation technologies to remove structurally similar impurities, the operational expenditure on specialized resins or extensive solvent recovery systems is drastically lowered. Additionally, the higher conversion rates mean that less raw material is wasted, optimizing the cost of goods sold (COGS) for every kilogram of 2,3-dihydroxy-6-chloroquinoxaline produced.
- Enhanced Supply Chain Reliability: The use of readily available starting materials like 4-chloro-o-phenylenediamine and diethyl oxalate ensures a stable supply chain, free from the bottlenecks associated with specialized nitro-compound precursors. The robustness of the vacuum reflux process also implies a wider operating window, reducing the risk of batch failures due to minor temperature fluctuations. This reliability is critical for maintaining consistent delivery schedules to downstream herbicide manufacturers, ensuring that the production of Quizalofop-p-ethyl is not interrupted by intermediate shortages.
- Scalability and Environmental Compliance: From an environmental perspective, the reduction in tar and heavy organic waste simplifies effluent treatment, aligning with increasingly stringent global environmental regulations. The process is inherently scalable; vacuum rectification is a standard unit operation in fine chemical plants, meaning that scaling from pilot batches to multi-ton commercial production involves minimal re-engineering. This ease of scale-up ensures that suppliers can rapidly respond to surges in market demand for herbicide intermediates during peak agricultural seasons.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis method. These insights are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing a clear picture of the technology's capabilities. Understanding these nuances is essential for technical teams evaluating the feasibility of integrating this process into existing manufacturing lines.
Q: Why is the vacuum reflux method superior to atmospheric reflux for this synthesis?
A: Vacuum reflux lowers the boiling point of the reaction system, preventing the formation of tar and side products associated with high-temperature atmospheric reflux, while simultaneously removing ethanol to shift equilibrium.
Q: What is the primary impurity challenge in traditional synthesis routes?
A: Traditional rearrangement routes often generate 5-10% of difficult-to-separate impurities like 2,3-dihydroxy-6-chloroquinoxaline derivatives, which compromise the quality of the final herbicide.
Q: How does the choice of base affect the reaction kinetics?
A: Using bases like sodium ethoxide or trioctylamine activates the reaction substrate, lowering the activation energy and promoting the cyclization process without requiring excessive thermal energy.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2,3-Dihydroxy-6-Chloroquinoxaline Supplier
At NINGBO INNO PHARMCHEM, we recognize that the quality of the final herbicide is inextricably linked to the purity of its intermediates. Our technical team has extensively analyzed the vacuum-reflux methodology described in CN111454223B and possesses the expertise to implement this advanced synthesis route effectively. We bring extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory success to industrial reality is seamless. Our facilities are equipped with state-of-the-art vacuum reactors and rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of 2,3-dihydroxy-6-chloroquinoxaline meets the exacting standards required for global agrochemical registration.
We invite potential partners to engage with our technical procurement team to discuss how this optimized synthesis route can enhance your supply chain resilience. By leveraging our manufacturing capabilities, you can secure a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to contact us today to request specific COA data and route feasibility assessments, ensuring that your production of Quizalofop-p-ethyl is built on a foundation of high-quality, reliably sourced intermediates.
