Scalable Metal-Free Synthesis of Fused Bisindole Derivatives for Advanced Optoelectronic Materials
Scalable Metal-Free Synthesis of Fused Bisindole Derivatives for Advanced Optoelectronic Materials
The rapid evolution of the organic electronics sector demands intermediates that combine high purity with sustainable manufacturing protocols. Patent CN109467559B introduces a groundbreaking methodology for the preparation of fused bisindole derivatives, a class of compounds critical for the development of next-generation LED polymers and functional materials. Unlike traditional pathways that rely on hazardous reagents and precious metal catalysts, this invention leverages a benign oxidative cyclization strategy using sodium nitrite and potassium persulfate. For R&D directors and procurement specialists seeking a reliable electronic chemical supplier, this technology represents a paradigm shift towards greener, more cost-effective production. The structural versatility of the starting indole compounds allows for the generation of a diverse library of derivatives, each tailored for specific optoelectronic applications. By addressing the longstanding issues of heavy metal contamination and operational complexity, this patent lays the foundation for a more robust supply chain in the high-value fine chemicals market.
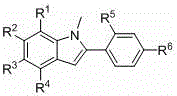
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of complex indole-based architectures has been plagued by significant technical and economic hurdles that hinder commercial viability. Conventional routes often necessitate the use of acetylene gas, a highly unstable and dangerous reagent that requires specialized handling infrastructure and poses severe safety risks in a manufacturing environment. Furthermore, many established protocols depend on expensive rhodium catalysts to drive the coupling reactions, which not only inflates the raw material costs but also introduces the risk of residual heavy metals in the final product. The presence of such metallic impurities is particularly detrimental in the field of optoelectronics, where trace contaminants can quench fluorescence or degrade device performance. Additionally, older methods frequently employ harsh oxidants like potassium permanganate or reducing agents like stannous chloride, generating substantial quantities of toxic waste that complicate disposal and increase environmental compliance costs. These factors collectively result in low overall yields and unpredictable batch-to-batch consistency, making scale-up a formidable challenge for supply chain managers.
The Novel Approach
The methodology disclosed in CN109467559B offers a transformative solution by replacing hazardous and expensive reagents with safe, commodity chemicals. This novel approach utilizes sodium nitrite as a nitrosating agent and potassium persulfate as an oxidant, both of which are inexpensive, stable, and readily available on a global scale. The reaction proceeds through a mild two-step sequence: an initial nitrosation followed by a reductive cyclization mediated by triethyl phosphite. This eliminates the need for transition metal catalysis entirely, thereby removing the costly and time-consuming step of heavy metal scavenging from the downstream processing. The operational simplicity is further enhanced by the use of common organic solvents such as acetone, acetonitrile, or dimethyl sulfoxide, which facilitates easy solvent recovery and recycling. For manufacturers aiming for cost reduction in electronic chemical manufacturing, this route provides a direct path to higher margins through reduced input costs and simplified waste management. The stability and controllability of the reaction conditions ensure that the process can be reliably transferred from the laboratory bench to multi-ton production scales without compromising product quality.
Mechanistic Insights into Sodium Nitrite-Mediated Oxidative Cyclization
The core of this innovation lies in the elegant mechanistic pathway that constructs the fused bisindole scaffold without external metal catalysis. The reaction initiates with the electrophilic nitrosation of the electron-rich indole ring, facilitated by the in situ generation of nitrosonium species from sodium nitrite and the persulfate oxidant. This step selectively functionalizes the indole nucleus, setting the stage for the subsequent ring-closing event. The use of potassium persulfate is critical here, as it serves as a clean one-electron oxidant that drives the formation of the reactive intermediate while minimizing side reactions. Following the nitrosation, the addition of triethyl phosphite triggers a deoxygenative cyclization. Triethyl phosphite acts as a potent oxygen acceptor, reducing the nitroso group and simultaneously promoting the formation of the new carbon-carbon bond that fuses the two indole units. This intramolecular coupling is highly efficient, driven by the thermodynamic stability of the resulting aromatic system and the formation of the strong phosphorus-oxygen bond in the phosphate byproduct.
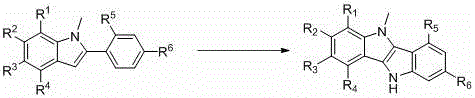
From an impurity control perspective, this metal-free mechanism offers distinct advantages for producing high-purity OLED material precursors. The absence of transition metals means there is no risk of catalyst-derived impurities that are notoriously difficult to remove to ppm levels. Furthermore, the reaction conditions are mild enough (60-130°C) to prevent thermal degradation of sensitive functional groups on the indole ring, such as halogens or nitro groups, which might be essential for downstream tuning of electronic properties. The byproducts of the reaction, primarily inorganic salts and phosphate esters, are generally polar and water-soluble, allowing for their efficient removal during the aqueous workup or column chromatography purification steps. This results in a cleaner crude product profile, reducing the burden on purification teams and increasing the overall recovery of the target molecule. For R&D teams focused on structure-activity relationships, this robustness allows for the exploration of a wider range of substituents without fear of incompatible reaction conditions.
How to Synthesize Fused Bisindole Derivatives Efficiently
The practical implementation of this synthesis route is designed for ease of execution, requiring standard laboratory equipment and straightforward operational procedures. The process begins with the dissolution of the selected indole substrate in a compatible solvent, followed by the sequential addition of sodium nitrite and potassium persulfate. The mixture is then heated to a temperature range of 60-120°C, depending on the specific reactivity of the substrate, and monitored via thin-layer chromatography until the starting material is fully consumed. Upon completion of the nitrosation step, the solvent is removed under reduced pressure, and the residue is treated with triethyl phosphite. The cyclization is effected by heating the mixture to 100-130°C, after which the crude product is isolated and purified using standard silica gel chromatography with a petroleum ether and ethyl acetate eluent system. The detailed standardized synthesis steps see the guide below.
- Conduct nitrosation of the indole substrate using sodium nitrite and potassium persulfate in a suitable solvent at 60-120°C.
- Remove the solvent via rotary evaporation after monitoring reaction completion by TLC.
- Add triethyl phosphite to the residue and heat to 100-130°C to effect cyclization, followed by purification via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented technology translates into tangible strategic benefits that extend beyond mere chemical yield. The primary advantage lies in the drastic simplification of the raw material portfolio; by eliminating the need for specialty gases like acetylene and precious metal catalysts like rhodium, companies can secure their supply lines against geopolitical volatility and price fluctuations associated with rare earth elements. The reliance on bulk commodities such as sodium nitrite and potassium persulfate ensures a stable and predictable cost structure, enabling more accurate long-term financial planning. Moreover, the simplified downstream processing reduces the consumption of auxiliary materials like scavenger resins and extensive filtration media, further driving down the cost of goods sold. This efficiency gain is critical for maintaining competitiveness in the fast-paced market of display and optoelectronic materials.
- Cost Reduction in Manufacturing: The elimination of expensive rhodium catalysts and hazardous acetylene gas removes two of the most significant cost drivers in traditional indole synthesis. Without the need for heavy metal removal steps, the consumption of specialized purification media is significantly reduced, leading to lower operational expenditures. The use of cheap, abundant reagents like sodium nitrite ensures that raw material costs remain minimal even at large production volumes. Additionally, the high overall yields reported in the patent examples mean less raw material is wasted per unit of product, maximizing the return on investment for every batch produced.
- Enhanced Supply Chain Reliability: Sourcing rhodium or high-purity acetylene can be subject to supply disruptions and strict regulatory controls, whereas sodium nitrite and potassium persulfate are produced globally in massive quantities. This abundance guarantees a continuous supply of key inputs, minimizing the risk of production stoppages due to material shortages. The robustness of the reaction conditions also means that the process is less sensitive to minor variations in reagent quality, providing a buffer against supply chain inconsistencies. For supply chain planners, this reliability translates into shorter lead times and the ability to commit to tighter delivery schedules with confidence.
- Scalability and Environmental Compliance: The mild reaction temperatures and absence of toxic heavy metals make this process inherently safer and easier to scale from pilot plants to commercial reactors. The waste stream is significantly cleaner, consisting mainly of inorganic salts and phosphate derivatives that are easier to treat than heavy metal sludge. This aligns perfectly with increasingly stringent environmental regulations, reducing the liability and cost associated with waste disposal. The simplicity of the workup, involving basic rotary evaporation and chromatography, allows for seamless technology transfer to manufacturing sites without the need for exotic equipment or specialized containment facilities.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this fused bisindole synthesis technology. These answers are derived directly from the experimental data and technical specifications outlined in the patent documentation, ensuring accuracy and relevance for potential partners. Understanding these details is crucial for evaluating the feasibility of integrating this route into existing production workflows. The insights provided here cover aspects ranging from reaction optimization to product application suitability.
Q: What are the key advantages of this metal-free synthesis over traditional rhodium-catalyzed methods?
A: This method eliminates the need for expensive rhodium catalysts and hazardous acetylene gas, significantly reducing raw material costs and safety risks while avoiding heavy metal contamination in the final product.
Q: What is the typical yield range for the fused bisindole derivatives produced via this route?
A: According to the patent data, the overall isolated yields for various substituted derivatives range consistently between 80% and 87%, demonstrating high efficiency and robustness across different substrates.
Q: Is this process suitable for large-scale commercial production of optoelectronic intermediates?
A: Yes, the process utilizes mild reaction conditions (60-130°C), common solvents like acetone or acetonitrile, and simple workup procedures, making it highly amenable to scale-up for industrial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Fused Bisindole Derivatives Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the metal-free synthesis route described in CN109467559B for the optoelectronics industry. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from lab-scale discovery to market-ready supply is seamless. Our state-of-the-art facilities are equipped to handle the specific solvent systems and thermal requirements of this process, while our rigorous QC labs enforce stringent purity specifications to meet the exacting standards of the LED and polymer sectors. We are committed to delivering high-purity intermediates that empower your R&D teams to push the boundaries of material performance without being constrained by supply limitations.
We invite you to collaborate with us to leverage this innovative chemistry for your next project. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating exactly how this metal-free route can optimize your budget. Please contact us to request specific COA data for our current inventory or to discuss route feasibility assessments for your proprietary derivatives. Together, we can build a more sustainable and efficient supply chain for the future of organic electronics.
