Efficient Iron-Catalyzed Synthesis of Thieno[3,4-b]indole Derivatives for Commercial Scale-up
Efficient Iron-Catalyzed Synthesis of Thieno[3,4-b]indole Derivatives for Commercial Scale-up
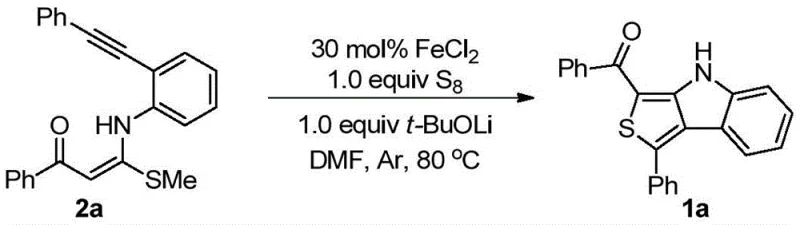
The rapid advancement of organic electronics and pharmaceutical sciences has intensified the demand for high-performance heterocyclic building blocks, specifically thieno[3,4-b]indole derivatives. A groundbreaking patent, CN111285881B, discloses a novel synthetic methodology that addresses the longstanding challenges associated with constructing these complex fused ring systems. This technology leverages a cost-effective iron-catalyzed oxidative cyclization strategy, utilizing elemental sulfur as a benign oxidant to facilitate the one-step transformation of 4-alkylthio-4-((2-(phenylethynyl)aryl)amino)but-3-en-2-one precursors into the target thieno[3,4-b]indole scaffold. For R&D directors and procurement specialists seeking reliable thieno[3,4-b]indole supplier partnerships, this innovation represents a paradigm shift towards more sustainable and economically viable manufacturing processes. The method not only simplifies the synthetic route but also ensures high purity and structural diversity, making it an ideal candidate for the commercial scale-up of complex electronic chemical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of thienoindole-containing compounds has relied heavily on multi-step sequences involving pre-functionalized thiophene or indole starting materials. These traditional pathways often necessitate the use of expensive transition metal catalysts, harsh reaction conditions, and rigorous purification protocols to remove toxic metal residues, which is particularly detrimental for electronic grade materials. Furthermore, the requirement for specialized starting materials limits the structural diversity of the final products, restricting the ability of chemists to fine-tune the optoelectronic properties necessary for next-generation organic photovoltaic cells. The reliance on precious metals also introduces significant volatility in supply chain costs and creates environmental compliance burdens related to heavy metal waste disposal. Consequently, the industry has been in urgent need of a direct, atom-economical approach that bypasses these inefficiencies while maintaining high yields and product quality standards required for high-purity OLED material production.
The Novel Approach
The methodology described in patent CN111285881B offers a transformative solution by enabling the direct construction of the thieno[3,4-b]indole core through a self-cyclization reaction of readily available enyne precursors. This innovative route employs inexpensive iron salts, specifically FeCl2, in conjunction with elemental sulfur (S8) and a base like lithium tert-butoxide to drive the oxidative cyclization under mild thermal conditions. By eliminating the need for pre-formed heterocycles, this approach drastically reduces the number of synthetic steps and minimizes waste generation. The versatility of this method is demonstrated by its broad substrate scope, accommodating various substituents on the aromatic rings without significant loss in efficiency. This streamlined process not only accelerates the timeline for cost reduction in electronic chemical manufacturing but also enhances the overall sustainability profile of the production workflow, aligning perfectly with modern green chemistry principles.
![General reaction scheme showing the conversion of 4-alkylthio-4-((2-(phenylethynyl)aryl)amino)but-3-en-2-one to thieno[3,4-b]indole derivatives using FeCl2 and S8](/insights/img/thieno-indole-synthesis-iron-catalysis-oled-supplier-20260303130523-02.webp)
Mechanistic Insights into FeCl2-Catalyzed Oxidative Cyclization
The core of this technological breakthrough lies in the unique mechanistic pathway facilitated by the iron catalyst and elemental sulfur. The reaction initiates with the coordination of the iron species to the alkyne and sulfur moieties of the substrate, activating the system for nucleophilic attack and subsequent ring closure. Elemental sulfur acts as both a sulfur source and an oxidant, promoting the formation of the thiophene ring fused to the indole system through a cascade of bond-forming events. This mechanism avoids the formation of stable off-cycle intermediates that often plague other catalytic cycles, thereby ensuring high turnover frequencies and robust reaction kinetics. The use of DMF as the solvent further stabilizes the charged intermediates, facilitating smooth progression to the final aromatic product. Understanding this mechanism is crucial for R&D teams aiming to optimize reaction parameters for specific substrate variants, ensuring consistent quality and minimizing the formation of regioisomeric impurities that could compromise device performance.
From an impurity control perspective, the mild nature of the iron-catalyzed system offers distinct advantages over harsher oxidative conditions. The selective activation of the enyne precursor prevents non-specific oxidation of sensitive functional groups, such as halogens or electron-donating methoxy groups, which are often present in advanced material designs. The reaction conditions, typically maintained around 80°C, are sufficiently energetic to drive the cyclization but gentle enough to prevent thermal degradation of the product. Post-reaction workup involves standard silica gel chromatography, which effectively separates the target thieno[3,4-b]indole from any unreacted starting material or minor byproducts. This high level of selectivity translates directly into reduced downstream processing costs and higher overall yields, providing a reliable foundation for the commercial production of high-purity organic semiconductor materials.
How to Synthesize Thieno[3,4-b]indole Derivatives Efficiently
Implementing this synthesis in a laboratory or pilot plant setting requires precise adherence to the optimized reaction parameters established in the patent data. The process begins with the careful weighing of the 4-alkylthio-4-((2-(phenylethynyl)aryl)amino)but-3-en-2-one substrate, ensuring a molar concentration within the range of 0.01-1.0M in the reaction solvent. The addition of the catalyst, oxidant, and base must be sequenced correctly to maximize initiation efficiency, typically under an inert atmosphere to prevent unwanted side reactions with atmospheric oxygen. The following guide outlines the standardized procedure derived from the exemplary embodiments, serving as a blueprint for process chemists to replicate the high yields reported in the literature.
- Prepare the reaction mixture by combining 4-alkylthio-4-((2-(phenylethynyl)aryl)amino)but-3-en-2-one with FeCl2 catalyst and elemental sulfur in DMF solvent.
- Add lithium tert-butoxide (t-BuOLi) as the base under an inert atmosphere to initiate the oxidative cyclization process.
- Heat the reaction mixture to 80°C for approximately 3 hours, then purify the resulting yellow solid via silica gel column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this iron-catalyzed synthesis route offers compelling strategic benefits that extend beyond simple chemical transformation. The shift from precious metal catalysts to abundant iron salts fundamentally alters the cost structure of the raw materials, removing exposure to the volatile pricing markets associated with palladium or rhodium. Additionally, the use of elemental sulfur, a commodity chemical with stable global supply, further insulates the production process from supply shocks. The simplicity of the reaction setup, requiring only standard heating and stirring equipment without the need for high-pressure reactors or specialized gas handling systems, lowers the barrier to entry for contract manufacturing organizations. This accessibility ensures a more resilient supply chain capable of meeting fluctuating demand schedules for critical electronic intermediates without significant lead time extensions.
- Cost Reduction in Manufacturing: The elimination of expensive noble metal catalysts results in substantial cost savings per kilogram of finished product, directly improving the gross margin for downstream applications. Furthermore, the simplified purification process reduces solvent consumption and waste disposal fees, contributing to a leaner operational expenditure model. The high atom economy of the one-step cyclization minimizes the loss of valuable carbon scaffolds, ensuring that raw material investments are converted efficiently into saleable inventory. These factors combined create a highly competitive cost position for manufacturers adopting this technology in the crowded organic electronics market.
- Enhanced Supply Chain Reliability: By relying on widely available reagents such as FeCl2, S8, and t-BuOLi, the risk of production stoppages due to material shortages is significantly mitigated. The robustness of the reaction conditions allows for flexible scheduling and batch sizing, enabling suppliers to respond rapidly to urgent orders from key accounts. The stability of the intermediates and the final product also facilitates easier storage and transportation, reducing the logistical complexities often associated with sensitive pharmaceutical or electronic intermediates. This reliability is paramount for maintaining continuous production lines in high-volume manufacturing environments.
- Scalability and Environmental Compliance: The mild reaction temperatures and ambient pressure conditions make this process inherently safer and easier to scale from gram to ton quantities without extensive re-engineering. The absence of toxic heavy metals simplifies the regulatory compliance landscape, reducing the burden of environmental reporting and hazardous waste management. This alignment with green chemistry principles not only satisfies corporate sustainability goals but also future-proofs the manufacturing process against tightening environmental regulations. Consequently, companies can achieve commercial scale-up of complex polymer additives and electronic materials with greater confidence and lower regulatory risk.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this thieno[3,4-b]indole synthesis technology. These insights are derived directly from the experimental data and beneficial effects outlined in the patent documentation, providing clarity for stakeholders evaluating the feasibility of this route for their specific applications. Understanding these details is essential for making informed decisions about process adoption and supplier selection.
Q: What are the primary advantages of using FeCl2 over precious metal catalysts for thienoindole synthesis?
A: The use of FeCl2 significantly reduces raw material costs compared to palladium or copper catalysts, while maintaining high efficiency and substrate adaptability for complex heterocyclic construction.
Q: Can this synthetic method be applied to substrates with diverse functional groups?
A: Yes, the method demonstrates excellent functional group tolerance, accommodating substituents such as fluorine, chlorine, methyl, and methoxy groups on the aromatic rings without compromising yield.
Q: What are the key applications for the synthesized thieno[3,4-b]indole derivatives?
A: These derivatives serve as critical building blocks for organic photovoltaic cells due to their strong fluorescence and light absorption properties, as well as potential applications in pharmaceutical chemistry.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Thieno[3,4-b]indole Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality heterocyclic intermediates play in the development of advanced organic photovoltaic cells and pharmaceutical agents. Our team of expert process chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless and efficient. We are committed to delivering products that meet stringent purity specifications through our rigorous QC labs, guaranteeing that every batch of thieno[3,4-b]indole derivative performs consistently in your final applications. Our state-of-the-art facilities are equipped to handle the specific requirements of iron-catalyzed reactions, maintaining the highest standards of safety and quality control throughout the manufacturing lifecycle.
We invite you to collaborate with us to leverage this cutting-edge synthesis technology for your next project. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements and purity needs. We encourage you to reach out today to obtain specific COA data and route feasibility assessments that will demonstrate the tangible value of our manufacturing capabilities. Let us help you secure a stable, cost-effective supply of these vital building blocks, empowering your R&D efforts and accelerating your time to market.
