Advanced Grignard Coupling Strategy for Scalable p-Phenylbutoxy Benzoic Acid Production
Advanced Grignard Coupling Strategy for Scalable p-Phenylbutoxy Benzoic Acid Production
The pharmaceutical industry constantly seeks robust synthetic routes for key intermediates that balance cost efficiency with environmental sustainability. Patent CN108558916B introduces a transformative synthesis process for p-phenylbutoxy benzoic acid, a critical precursor in the manufacturing of the anti-asthma drug Pranlukast. This technical disclosure outlines a streamlined three-step methodology that leverages Grignard chemistry to bypass the limitations of traditional Friedel-Crafts alkylation and expensive alcohol-based condensations. By shifting the synthetic logic to a Grignard coupling followed by nucleophilic substitution, the process eliminates the need for hazardous Lewis acids and reduces the overall step count. For R&D directors and procurement specialists, this represents a significant opportunity to optimize the supply chain for asthma medication intermediates while adhering to stricter environmental regulations regarding waste disposal and toxic reagent handling.
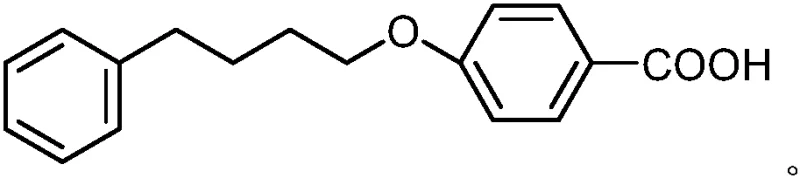
The molecular architecture of p-phenylbutoxy benzoic acid features a four-carbon linker connecting a phenyl ring to a para-substituted benzoic acid via an ether bond. Achieving this specific connectivity with high regioselectivity and minimal byproduct formation has historically been a challenge in process chemistry. The patent highlights that previous literature methods often suffered from low atom economy or required harsh conditions that compromised safety profiles. In contrast, the disclosed method utilizes readily available chlorobenzene as the aromatic source, initiating a sequence that builds the carbon chain efficiently before installing the carboxylic acid functionality. This strategic disconnection allows for better control over impurities and facilitates a more straightforward purification protocol, which is essential for meeting the stringent quality standards required for active pharmaceutical ingredient (API) precursors.
The Limitations of Conventional Methods vs. The Novel Approach
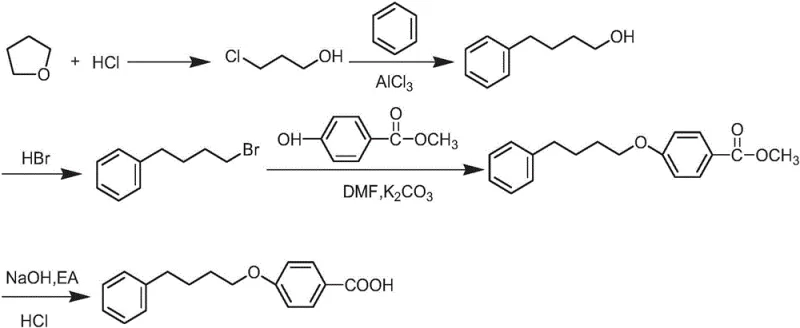
The Limitations of Conventional Methods
Historically, the synthesis of this intermediate has relied on pathways that introduce significant operational hazards and cost burdens. One prominent prior art method involves a Friedel-Crafts acylation or alkylation strategy using benzene and gamma-butyrolactone or similar derivatives, often catalyzed by aluminum trichloride. As illustrated in the reaction schemes of Document 1, these processes generate substantial quantities of acidic wastewater due to the hydrolysis of the aluminum catalyst complexes. Furthermore, the intermediates involved, such as 4-phenyl-1-butyl bromide, are highly toxic and pose severe handling risks in a manufacturing environment. Another common approach utilizes 4-phenyl-1-butanol as a starting material, reacting it with halogenated benzonitriles or benzoates. While chemically feasible, this route is economically disadvantaged because phenylbutanol is a high-cost specialty chemical compared to bulk commodities. Additionally, variations of this alcohol-based route can generate pungent and corrosive hydrobromic acid gas as a byproduct, necessitating expensive scrubbing systems and corrosion-resistant reactor equipment.
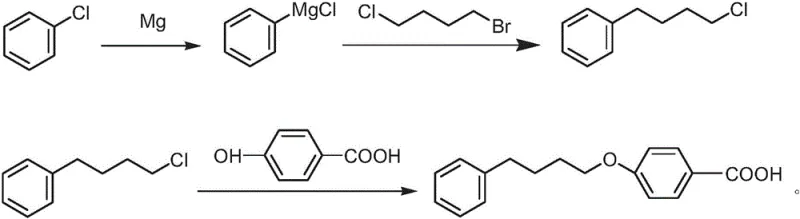
The Novel Approach
The innovation presented in patent CN108558916B fundamentally restructures the synthesis to prioritize safety and cost reduction. Instead of starting with functionalized alcohols or relying on Lewis acid catalysis, the new process begins with the formation of a Grignard reagent from chlorobenzene and magnesium. This organometallic species is then coupled with 1-bromo-4-chlorobutane, a bifunctional alkylating agent that selectively reacts at the bromine position while preserving the chlorine atom for the subsequent etherification step. This chemoselectivity is the cornerstone of the method's efficiency, allowing the construction of the 1-chloro-4-phenylbutane intermediate in high yield without protecting group strategies. The final step involves a condensation reaction with p-hydroxybenzoic acid under basic conditions, utilizing phase transfer catalysis to drive the ether formation. This approach not only simplifies the operational workflow but also drastically reduces the environmental footprint by avoiding heavy metal waste streams and toxic gas evolution.
Mechanistic Insights into Grignard Coupling and Etherification
The success of this synthetic route relies heavily on the precise control of the Grignard formation and the subsequent cross-coupling reaction. The initiation step involves the insertion of magnesium into the carbon-chlorine bond of chlorobenzene within a tetrahydrofuran solvent system. The patent specifies maintaining a reaction temperature between -20°C and 0°C during the addition of chlorobenzene to manage the exothermic nature of Grignard formation and prevent Wurtz-type homocoupling side reactions which would generate biphenyl impurities. Once the phenylmagnesium chloride is formed, it acts as a potent nucleophile in the second step. When reacted with 1-bromo-4-chlorobutane, the Grignard reagent preferentially displaces the bromide ion due to the superior leaving group ability of bromine compared to chlorine. This kinetic selectivity is crucial as it preserves the terminal chloride, which serves as the electrophilic handle for the final etherification. The reaction is quenched carefully with dilute hydrochloric acid to protonate any remaining organometallic species and facilitate phase separation, yielding the alkyl chloride intermediate as a reddish-brown liquid that is purified via distillation.
In the final condensation stage, the mechanism shifts to a classic nucleophilic aromatic substitution or aliphatic SN2 reaction depending on the specific activation of the phenol. Under the influence of a base such as potassium carbonate or sodium carbonate, p-hydroxybenzoic acid is deprotonated to form a phenoxide anion. This anion attacks the terminal carbon of the 1-chloro-4-phenylbutane chain, displacing the chloride ion to form the ether linkage. The inclusion of a phase transfer catalyst, such as polyethylene glycol or tetrabutylammonium bromide, is critical here as it enhances the solubility of the inorganic base in the organic xylene solvent, thereby accelerating the reaction rate and ensuring complete conversion. The reaction is typically refluxed at 130-140°C for approximately 24 hours to drive the equilibrium towards the product. Post-reaction workup involves washing with liquid alkali to remove unreacted phenolic acids, followed by recrystallization from methanol to achieve the high purity specifications required for pharmaceutical applications.
How to Synthesize p-Phenylbutoxy Benzoic Acid Efficiently
Implementing this synthesis requires strict adherence to anhydrous conditions during the Grignard phase and precise temperature control throughout the coupling sequence. The process is designed to be scalable, moving from laboratory glassware to industrial reactors with minimal modification to the core chemical parameters. Operators must ensure that the magnesium surface is activated, often using iodine as an initiator, to guarantee consistent Grignard reagent formation. The subsequent coupling with the dibromo-chloro alkane must be monitored via HPLC to prevent over-reaction or decomposition of the sensitive organometallic intermediate. Finally, the etherification step benefits from the use of recyclable solvents like xylene and methanol, aligning with green chemistry principles.
- Prepare the Grignard reagent by reacting chlorobenzene with magnesium chips in tetrahydrofuran at -20 to 0°C under nitrogen protection.
- Perform a coupling reaction by adding 1-bromo-4-chlorobutane to the Grignard reagent at low temperature to form 1-chloro-4-phenylbutane.
- Execute a condensation reaction between 1-chloro-4-chlorobutane and p-hydroxybenzoic acid using a phase transfer catalyst and base to yield the final acid.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the shift to this Grignard-based methodology offers compelling economic and logistical benefits that extend beyond simple yield improvements. The primary driver for cost reduction lies in the raw material selection; replacing expensive 4-phenyl-1-butanol with commodity-grade chlorobenzene and 1-bromo-4-chlorobutane significantly lowers the bill of materials. Chlorobenzene is a high-volume petrochemical derivative with stable pricing and abundant global availability, insulating the supply chain from the volatility often seen with specialized fine chemical intermediates. Furthermore, the elimination of aluminum trichloride removes the downstream costs associated with neutralizing and disposing of large volumes of aluminum-containing sludge, which is a regulated hazardous waste in many jurisdictions. This reduction in waste treatment complexity translates directly into lower operational expenditures and reduced liability for the manufacturing site.
- Cost Reduction in Manufacturing: The economic model of this process is strengthened by the avoidance of stoichiometric Lewis acid catalysts and expensive alcohol starting materials. By utilizing a catalytic amount of phase transfer agent and inexpensive inorganic bases like potassium carbonate, the variable cost per kilogram of product is substantially decreased. Additionally, the high selectivity of the Grignard coupling minimizes the formation of difficult-to-separate byproducts, which improves the overall mass balance and reduces the loss of valuable materials during purification. This efficiency gain means that less raw material is required to produce the same amount of finished goods, effectively stretching the purchasing budget and improving the gross margin profile of the intermediate.
- Enhanced Supply Chain Reliability: Sourcing reliability is markedly improved because the key inputs are bulk chemicals rather than niche custom synthetics. Chlorobenzene and magnesium are produced by multiple major chemical manufacturers worldwide, reducing the risk of single-source supply disruptions. The process also avoids the use of highly toxic reagents like phenylbutyl bromide, which often require special shipping classifications and storage facilities that can bottleneck logistics. By simplifying the hazard profile of the inventory, the manufacturing facility can operate with greater flexibility and resilience, ensuring consistent delivery schedules to downstream API producers even during periods of market tightness or regulatory scrutiny.
- Scalability and Environmental Compliance: From a scale-up perspective, the reaction conditions are robust and tolerant of minor variations, making the transition from pilot plant to commercial production smoother. The absence of corrosive hydrobromic acid gas generation eliminates the need for specialized Hastelloy reactors or extensive gas scrubbing infrastructure, allowing the process to be run in standard glass-lined steel equipment. This compatibility with existing infrastructure accelerates the timeline for technology transfer and capacity expansion. Moreover, the reduced wastewater load and absence of heavy metal contaminants simplify the environmental permitting process, ensuring long-term operational continuity in regions with strict ecological compliance standards.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis route. These answers are derived directly from the experimental data and comparative analysis provided in the patent documentation, offering clarity on performance metrics and operational requirements. Understanding these details is crucial for technical teams evaluating the feasibility of adopting this process for their own manufacturing lines or for procurement teams negotiating supply contracts based on these specifications.
Q: How does this Grignard route improve upon traditional Friedel-Crafts synthesis?
A: Traditional methods often utilize aluminum trichloride catalysts which generate significant wastewater and require complex treatment. This novel Grignard pathway avoids heavy metal catalysts and highly toxic intermediates like phenylbutyl bromide, resulting in a cleaner process with easier waste management.
Q: Does this process require expensive starting materials like phenylbutanol?
A: No, a key advantage of this patent is the avoidance of expensive phenylbutanol. Instead, it utilizes commodity chemicals such as chlorobenzene and 1-bromo-4-chlorobutane, which significantly lowers the raw material cost basis for large-scale production.
Q: What purity levels can be achieved with this synthesis method?
A: The patent data indicates that through optimized crystallization and washing steps, the process consistently achieves high purity levels exceeding 98%, which is critical for downstream API synthesis such as Pranlukast.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable p-Phenylbutoxy Benzoic Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the successful development of respiratory therapeutics like Pranlukast. Our technical team has extensively analyzed the Grignard-based synthesis route described in CN108558916B and possesses the expertise to execute this chemistry with precision and consistency. We bring extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive a steady supply of material regardless of market fluctuations. Our facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, including the detection of trace Grignard coupling byproducts and residual solvents, guaranteeing that every batch meets the exacting standards required for pharmaceutical grade intermediates.
We invite potential partners to engage with our technical procurement team to discuss how this optimized synthesis route can benefit your specific supply chain needs. By leveraging our manufacturing capabilities, you can secure a Customized Cost-Saving Analysis that quantifies the potential reductions in raw material and waste disposal costs for your organization. We encourage you to contact us to request specific COA data from our recent pilot runs and to schedule a consultation for detailed route feasibility assessments tailored to your project timelines and volume requirements.
