Advanced Ultrasonic Synthesis of Z-Beta-Selenocyanato Thioesters for Commercial Pharmaceutical Applications
The pharmaceutical and fine chemical industries are constantly seeking robust methodologies for incorporating selenium into organic frameworks, given the element's profound biological significance in enzyme modulation and antioxidant activity. Patent CN109438308B introduces a groundbreaking approach for the synthesis of (Z)-β-selenocyanatopropene thioester compounds, a novel class of organic intermediates that possess both a reactive selenocyanate group and a modifiable thioester moiety. This technology leverages a synergistic combination of ultrasonic radiation and biomass-derived deep eutectic solvents (DES) to facilitate a three-component addition reaction. Unlike traditional methods that often rely on toxic volatile organic solvents or expensive transition metal catalysts, this innovation utilizes a Choline Chloride (ChCl) and glycolic acid system to achieve high selectivity and yield under ambient conditions. For R&D directors and procurement specialists, this represents a paradigm shift towards greener, more cost-effective manufacturing of high-value selenium-containing scaffolds.
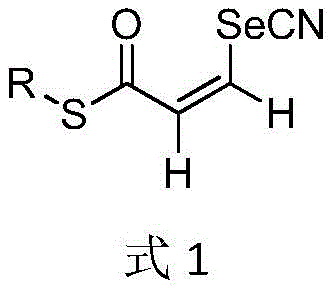
The structural versatility of these compounds, as depicted in the general formula, allows for extensive downstream functionalization, making them invaluable precursors for drug discovery programs targeting various therapeutic areas. The presence of the Z-configuration is critical for specific biological interactions, and this patent provides a reliable method to access this stereoisomer exclusively.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of organoselenium compounds has been fraught with significant challenges that hinder their widespread adoption in large-scale pharmaceutical manufacturing. Conventional routes frequently necessitate the use of hazardous reagents such as elemental selenium powder or unstable selenol derivatives, which pose severe safety risks and require specialized handling equipment. Furthermore, traditional catalytic systems often depend on precious metals like palladium or copper, which not only inflate the raw material costs but also introduce the risk of heavy metal contamination in the final Active Pharmaceutical Ingredient (API). Removing these trace metals to meet stringent regulatory limits adds complex purification steps, such as scavenging or repeated recrystallization, thereby extending the overall production cycle. Additionally, many existing protocols require elevated temperatures or inert atmospheres, leading to substantial energy consumption and operational complexity that is ill-suited for sustainable chemical production.
The Novel Approach
The methodology disclosed in CN109438308B effectively circumvents these historical bottlenecks by employing a biomimetic catalytic system driven by ultrasonic energy. By utilizing a deep eutectic solvent composed of choline chloride and glycolic acid, the reaction proceeds through a highly efficient three-component coupling of propyne thioesters, potassium selenocyanate, and water. This approach eliminates the need for external transition metal catalysts entirely, relying instead on the unique hydrogen-bonding network of the DES to activate the nucleophiles. The integration of ultrasonic irradiation further enhances the reaction kinetics through acoustic cavitation, which improves mass transfer and ensures uniform mixing without the need for thermal input. Consequently, this novel pathway delivers superior regioselectivity for the Z-isomer while operating at room temperature, offering a streamlined, one-pot solution that drastically simplifies the workflow for producing high-purity organoselenium intermediates.
Mechanistic Insights into Ultrasonic-Assisted DES Catalysis
The core of this technological breakthrough lies in the dual functionality of the ChCl/glycolic acid deep eutectic solvent, which acts simultaneously as the reaction medium and the catalyst. Mechanistically, the hydrogen bond donor (glycolic acid) and acceptor (choline chloride) create a structured solvent cage that stabilizes the transition state of the addition reaction. The acidic protons in the glycolic acid likely activate the triple bond of the propyne thioester substrate, making it more susceptible to nucleophilic attack by the selenocyanate anion generated in situ from potassium selenocyanate. This activation lowers the energy barrier for the addition step, allowing the reaction to proceed rapidly even at ambient temperatures. The specific molar ratio of 1:2 between ChCl and glycolic acid is critical, as deviations from this stoichiometry disrupt the optimal hydrogen bonding network, leading to diminished yields. This precise tuning of the solvent environment ensures that the reaction follows a specific trajectory that favors the formation of the thermodynamically stable Z-configuration.
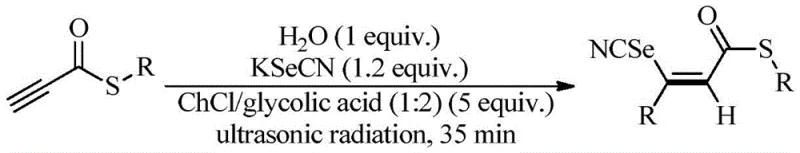
Furthermore, the ultrasonic waves play a pivotal role in the mechanistic pathway by generating microscopic bubbles that collapse violently, creating localized hot spots and high-pressure zones. These physical phenomena enhance the solubility of the inorganic salt (KSeCN) within the organic-compatible DES matrix, ensuring a homogeneous reaction mixture that maximizes collision frequency between reactants. This physical activation complements the chemical catalysis provided by the DES, resulting in a synergistic effect that drives the conversion to completion within a remarkably short timeframe of 35 minutes. The absence of radical initiators or harsh oxidants further preserves the integrity of sensitive functional groups on the thioester substrate, demonstrating excellent functional group tolerance.
How to Synthesize (Z)-β-Selenocyanato Propylene Thioester Efficiently
To implement this synthesis in a laboratory or pilot plant setting, operators must adhere to specific parameters regarding reagent ratios and ultrasonic settings to maximize efficiency. The process begins with the preparation of the deep eutectic solvent by mixing choline chloride and glycolic acid, followed by the addition of the propyne thioester substrate and potassium selenocyanate. Water is introduced as a co-reactant in a stoichiometric amount to facilitate the hydro-selenation process. The mixture is then subjected to ultrasonic radiation at a power of 35W and a frequency of 40KHz, which has been empirically determined to be the optimal condition for balancing reaction rate and product purity. Detailed standardized synthesis steps follow below to ensure reproducibility and safety during operation.
- Prepare the reaction mixture by combining propyne thioester, potassium selenocyanate, water, and ChCl/glycolic acid deep eutectic solvent in a round-bottom flask.
- Subject the mixture to ultrasonic radiation at optimized power (35W) and frequency (40KHz) for approximately 35 minutes at room temperature.
- Extract the product with ethyl acetate, concentrate the filtrate, and purify via silica gel column chromatography to obtain the high-purity target compound.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this ultrasonic-assisted DES methodology offers compelling economic and logistical benefits that directly impact the bottom line. The primary advantage stems from the elimination of expensive transition metal catalysts and volatile organic solvents, which traditionally constitute a significant portion of the raw material budget. By replacing these with inexpensive, bio-based components like choline chloride and glycolic acid, manufacturers can achieve substantial cost savings in reagent procurement. Moreover, the simplified workup procedure, which avoids complex metal scavenging steps, reduces the consumption of auxiliary materials such as silica gel and filtration media, further driving down the cost of goods sold (COGS). The mild reaction conditions also translate to lower energy expenditures, as there is no need for heating mantles or cryogenic cooling systems, aligning perfectly with corporate sustainability goals.
- Cost Reduction in Manufacturing: The removal of precious metal catalysts eliminates the need for costly purification protocols required to meet residual metal specifications in pharmaceuticals. This streamlining of the downstream processing significantly reduces labor hours and waste disposal costs associated with heavy metal containment. Additionally, the high atom economy of the three-component reaction ensures that the majority of the starting materials are incorporated into the final product, minimizing raw material waste and maximizing overall process efficiency.
- Enhanced Supply Chain Reliability: The starting materials, including propyne thioesters and potassium selenocyanate, are commercially available commodities with stable supply chains, reducing the risk of production delays due to raw material shortages. The robustness of the reaction against moisture and oxygen, facilitated by the DES medium, means that strict anhydrous conditions are not required, simplifying storage and handling logistics. This resilience allows for more flexible scheduling and inventory management, ensuring a consistent flow of high-quality intermediates to downstream synthesis units.
- Scalability and Environmental Compliance: The use of non-volatile, biodegradable deep eutectic solvents significantly reduces the emission of Volatile Organic Compounds (VOCs), making it easier for facilities to comply with increasingly stringent environmental regulations. The scalability of ultrasonic reactors is well-established in the chemical industry, allowing for a seamless transition from gram-scale laboratory optimization to multi-kilogram commercial production without losing reaction efficiency. This scalability ensures that the supply of these critical selenium intermediates can be ramped up quickly to meet market demand without compromising on quality or safety standards.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and optimization of this synthetic route, derived directly from the experimental data and beneficial effects outlined in the patent documentation. Understanding these nuances is essential for process chemists aiming to integrate this technology into their existing workflows. The answers provide clarity on solvent selection, reaction parameters, and product isolation strategies to ensure successful adoption.
Q: What are the key advantages of using biomass deep eutectic solvents in this synthesis?
A: The use of ChCl/glycolic acid deep eutectic solvents eliminates the need for volatile organic solvents and expensive transition metal catalysts. This dual-function medium acts as both catalyst and solvent, significantly simplifying downstream purification and reducing environmental impact while maintaining high reaction efficiency.
Q: How does ultrasonic assistance improve the reaction yield compared to conventional stirring?
A: Ultrasonic radiation generates cavitation effects that enhance mass transfer and activate the reagents more effectively than mechanical stirring. Experimental data indicates that optimizing ultrasonic power to 35W and frequency to 40KHz drastically shortens reaction time to 35 minutes while boosting yields to over 80%.
Q: Is this synthetic route scalable for industrial production of API intermediates?
A: Yes, the process operates under mild room temperature conditions and utilizes commercially available raw materials like propyne thioesters and potassium selenocyanate. The absence of harsh conditions and the use of recyclable solvents make this method highly suitable for commercial scale-up of complex pharmaceutical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable (Z)-β-Selenocyanato Propylene Thioester Supplier
As the global demand for specialized organoselenium intermediates continues to rise, partnering with an experienced CDMO like NINGBO INNO PHARMCHEM ensures access to cutting-edge synthetic technologies and reliable supply. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, leveraging our state-of-the-art facilities to deliver products with stringent purity specifications. Our rigorous QC labs are equipped to analyze complex selenium-containing compounds, ensuring that every batch meets the highest international standards for pharmaceutical applications. By adopting the advanced ultrasonic-assisted DES methodology, we can offer our clients a competitive edge through faster turnaround times and superior cost-efficiency.
We invite potential partners to contact our technical procurement team to discuss how this innovative synthesis can be tailored to your specific project needs. Request a Customized Cost-Saving Analysis today to understand the economic impact of switching to this green chemistry platform. Our experts are ready to provide specific COA data and route feasibility assessments to support your R&D and commercialization efforts, ensuring a seamless transition from benchtop discovery to full-scale manufacturing.
