Advanced Metal-Free Synthesis Strategy for High-Purity Nitrogen-Containing Aryl Intermediates
The pharmaceutical and fine chemical industries are constantly seeking robust, cost-effective methodologies for constructing carbon-nitrogen bonds, which are fundamental motifs in bioactive molecules. Patent CN115043736A introduces a groundbreaking approach for preparing nitrogen-containing aryl compounds that circumvents the traditional reliance on expensive transition metal catalysts during the critical C-N bond formation step. This innovation leverages a metal-free nucleophilic substitution strategy using sodium hydride and o-diiodobenzene to generate protected ortho-iodoaniline intermediates with high efficiency. For R&D directors and procurement managers, this represents a significant shift towards more sustainable and economically viable synthetic routes. The technology not only simplifies the purification process by eliminating heavy metal contaminants but also utilizes readily available starting materials, ensuring a stable supply chain for complex pharmaceutical intermediates. By integrating this methodology into existing production workflows, manufacturers can achieve substantial operational improvements while maintaining stringent quality standards required for API synthesis.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the formation of C-N bonds in aryl systems has been dominated by palladium-catalyzed cross-coupling reactions, such as the Buchwald-Hartwig amination. While effective, these methods suffer from inherent drawbacks that impact both cost and environmental sustainability. The reliance on precious metal catalysts like palladium, often paired with sophisticated and costly phosphine ligands, drives up the raw material expenditure significantly. Furthermore, the presence of residual heavy metals in the final product poses a severe regulatory challenge, necessitating complex and expensive purification steps to meet strict ppm limits set by health authorities. Additionally, conventional methods often require harsh reaction conditions, including high temperatures and inert atmospheres, which increase energy consumption and operational complexity. The risk of over-coupling or side reactions in metal-catalyzed systems can also lead to lower yields and difficult-to-separate impurity profiles, complicating the scale-up process for commercial manufacturing.
The Novel Approach
In stark contrast, the methodology disclosed in CN115043736A offers a streamlined, metal-free alternative for the initial arylation step. By reacting nitrogen-containing carbonyl compounds (specifically Boc-protected amines) with o-diiodobenzene in the presence of sodium hydride, the process achieves efficient C-N coupling without any transition metal catalyst. This reaction proceeds under mild conditions, typically at room temperature or slightly elevated temperatures around 40°C, using tetrahydrofuran (THF) as a solvent. The absence of metal catalysts not only drastically reduces the cost of goods but also simplifies the workup procedure, as there is no need for specialized scavengers to remove palladium residues. The use of o-diiodobenzene, a commercially available and chemically stable reagent, ensures consistent quality and supply reliability. This novel approach effectively bypasses the economic and environmental bottlenecks associated with traditional metal catalysis, providing a cleaner and more direct route to valuable ortho-iodoaniline scaffolds.
Mechanistic Insights into Metal-Free Nucleophilic Aromatic Substitution
The core mechanistic advantage of this technology lies in the activation of the nitrogen nucleophile through deprotonation by a strong base, sodium hydride. In the first step, the nitrogen-containing carbonyl compound, such as a Boc-protected amine, is treated with NaH in THF. This generates a highly reactive nitrogen anion species which then attacks the electron-deficient aromatic ring of o-diiodobenzene. Unlike metal-catalyzed cycles that involve oxidative addition and reductive elimination steps prone to catalyst deactivation, this direct nucleophilic substitution is robust and less sensitive to trace impurities. The reaction stoichiometry is carefully optimized, with a molar ratio of o-diiodobenzene to amine to base typically around 2.5:1:2.5, ensuring complete conversion while minimizing waste. The resulting protected intermediate retains the iodine functionality at the ortho position, which is crucial for subsequent diversification. This mechanistic simplicity translates to high reproducibility and ease of control, key factors for maintaining batch-to-batch consistency in large-scale production environments.
Following the initial coupling, the protocol involves a strategic deprotection step to unlock the reactivity of the amine for further transformations. The Boc protecting group is efficiently removed using trifluoroacetic acid (TFA) in dichloromethane (DCM) at room temperature. This mild acidic condition cleaves the carbamate linkage without affecting the sensitive aryl-iodide bond, yielding the free ortho-iodoaniline derivative. The preservation of the iodine handle is vital, as it serves as a versatile pivot point for downstream functionalization. From a purity perspective, this two-step sequence (coupling followed by deprotection) allows for intermediate purification, effectively removing any unreacted starting materials or side products before the final diversification steps. This modular approach ensures that the final nitrogen-containing aryl compounds meet high-purity specifications, minimizing the burden on final crystallization or chromatography steps.
How to Synthesize Nitrogen-Containing Aryl Compounds Efficiently
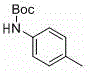
The synthesis of these valuable intermediates follows a logical and scalable sequence designed for maximum yield and minimal waste generation. The process begins with the protection of the amine substrate, followed by the key metal-free coupling reaction, and concludes with deprotection and optional diversification. Detailed standard operating procedures for each stage, including precise reagent quantities, temperature controls, and workup protocols, are essential for successful implementation. The following guide outlines the critical phases of this synthesis, ensuring that technical teams can replicate the high yields reported in the patent data, such as the 91% yield observed for tert-butyl (2-iodophenyl)(p-tolyl)carbamate.
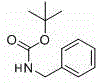
- Protect the amine starting material (e.g., benzylamine, p-toluidine) using di-tert-butyl dicarbonate ((Boc)2O) in ethanol under reflux to form the Boc-protected carbamate.
- React the Boc-protected amine with o-diiodobenzene in the presence of sodium hydride (NaH) in THF at room temperature to 40°C to achieve metal-free C-N bond formation.
- Deprotect the resulting intermediate using trifluoroacetic acid (TFA) in DCM to yield the free ortho-iodoaniline, which serves as a versatile scaffold for further functionalization.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this metal-free synthesis route offers tangible benefits that directly impact the bottom line and operational resilience. The elimination of palladium catalysts in the primary C-N bond forming step removes a major cost driver and supply chain vulnerability associated with precious metals. Since the price of palladium is subject to significant market volatility, replacing it with inexpensive bases like sodium hydride stabilizes production costs and improves margin predictability. Furthermore, the reagents used, such as o-diiodobenzene and Boc-anhydride, are commodity chemicals with established global supply chains, reducing the risk of raw material shortages. The mild reaction conditions also contribute to energy efficiency, lowering utility costs associated with heating and cooling large reactors. Overall, this technology aligns perfectly with green chemistry principles, potentially reducing waste disposal costs and enhancing the company's sustainability profile.
- Cost Reduction in Manufacturing: The most significant economic advantage stems from the complete avoidance of transition metal catalysts in the initial coupling phase. Traditional methods require expensive palladium sources and specialized ligands, which can account for a substantial portion of the raw material cost. By substituting these with sodium hydride, the direct material cost is drastically reduced. Additionally, the simplified purification process eliminates the need for expensive metal scavenging resins or complex extraction protocols designed to lower metal residues below regulatory limits. This streamlining of the downstream processing further reduces labor and consumable costs, leading to a more competitive cost structure for the final active pharmaceutical ingredients or intermediates.
- Enhanced Supply Chain Reliability: Relying on precious metal catalysts often introduces supply chain fragility due to geopolitical factors and mining constraints. This new methodology utilizes widely available organic and inorganic reagents that are produced by multiple suppliers globally. The stability of o-diiodobenzene allows for long-term storage and bulk purchasing, enabling better inventory management and hedging against price fluctuations. Moreover, the robustness of the reaction conditions means that the process is less susceptible to variations in raw material quality, ensuring consistent output even when sourcing from different vendors. This reliability is crucial for maintaining continuous production schedules and meeting tight delivery deadlines for downstream customers.
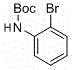
- Scalability and Environmental Compliance: Scaling chemical processes often amplifies safety and environmental challenges, particularly those involving pyrophoric catalysts or high-pressure hydrogenation. The described metal-free coupling operates at atmospheric pressure and moderate temperatures, significantly reducing the engineering controls required for safe operation. The absence of heavy metals simplifies wastewater treatment and solid waste disposal, ensuring easier compliance with increasingly stringent environmental regulations. The high yields reported, such as 98% for certain bromo-iodo derivatives, indicate excellent atom economy and minimal byproduct formation. This efficiency translates to higher throughput per batch and reduced solvent consumption, making the process highly attractive for multi-ton commercial manufacturing campaigns.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel synthesis technology. These answers are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity on the practical application of the method. Understanding these details helps stakeholders evaluate the feasibility of integrating this route into their existing manufacturing portfolios for producing high-value nitrogen-containing scaffolds.
Q: What are the advantages of this metal-free C-N coupling method over traditional palladium catalysis?
A: The primary advantage is the elimination of expensive palladium catalysts and phosphine ligands in the initial coupling step, significantly reducing raw material costs and simplifying the removal of heavy metal residues, which is critical for pharmaceutical compliance.
Q: Can the ortho-iodoaniline intermediates produced be used for diverse downstream reactions?
A: Yes, the patent demonstrates that the deprotected ortho-iodoaniline intermediates are highly versatile scaffolds that can undergo Suzuki coupling, Sonogashira coupling, and intramolecular cyclization to form complex heterocycles like carbazoles and benzothiazoles.
Q: Is this process suitable for large-scale commercial manufacturing?
A: The process utilizes mild reaction conditions (room temperature to 40°C) and commercially available, stable reagents like o-diiodobenzene and sodium hydride, making it highly scalable and safer for industrial production compared to high-temperature or high-pressure alternatives.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Nitrogen-Containing Aryl Compound Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of metal-free synthetic methodologies in modern pharmaceutical manufacturing. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative lab-scale discoveries like CN115043736A are successfully translated into robust industrial processes. Our state-of-the-art facilities are equipped to handle the specific reagents and conditions required for this chemistry, including the safe handling of sodium hydride and the efficient management of halogenated solvents. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch of nitrogen-containing aryl intermediates meets the highest quality standards, free from unacceptable levels of metallic impurities.
We invite pharmaceutical companies and research institutions to collaborate with us to leverage this cost-effective and environmentally friendly technology for your next project. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific target molecules, demonstrating how this metal-free route can optimize your budget without compromising quality. Please contact our technical procurement team to request specific COA data for similar intermediates and comprehensive route feasibility assessments. Together, we can accelerate the development of your drug candidates while achieving significant operational efficiencies and sustainability goals.
