Revolutionizing Naphthalene Derivative Synthesis: Scalable Production of High-Purity API Intermediates
The Chinese patent CN109734642B introduces a groundbreaking synthetic methodology for naphthone-sulfoxide ylide hybrids, representing a significant advancement in the field of functionalized naphthalene derivative production. This innovative approach addresses longstanding challenges in conventional synthesis routes by providing a single-step tandem reaction that efficiently generates key intermediates for various biologically active compounds. The technology enables precise structural modifications through multiple divergent pathways, offering unprecedented flexibility for pharmaceutical manufacturers seeking high-purity intermediates with complex substitution patterns. By leveraging rhodium-catalyzed chemistry under mild conditions, this method eliminates several cumbersome steps typically required in traditional approaches while maintaining excellent atom economy and operational simplicity. The resulting intermediates serve as versatile building blocks for numerous pharmaceutical applications, particularly in the development of compounds requiring specific hydroxyl and carbonyl functionalization patterns on naphthalene scaffolds.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic routes for functionalized naphthalene derivatives such as 4-hydroxy-1-naphthoates, 3-hydroxy-4-oxo-1-naphthoates, and related compounds have been plagued by multiple significant limitations that hinder their practical application in pharmaceutical manufacturing. These conventional approaches typically require lengthy multi-step sequences with poor atom economy, often necessitating harsh reaction conditions that compromise both yield and purity. The use of expensive reagents and specialized catalysts further increases production costs while introducing potential contamination risks that complicate regulatory compliance for pharmaceutical applications. Additionally, many existing methods suffer from limited substrate scope and poor functional group tolerance, restricting their applicability to specific derivative types rather than providing a comprehensive solution for diverse naphthalene-based intermediates. The requirement for multiple protection/deprotection steps not only extends manufacturing timelines but also generates substantial waste streams that conflict with modern green chemistry principles and environmental regulations.
The Novel Approach
The patented methodology overcomes these limitations through an elegant rhodium-catalyzed tandem reaction between arylformyl sulfoxide ylides and alpha-diazocarbonyl compounds that directly produces naphthone-sulfoxide ylide hybrids in a single operation. This innovative approach operates under relatively mild conditions (80-120°C) with excellent functional group compatibility across diverse substituents including halogens, alkyl groups, and various aryl moieties. The process demonstrates remarkable versatility through multiple downstream transformation pathways that efficiently generate different classes of functionalized naphthalene derivatives from the same key intermediate. Crucially, the method achieves high atom economy without requiring additional acid or base catalysts during the core transformation step, significantly simplifying process design and reducing potential impurity formation. The compatibility with fluorinated alcohol solvents like TFE and HFIP further enhances practical utility by enabling cleaner reactions with fewer side products and simplified purification protocols.
Mechanistic Insights into Rhodium-Catalyzed Tandem Reaction
The core innovation lies in the rhodium-catalyzed tandem reaction mechanism that enables direct formation of naphthone-sulfoxide ylide hybrids from readily available starting materials. The process begins with rhodium complex activation of the diazo compound, followed by carbene transfer to the sulfoxide ylide component through a carefully orchestrated sequence of bond formations and rearrangements. This cascade reaction proceeds through a series of well-defined intermediates that ultimately deliver the fused naphthalene ring system with precise regiochemical control. The rhodium catalyst's compatibility with various forms ([RhCp*(MeCN)3](SbF6)2, [RhCp*(OAc)2]2, [RhCp*Cl2]2) provides flexibility for process optimization while maintaining consistent performance across different substrate combinations. The mechanism avoids problematic side reactions through careful control of electronic effects imparted by both the sulfoxide moiety and the rhodium catalyst system.
Impurity control is achieved through multiple complementary mechanisms inherent to this synthetic approach. The use of fluorinated alcohol solvents creates a unique microenvironment that suppresses common side reactions while promoting selective product formation. The precise stoichiometric control between reactants (1-2:1 ratio of sulfoxide ylide to diazo compound) prevents dimerization or oligomerization pathways that typically generate impurities in similar transformations. Additionally, the absence of strong acids or bases during the key cyclization step eliminates common degradation pathways that would otherwise compromise product purity. The resulting intermediates demonstrate excellent stability profiles that facilitate straightforward purification through standard chromatographic techniques without requiring specialized equipment or hazardous reagents.
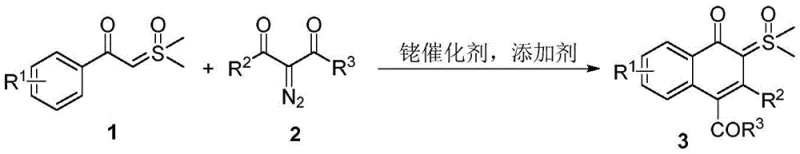
How to Synthesize Naphthone-Sulfoxide Ylide Hybrid Efficiently
This patented synthesis route represents a significant advancement in producing high-value naphthalene-based intermediates through a streamlined process that eliminates multiple steps required by conventional methods. The methodology leverages readily available starting materials and operates under practical manufacturing conditions that can be readily implemented at commercial scale. Detailed standardized synthesis procedures are provided below to ensure consistent production quality and yield across different manufacturing environments.
- Combine arylformyl sulfoxide ylide with alpha-diazocarbonyl compound in rhodium-catalyzed tandem reaction using appropriate solvent system
- Optimize reaction conditions including temperature control between 80-120°C under inert atmosphere protection
- Purify resulting naphthone-sulfoxide ylide hybrid intermediate through silica gel column chromatography
Commercial Advantages for Procurement and Supply Chain Teams
This innovative synthesis platform addresses critical pain points in pharmaceutical supply chains by providing reliable access to complex naphthalene intermediates through a robust manufacturing process that minimizes common production bottlenecks. The methodology delivers significant operational improvements that directly impact procurement efficiency and supply chain resilience while maintaining strict quality standards required for pharmaceutical applications.
- Cost Reduction in Manufacturing: The elimination of multiple synthetic steps reduces overall production costs through decreased raw material consumption, lower energy requirements, and reduced waste generation. The process operates without requiring expensive transition metal catalysts in downstream transformations, avoiding costly metal removal steps that typically add significant expense to traditional manufacturing routes. The compatibility with standard processing equipment further enhances cost efficiency by eliminating capital expenditure requirements for specialized manufacturing infrastructure.
- Enhanced Supply Chain Reliability: The use of readily available starting materials with established supply chains ensures consistent access to critical raw materials regardless of market fluctuations. The robust nature of the transformation tolerates minor variations in starting material quality while maintaining consistent product specifications, reducing quality-related supply disruptions. Multiple solvent options provide flexibility to adapt to regional material availability constraints without requiring process revalidation.
- Scalability and Environmental Compliance: The process demonstrates excellent scalability from laboratory to commercial production with consistent yield and purity profiles across different batch sizes. The reduced number of processing steps minimizes solvent usage and waste generation compared to conventional routes, aligning with green chemistry principles and reducing environmental compliance burdens. The absence of hazardous reagents simplifies waste treatment protocols while improving workplace safety metrics.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial concerns regarding implementation of this patented technology in pharmaceutical manufacturing environments.
Q: How does this novel synthesis method improve upon conventional approaches for naphthalene derivatives?
A: The rhodium-catalyzed tandem reaction provides a single-step synthesis route that eliminates multiple protection/deprotection steps required in conventional methods, significantly improving atom economy while maintaining high purity levels without requiring harsh reaction conditions.
Q: What specific advantages does this technology offer for pharmaceutical manufacturing scale-up?
A: The process demonstrates excellent substrate scope with various functional groups while maintaining consistent yields across different solvents, particularly in fluorinated alcohols which enable simplified purification and reduced solvent waste during commercial scale-up.
Q: How does this approach address purity requirements for API intermediates?
A: The selective transformation pathways allow for precise control over regiochemistry without requiring additional purification steps, while the absence of transition metal catalysts in downstream transformations eliminates potential metal contamination concerns in final pharmaceutical products.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Naphthone-Sulfoxide Ylide Hybrid Supplier
NINGBO INNO PHARMCHEM brings extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production of complex pharmaceutical intermediates like naphthone-sulfoxide ylide hybrids. Our commitment to stringent purity specifications ensures consistent product quality meeting global regulatory requirements, supported by state-of-the-art QC labs equipped for comprehensive analytical testing including advanced chromatographic methods and structural verification techniques. Our technical team works closely with clients to optimize manufacturing processes for specific requirements while maintaining robust supply chain continuity through strategic raw material sourcing and redundant production capabilities.
Request our Customized Cost-Saving Analysis today to understand how our manufacturing expertise can optimize your supply chain for naphthone-sulfoxide ylide hybrid intermediates. Contact our technical procurement team to receive specific COA data and route feasibility assessments tailored to your production requirements.
