Advanced Rhodium-Catalyzed Synthesis of Planar Chiral Ferrocene Compounds for Commercial Pharmaceutical Applications
The pharmaceutical and fine chemical industries are constantly seeking robust methodologies for constructing complex chiral scaffolds, particularly those exhibiting planar chirality which is increasingly recognized for its unique pharmacological properties. Patent CN109678911B introduces a groundbreaking preparation method for ferrocene compounds that addresses long-standing challenges in asymmetric synthesis. This technology leverages a rhodium-catalyzed asymmetric arylation strategy to directly install planar chirality onto the ferrocene backbone without the need for cumbersome chiral auxiliary groups. By utilizing a specialized chiral phosphine ligand system in conjunction with a rhodium catalyst and a strong base, the process achieves exceptional enantioselectivity and reaction efficiency. For R&D directors and procurement specialists, this represents a significant leap forward in accessing high-purity ferrocene intermediates essential for next-generation medicinal chemistry programs and advanced material applications.
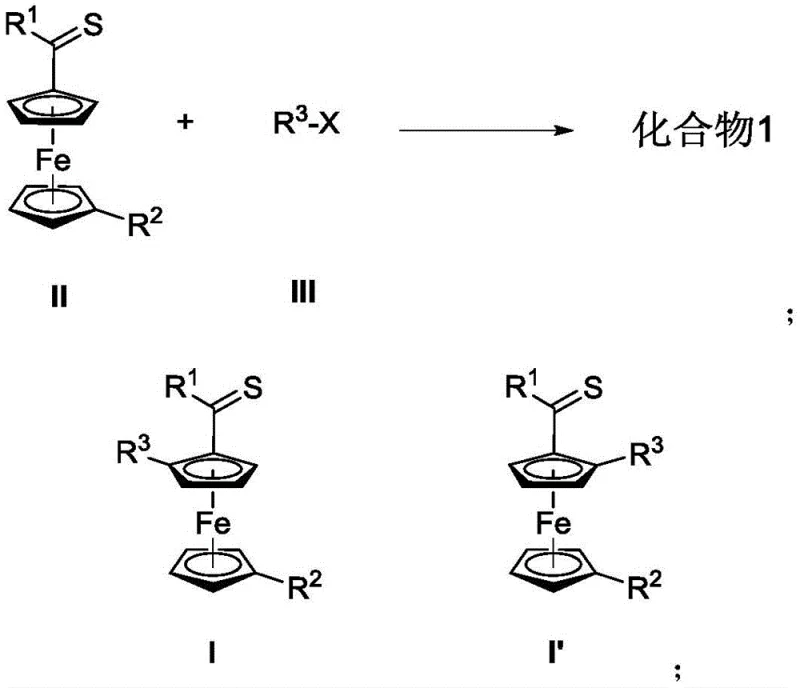
The development of planar chiral ferrocene derivatives has historically been hindered by synthetic limitations that increased both cost and complexity. Conventional strategies often relied on the use of chiral prosthetic groups to induce diastereoselective ortho-metallation, a approach that necessitates the introduction of central chirality into the ferrocene scaffold in advance. This multi-step requirement not only extends the synthetic timeline but also generates significant chemical waste and reduces overall atom economy. Furthermore, existing palladium-catalyzed coupling methods frequently suffer from poor substrate universality or inability to strictly control enantioselectivity across diverse electronic environments. The novel approach disclosed in the patent circumvents these issues by employing a direct C-H activation mechanism. This allows for the coupling of simple ferrocenyl thioamides with a wide array of aryl halides, including those with electron-withdrawing or electron-donating substituents, under relatively mild thermal conditions.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional pathways for synthesizing planar chiral ferrocenes are often plagued by inefficiencies that make them unsuitable for modern, cost-sensitive manufacturing environments. The reliance on stoichiometric chiral inducers means that every batch requires the synthesis and subsequent removal of these auxiliary groups, adding at least two extra synthetic steps to the process flow. Additionally, many prior art methods utilize palladium catalysts which, while effective for certain substrates, often struggle with steric hindrance when dealing with ortho-substituted aryl halides. The lack of broad substrate scope forces process chemists to develop custom conditions for each new target molecule, leading to unpredictable timelines and increased R&D expenditure. Moreover, the enantiomeric excess (ee) achieved in older methods often fluctuates significantly, requiring costly recrystallization or chiral chromatography steps to meet the stringent purity specifications required for active pharmaceutical ingredients.
The Novel Approach
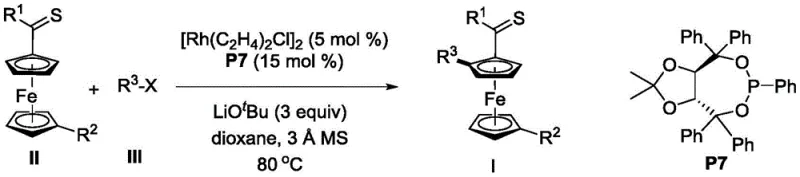
In stark contrast, the methodology described in CN109678911B utilizes a highly efficient rhodium catalytic system that operates with excellent turnover numbers and selectivity. The core innovation lies in the use of tartaric acid-derived chiral phosphite ligands, which create a precise chiral environment around the rhodium center. This setup facilitates the selective activation of the C-H bond on the ferrocene ring adjacent to the thioamide directing group. The reaction proceeds smoothly in common ether solvents like 1,4-dioxane or tetrahydrofuran at temperatures around 80°C. This thermal profile is energy-efficient and compatible with standard glass-lined reactors used in commercial plants. The method demonstrates remarkable tolerance for various functional groups on the aryl halide partner, including halogens, cyano groups, and trifluoromethyl moieties, thereby expanding the chemical space accessible to medicinal chemists without compromising yield or optical purity.
Mechanistic Insights into Rhodium-Catalyzed Asymmetric C-H Activation
The success of this transformation is deeply rooted in the synergistic interaction between the rhodium precursor and the bulky chiral phosphite ligand. Mechanistically, the reaction likely initiates with the formation of an active rhodium(I) species coordinated by the chiral ligand. This complex then undergoes oxidative addition with the aryl halide substrate, followed by the coordination of the ferrocenyl thioamide through the sulfur atom of the thioamide group. The thioamide acts as a powerful directing group, guiding the rhodium center to the proximal C-H bond on the cyclopentadienyl ring. Subsequent C-H activation generates a rhodacycle intermediate, which is the key stereochemistry-determining step. The steric bulk of the ligand, particularly the substituents on the tartrate backbone, effectively blocks one face of the ferrocene ring, ensuring that the aryl group is installed with high facial selectivity to yield the desired planar chiral configuration.
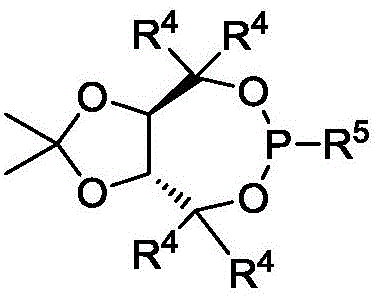
Impurity control in this system is managed through the careful selection of reaction additives and conditions. The inclusion of molecular sieves plays a pivotal role in maintaining the integrity of the catalytic cycle by sequestering trace water that could otherwise hydrolyze the sensitive organometallic intermediates or deactivate the lithium tert-butoxide base. Furthermore, the use of specific ligand architectures, such as the P7 ligand shown in the patent examples, minimizes the formation of regioisomers or homocoupling byproducts. The high enantioselectivity observed, often exceeding 90% ee and reaching up to 99% ee in optimized cases, suggests that the transition state for the reductive elimination step is tightly controlled. This level of precision reduces the burden on downstream purification processes, allowing for the isolation of high-purity products via standard silica gel chromatography without the need for extensive chiral resolution techniques.
How to Synthesize Planar Chiral Ferrocene Efficiently
The synthesis protocol outlined in the patent provides a clear roadmap for reproducing these high-value intermediates in a laboratory or pilot plant setting. The procedure emphasizes the importance of anhydrous conditions and inert atmosphere handling to maximize catalyst longevity. By adhering to the specified molar ratios of catalyst to substrate and utilizing the recommended base equivalents, operators can consistently achieve yields ranging from 50% to over 80% depending on the specific substrate electronics. The detailed experimental section offers variations for different aryl halides, demonstrating the robustness of the method across a diverse library of targets. For comprehensive operational details regarding reagent addition sequences and workup procedures, please refer to the standardized synthesis guide below.
- Prepare the reaction mixture by combining the ferrocenyl thioamide substrate, aryl halide, rhodium catalyst, chiral phosphite ligand, and lithium tert-butoxide base in anhydrous dioxane under inert atmosphere.
- Add activated molecular sieves to the reaction vessel to scavenge moisture and maintain anhydrous conditions critical for catalyst stability.
- Heat the reaction mixture to 80°C for the specified duration, then proceed with workup involving dilution, filtration, and column chromatography purification.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this rhodium-catalyzed technology offers substantial benefits that align with the strategic goals of cost reduction and supply chain resilience. The shift away from multi-step auxiliary-based syntheses to a direct catalytic coupling significantly compresses the manufacturing timeline. This compression translates directly into lower labor costs and reduced occupancy time in production facilities, which are critical factors in determining the final cost of goods sold. Furthermore, the catalyst loading is kept relatively low, typically around 5 mol%, which helps mitigate the expense associated with precious metal usage. Since the rhodium catalyst can be sourced from established suppliers and the ligands are derived from abundant natural tartaric acid, the raw material supply chain is stable and less prone to geopolitical disruptions compared to more exotic catalytic systems.
- Cost Reduction in Manufacturing: The elimination of chiral auxiliary synthesis and removal steps drastically simplifies the process flow, leading to significant savings in raw material consumption and waste disposal costs. By avoiding the use of stoichiometric amounts of chiral inducers, the overall mass intensity of the process is improved, which is a key metric for green chemistry and cost efficiency. Additionally, the ability to use standard solvents like dioxane and toluene avoids the need for specialized or hazardous solvent handling infrastructure, further reducing capital and operational expenditures associated with plant modifications.
- Enhanced Supply Chain Reliability: The reagents required for this transformation, including the rhodium dimer and lithium tert-butoxide, are commodity chemicals available from multiple global vendors. This multi-sourcing capability ensures that production schedules are not held hostage by single-supplier bottlenecks. The robustness of the reaction conditions also means that the process is less sensitive to minor variations in reagent quality, providing a buffer against supply chain fluctuations. Consequently, manufacturers can maintain consistent inventory levels and meet delivery commitments to downstream pharmaceutical clients with greater confidence.
- Scalability and Environmental Compliance: The reaction operates at moderate temperatures and does not require high-pressure equipment, making it inherently safer and easier to scale from kilogram to tonne quantities. The use of molecular sieves as a recyclable additive minimizes liquid waste generation compared to traditional drying agents. Moreover, the high selectivity of the reaction reduces the formation of difficult-to-separate byproducts, simplifying the purification workflow and reducing the volume of organic solvents required for chromatography. This aligns well with increasingly stringent environmental regulations regarding solvent emissions and chemical waste management in the fine chemical sector.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and optimization of this ferrocene synthesis technology. These answers are derived directly from the experimental data and embodiments provided in the patent documentation to ensure accuracy and relevance for process development teams. Understanding these nuances is essential for successfully transferring this chemistry from the bench to the pilot plant.
Q: What represents the primary advantage of this Rh-catalyzed method over traditional palladium systems?
A: This method utilizes a readily available rhodium catalyst system combined with tartaric acid-derived phosphite ligands to achieve direct C-H activation, eliminating the need for pre-installed chiral auxiliaries and significantly simplifying the synthetic route while maintaining high enantioselectivity.
Q: How does the choice of molecular sieves impact the reaction efficiency?
A: The addition of molecular sieves, particularly 3Å or 4Å types, is critical for scavenging trace moisture that could deactivate the sensitive rhodium catalyst or interfere with the organolithium base, thereby ensuring consistent high yields and reproducibility in large-scale batches.
Q: Is this synthesis protocol scalable for industrial production of API intermediates?
A: Yes, the protocol employs commercially available reagents, mild reaction temperatures around 80°C, and standard organic solvents like dioxane, making it highly amenable to scale-up from gram-scale laboratory synthesis to multi-kilogram commercial manufacturing without requiring exotic equipment.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Ferrocene Compound Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality chiral intermediates play in the development of innovative therapeutics and advanced materials. Our team of expert process chemists has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from discovery to market. We are committed to delivering products that meet stringent purity specifications through our rigorous QC labs, which are equipped with state-of-the-art analytical instrumentation for chiral separation and structural verification. Our facility is designed to handle sensitive organometallic reactions safely and efficiently, providing a secure environment for the manufacture of complex ferrocene derivatives.
We invite you to collaborate with us to leverage this advanced rhodium-catalyzed technology for your specific application needs. Whether you require custom synthesis of novel analogs or reliable supply of established intermediates, our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your project volume. Please contact us today to request specific COA data and route feasibility assessments, and let us demonstrate how our manufacturing capabilities can accelerate your development timeline while optimizing your budget.
