Scalable Copper-Catalyzed Synthesis of 4,4-Dimethyl-4,5-Dihydropyridazin-3-One Intermediates
Introduction to Advanced Pyridazinone Scaffold Construction
The pharmaceutical industry continuously seeks robust and scalable methodologies for constructing nitrogen-containing heterocycles, which serve as critical scaffolds in a vast array of bioactive molecules. A recent technological breakthrough documented in patent CN113845481A introduces a novel synthetic pathway for generating 4,4-dimethyl-4,5-dihydropyridazin-3-one derivatives directly from alkynes. This innovation addresses long-standing challenges in heterocyclic chemistry by replacing hazardous traditional reagents with a modular, copper-catalyzed system. For R&D directors and procurement specialists, this represents a significant opportunity to streamline the supply chain for histamine H3A receptor inverse agonists and other therapeutic agents. By leveraging di-tert-butyl azodicarboxylate and dimethyl azodiisobutyrate in the presence of cuprous bromide, this method offers a greener, more efficient alternative that aligns with modern green chemistry principles while maintaining high structural diversity.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 4,4-dimethyl-4,5-dihydropyridazin-3-one cores has relied heavily on the condensation of hydrazine hydrate with specific carbonyl precursors, a process fraught with significant safety and environmental liabilities. Traditional routes often necessitate the preparation of 2,2-dimethyl-4-carbonyl-1-butyrate structures through the addition of water-sensitive aryl Grignard reagents to succinic anhydrides, requiring stringent anhydrous conditions and cryogenic temperatures that drive up operational costs. Furthermore, the reliance on hydrazine hydrate introduces severe toxicity concerns, mandating expensive containment protocols and complex waste treatment procedures to ensure worker safety and regulatory compliance. These legacy methods frequently suffer from limited substrate scope and poor atom economy, making them ill-suited for the rapid iteration required in modern drug discovery pipelines. Consequently, the industry has faced a persistent bottleneck in accessing diverse pyridazinone libraries efficiently and safely.
The Novel Approach
In stark contrast, the methodology disclosed in CN113845481A revolutionizes this landscape by utilizing a direct three-component coupling strategy that bypasses the need for pre-functionalized carbonyl intermediates. This innovative route employs readily available terminal alkynes, di-tert-butyl azodicarboxylate, and dimethyl azodiisobutyrate as building blocks, facilitated by a catalytic amount of cuprous bromide in acetonitrile. The reaction proceeds under mild thermal conditions, typically around 80°C, to generate a vinyl hydrazine intermediate which is subsequently cyclized under acidic conditions. 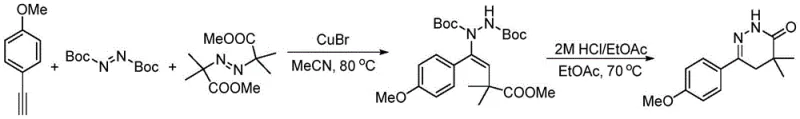 This transformation not only simplifies the synthetic sequence by reducing step count but also dramatically improves the safety profile by eliminating toxic hydrazine and moisture-sensitive organometallics. The versatility of this approach is evidenced by its compatibility with a wide range of substituted alkynes, enabling the rapid generation of structurally diverse analogues for SAR studies.
This transformation not only simplifies the synthetic sequence by reducing step count but also dramatically improves the safety profile by eliminating toxic hydrazine and moisture-sensitive organometallics. The versatility of this approach is evidenced by its compatibility with a wide range of substituted alkynes, enabling the rapid generation of structurally diverse analogues for SAR studies.
Mechanistic Insights into CuBr-Catalyzed Radical Cyclization
The core of this synthetic advancement lies in the copper-catalyzed radical addition mechanism that orchestrates the assembly of the pyridazinone ring system. The reaction initiates with the activation of the azo compounds by the cuprous bromide catalyst, generating reactive radical species that undergo regioselective addition across the triple bond of the alkyne substrate. This radical cascade effectively constructs the carbon-nitrogen framework necessary for the heterocycle, forming a stable vinyl hydrazine intermediate bearing the requisite Boc protecting groups. The use of CuBr at 20 mol% loading ensures efficient turnover while minimizing metal contamination in the final product, a critical factor for pharmaceutical applications. Following the initial coupling, the intermediate undergoes an acid-mediated deprotection and cyclization sequence upon treatment with hydrochloric acid in ethyl acetate at 70°C. 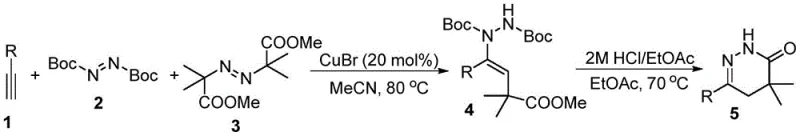 This second step cleaves the tert-butyl carbamate groups and promotes intramolecular nucleophilic attack to close the six-membered ring, yielding the target 4,4-dimethyl-4,5-dihydropyridazin-3-one scaffold with high fidelity.
This second step cleaves the tert-butyl carbamate groups and promotes intramolecular nucleophilic attack to close the six-membered ring, yielding the target 4,4-dimethyl-4,5-dihydropyridazin-3-one scaffold with high fidelity.
From an impurity control perspective, this mechanism offers distinct advantages over traditional condensation reactions which often produce complex mixtures of hydrazine byproducts. The radical nature of the initial coupling is highly selective for the alkyne functionality, minimizing side reactions with other potential functional groups present on the aromatic ring, such as methoxy, chloro, or cyano substituents. The subsequent acid treatment is clean and efficient, primarily generating volatile isobutylene and carbon dioxide as byproducts during Boc deprotection, which simplifies downstream purification. This mechanistic clarity allows process chemists to predict and manage impurity profiles more effectively, ensuring that the final API intermediate meets stringent purity specifications required for clinical development. The robustness of the catalytic cycle against various electronic environments on the alkyne further underscores its utility for synthesizing complex drug candidates.
How to Synthesize 4,4-Dimethyl-4,5-Dihydropyridazin-3-One Efficiently
Implementing this synthesis requires careful attention to reaction parameters to maximize yield and reproducibility across different substrate classes. The process begins by dissolving the catalyst and azo reagents in anhydrous acetonitrile under an inert atmosphere, followed by the sequential addition of the alkyne and the diester component. Maintaining the reaction temperature at 80°C for 6 to 9 hours is crucial for complete conversion to the vinyl hydrazine intermediate, after which simple filtration and concentration afford the crude material. The subsequent cyclization step involves suspending the crude intermediate in ethyl acetate and treating it with a 2M hydrochloric acid solution, heating the mixture to 70°C for 1 to 8 hours depending on the specific substrate reactivity. Detailed standardized operating procedures for this transformation are provided below to assist technical teams in replicating these results.
- Dissolve cuprous bromide and di-tert-butyl azodicarboxylate in acetonitrile, add alkyne and dimethyl azodiisobutyrate, then heat to 80°C for 6-9 hours to form the vinyl hydrazine intermediate.
- Treat the crude intermediate with a solvent and 2M hydrochloric acid/ethyl acetate solution, heating to 70°C for 1-8 hours to effect cyclization and deprotection.
- Perform standard post-reaction workup including neutralization, extraction, and purification via silica gel column chromatography to isolate the final pyridazinone product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this copper-catalyzed methodology translates into tangible strategic benefits regarding cost stability and sourcing reliability. By shifting away from water-sensitive Grignard reagents and toxic hydrazine hydrate, manufacturers can significantly reduce the overhead costs associated with specialized storage, handling, and hazardous waste disposal. The raw materials required for this process, including various substituted phenylacetylenes and azo compounds, are commodity chemicals available from multiple global suppliers, thereby mitigating the risk of single-source bottlenecks. Furthermore, the mild reaction conditions eliminate the need for energy-intensive cryogenic cooling or high-pressure equipment, leading to substantial reductions in utility consumption and capital expenditure for reactor infrastructure. These factors collectively enhance the overall economic viability of producing pyridazinone intermediates at scale.
- Cost Reduction in Manufacturing: The elimination of expensive and hazardous reagents like hydrazine hydrate and aryl Grignard reagents directly lowers the bill of materials for every batch produced. Additionally, the simplified workup procedure, which avoids complex quenching steps for organometallics, reduces labor hours and solvent usage, contributing to a leaner manufacturing cost structure. The use of a base metal catalyst like copper bromide instead of precious metals further ensures that catalyst costs remain negligible even at large production volumes.
- Enhanced Supply Chain Reliability: Sourcing stability is greatly improved as the key starting materials are bulk chemicals with established supply chains, unlike specialized organometallic reagents that may have long lead times. The robustness of the reaction conditions means that production is less susceptible to disruptions caused by minor variations in environmental humidity or temperature, ensuring consistent output quality. This reliability allows supply chain planners to forecast inventory needs with greater confidence and maintain optimal stock levels of critical pharmaceutical intermediates.
- Scalability and Environmental Compliance: The process is inherently scalable due to its operation at atmospheric pressure and moderate temperatures, facilitating seamless technology transfer from laboratory to pilot and commercial plants. From an environmental standpoint, the avoidance of toxic hydrazine and the generation of benign gaseous byproducts during deprotection align with increasingly strict global environmental regulations. This compliance reduces the regulatory burden on manufacturing sites and minimizes the risk of production shutdowns due to environmental non-compliance issues.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel synthetic route. These insights are derived directly from the experimental data and scope defined in the patent literature, providing a factual basis for decision-making. Understanding these nuances is essential for evaluating the feasibility of integrating this technology into existing production workflows.
Q: What are the safety advantages of this new pyridazinone synthesis method?
A: This method eliminates the use of toxic hydrazine hydrate and water-sensitive organometallic reagents like Grignard reagents, significantly improving operational safety and reducing hazardous waste disposal costs.
Q: What types of alkyne substrates are compatible with this copper-catalyzed route?
A: The process demonstrates broad substrate applicability, successfully accommodating phenylacetylenes with various substituents (methoxy, chloro, fluoro, ethyl), heterocyclic alkynes like ethynylpyridine and ethynylthiophene, and functionalized alkynes such as 4-cyanomethylphenylacetylene.
Q: Is this synthesis method suitable for large-scale commercial production?
A: Yes, the reaction utilizes mild temperatures (80°C and 70°C), inexpensive catalysts (CuBr), and readily available raw materials, making it highly amenable to commercial scale-up without requiring specialized low-temperature or anhydrous infrastructure.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4,4-Dimethyl-4,5-Dihydropyridazin-3-One Supplier
As a leader in the fine chemical sector, NINGBO INNO PHARMCHEM possesses the technical expertise and infrastructure to bring this advanced synthetic methodology to commercial reality. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with unwavering consistency. We operate stringent purity specifications and utilize rigorous QC labs to guarantee that every batch of 4,4-dimethyl-4,5-dihydropyridazin-3-one intermediate meets the highest industry standards. Our commitment to quality assurance means that you can rely on us for critical project milestones, from early-stage clinical trials to full-scale market launch.
We invite you to collaborate with us to leverage this cutting-edge technology for your next drug development program. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are prepared to provide specific COA data and comprehensive route feasibility assessments to demonstrate how this copper-catalyzed process can optimize your supply chain and accelerate your time to market.
