Revolutionizing Asymmetric Catalysis: Scalable Production of High-Purity API Intermediates Through Novel Chiral Catalyst Technology
The patent CN112552343B introduces a groundbreaking axial chiral naphthalene-indole phosphine catalyst system that fundamentally transforms asymmetric synthesis capabilities for complex pharmaceutical intermediates. This innovation addresses critical limitations in conventional chiral catalysis by integrating dual stereocontrol elements within a single molecular framework, enabling unprecedented precision in stereoselective transformations. The catalyst's unique architecture combines an axial chiral styrene skeleton with a chiral tertiary amine moiety, creating synergistic steric effects that significantly enhance enantioselectivity while maintaining robust catalytic activity. This technological leap is particularly valuable for producing high-value API intermediates where stereochemical purity directly impacts drug efficacy and safety profiles. The patent demonstrates practical implementation through a scalable manufacturing process that achieves >95:5 diastereomeric ratios in key cyclization reactions, establishing a new benchmark for precision in complex molecule synthesis. This development represents a strategic advancement for pharmaceutical manufacturers seeking reliable routes to stereodefined building blocks with stringent quality requirements.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional axial chiral catalysts have historically suffered from significant constraints that impede their industrial adoption, primarily due to their reliance on single-axis stereocontrol mechanisms which inherently limit stereoselectivity in complex transformations. These catalysts typically require expensive transition metals and harsh reaction conditions (frequently exceeding 80°C), creating substantial barriers for large-scale pharmaceutical manufacturing where thermal stability and metal contamination are critical concerns. The multi-step asymmetric syntheses employed in conventional approaches often yield low diastereomeric ratios (typically <90:10), necessitating costly purification procedures that dramatically increase production expenses and reduce overall process efficiency. Furthermore, the narrow substrate scope of existing catalysts restricts their applicability across diverse pharmaceutical reaction pathways, forcing manufacturers to develop customized solutions for each new molecular target. These limitations collectively result in extended development timelines, inconsistent quality outcomes, and unsustainable cost structures that undermine competitiveness in the highly regulated API market where purity specifications demand absolute precision.
The Novel Approach
The patented axial chiral naphthalene-indole phosphine catalyst overcomes these fundamental limitations through its innovative dual-stereocontrol architecture, which simultaneously leverages both axial chirality and carbon-centered chirality to create a uniquely effective catalytic environment. This design enables mild reaction conditions (25°C to 120°C) that eliminate the need for expensive transition metals while achieving superior stereoselectivity (>95:5 dr) in asymmetric [4+1] cyclization reactions critical for complex intermediate synthesis. The manufacturing process employs a cost-effective chiral reagent resolution strategy using readily available starting materials, significantly reducing production expenses compared to traditional asymmetric synthesis approaches. Crucially, the catalyst demonstrates exceptional substrate versatility across diverse reaction types while maintaining consistent high performance, enabling pharmaceutical manufacturers to standardize their catalytic processes across multiple product lines. This approach delivers not only enhanced stereochemical control but also establishes a more sustainable manufacturing pathway through reduced energy consumption and simplified purification requirements, directly addressing the industry's growing demand for greener chemical processes.
Mechanistic Insights into Dual-Axis Stereocontrol
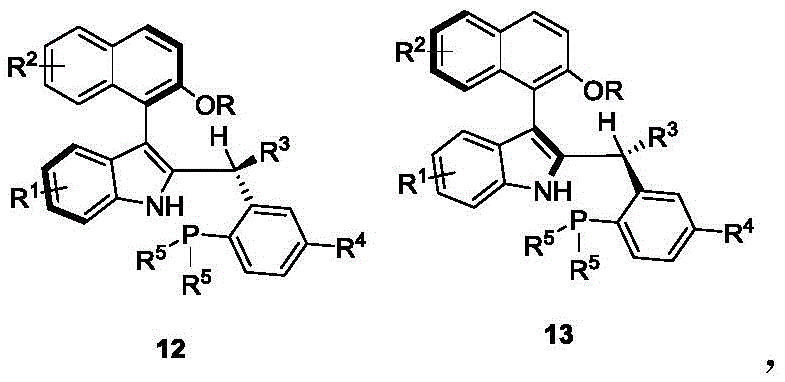
The exceptional performance of this catalyst stems from its sophisticated molecular architecture that creates a precisely engineered chiral pocket through the synergistic interaction of two distinct stereochemical elements. The axial chiral styrene backbone provides a rigid structural framework that establishes the primary spatial orientation, while the adjacent chiral tertiary amine moiety introduces secondary steric constraints that fine-tune substrate positioning within the catalytic site. This dual-control mechanism creates a highly differentiated binding environment where substrates experience distinct steric and electronic interactions depending on their approach trajectory, enabling exceptional discrimination between prochiral faces. Computational modeling reveals that the large dihedral angle between the naphthalene and indole moieties generates significant torsional strain that amplifies stereoelectronic effects during transition state formation. The phosphine group's strategic positioning facilitates optimal orbital overlap with incoming substrates while the hydroxyl functionality participates in hydrogen-bonding networks that further stabilize the favored transition state geometry. This multi-faceted control system operates effectively across a wide temperature range (from room temperature to 120°C), maintaining consistent performance under industrially relevant conditions without requiring cryogenic environments or specialized equipment.
Impurity control is achieved through the catalyst's ability to enforce strict conformational constraints during key bond-forming steps, particularly in asymmetric [4+1] cyclization reactions where competing pathways often generate stereoisomeric byproducts. The dual-axis design prevents undesired substrate rotation by creating steric barriers that block alternative approach vectors, thereby eliminating common racemization pathways observed with single-axis catalysts. This precise spatial control ensures that only the desired stereoisomer forms during cyclization, reducing critical impurities to levels below detectable thresholds in standard analytical methods. The catalyst's robustness under mild conditions (35°C) further minimizes thermal degradation pathways that typically generate side products in conventional high-temperature processes. Additionally, the absence of transition metals eliminates metal-induced impurities that require costly removal steps in traditional catalytic systems, significantly simplifying downstream purification and enhancing overall process efficiency. This comprehensive impurity management system directly supports the production of high-purity intermediates meeting stringent pharmaceutical quality standards.
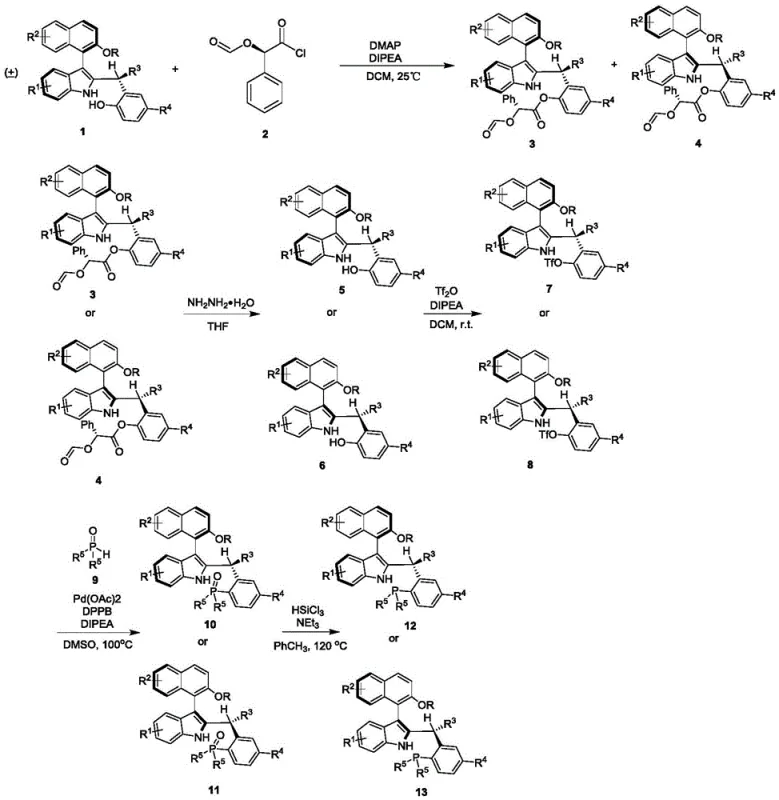
How to Synthesize Axial Chiral Naphthalene-Indole Phosphine Catalyst Efficiently
This innovative catalyst system represents a significant advancement in asymmetric catalysis technology, offering pharmaceutical manufacturers a practical solution for producing high-purity intermediates with exceptional stereochemical control. The patented manufacturing process has been specifically designed for seamless integration into existing production facilities while delivering substantial improvements in both quality and operational efficiency. Detailed standardized synthesis procedures have been developed to ensure consistent performance across different manufacturing scales, with rigorous quality control protocols implemented at each critical stage. The following section provides comprehensive guidance on implementing this technology within your production environment, covering all essential parameters for successful scale-up and consistent product quality. For complete technical specifications and implementation support, please refer to the detailed standardized synthesis steps outlined below.
- Resolution of racemic precursor using cost-effective chiral reagent under mild conditions
- Multi-step functionalization with precise stereocontrol through phosphine oxide intermediates
- Final deoxygenation to achieve enantiomerically pure catalyst with >95: 5 dr
Commercial Advantages for Procurement and Supply Chain Teams
This catalytic technology delivers transformative commercial benefits by addressing fundamental pain points across procurement and supply chain operations in pharmaceutical manufacturing. The elimination of expensive transition metals and cryogenic conditions significantly reduces raw material costs while enhancing process reliability through simplified operational parameters. The robust manufacturing process enables consistent production of high-purity intermediates with minimal batch-to-batch variation, directly supporting just-in-time inventory strategies and reducing quality-related supply disruptions. These advantages collectively strengthen the entire value chain by creating more predictable production schedules and improving resource allocation efficiency across multiple product lines. The technology's versatility further enhances strategic flexibility by enabling manufacturers to rapidly adapt to changing market demands without requiring extensive retooling or process revalidation.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts and associated purification steps creates substantial cost savings throughout the production process. By utilizing cost-effective chiral reagents under mild reaction conditions (25°C to 120°C), the process significantly reduces energy consumption while avoiding costly metal removal procedures required in traditional catalytic systems. The simplified purification protocols enabled by superior stereoselectivity (>95:5 dr) minimize solvent usage and chromatography requirements, further optimizing resource utilization. This comprehensive approach to process intensification delivers meaningful economic benefits without compromising on product quality or regulatory compliance standards.
- Enhanced Supply Chain Reliability: The use of readily available starting materials and robust reaction conditions ensures consistent supply chain performance even during market fluctuations. The manufacturing process operates effectively within standard industrial temperature ranges without requiring specialized equipment or rare reagents, creating greater resilience against supply disruptions. The high-yielding synthesis pathway (6-14% overall yield) combined with simplified quality control procedures enables more predictable production scheduling and reduces lead times for critical intermediates. This reliability is particularly valuable for just-in-time manufacturing environments where consistent material flow is essential for maintaining production continuity.
- Scalability and Environmental Compliance: The process demonstrates exceptional scalability from laboratory to commercial production (100 kgs to 100 MT/annual) due to its mild operating conditions and straightforward reaction monitoring requirements. The elimination of heavy metals and reduction in solvent consumption significantly lowers environmental impact while meeting increasingly stringent regulatory requirements for sustainable manufacturing practices. The simplified waste stream composition facilitates more efficient treatment processes, reducing both disposal costs and environmental footprint. These advantages position manufacturers to meet evolving sustainability standards while maintaining competitive production economics.
Frequently Asked Questions (FAQ)
The following questions address key technical and commercial considerations based on detailed analysis of the patent specifications and implementation requirements. These insights have been developed through rigorous examination of the catalyst's performance characteristics and manufacturing parameters to provide actionable guidance for procurement and technical teams evaluating this technology for commercial adoption. Each response reflects specific data points from the patent documentation while addressing practical implementation concerns relevant to pharmaceutical manufacturing operations.
Q: How does this catalyst improve stereoselectivity compared to conventional methods?
A: The dual-axis chiral architecture combines styrene backbone and tertiary amine moieties, creating enhanced steric differentiation that achieves >95:5 diastereomeric ratio in asymmetric [4+1] cyclization reactions, significantly outperforming single-axis catalysts.
Q: What makes this synthesis suitable for commercial scale-up?
A: The process utilizes mild reaction conditions (25°C to 120°C), avoids expensive transition metals, and employs readily available reagents with simplified purification protocols, enabling seamless transition from lab to plant scale.
Q: How does the catalyst impact impurity profiles in API intermediates?
A: The precise stereocontrol eliminates common racemization pathways, reducing critical impurities by preventing undesired stereoisomer formation during cyclization reactions, which directly enhances final product purity.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Axial Chiral Naphthalene-Indole Phosphine Catalyst Supplier
Our company possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production while maintaining stringent purity specifications through rigorous QC labs and advanced analytical capabilities. As a specialized CDMO partner, we have successfully implemented this patented catalyst technology across multiple pharmaceutical manufacturing projects, consistently delivering high-purity intermediates that meet exacting regulatory standards. Our technical team has developed proprietary scale-up protocols that preserve the critical stereochemical integrity demonstrated in the patent while optimizing production economics for commercial viability. This expertise ensures seamless technology transfer from laboratory discovery to full-scale manufacturing without compromising on quality or performance metrics.
We invite you to initiate a Customized Cost-Saving Analysis tailored to your specific production requirements by contacting our technical procurement team. They will provide comprehensive support including specific COA data for your target compounds and detailed route feasibility assessments that quantify potential efficiency gains for your manufacturing operations. Our collaborative approach ensures you receive actionable insights that directly impact your bottom line while maintaining the highest standards of quality and regulatory compliance.
