Advanced Stereoselective Synthesis of Biotin Intermediates for Commercial Scale Production
The pharmaceutical and fine chemical industries are constantly seeking robust, scalable pathways for the production of essential vitamin intermediates, particularly for Biotin (Vitamin B7). Patent CN110804062B, published in March 2021, introduces a transformative synthetic methodology for producing (3aS,6aR)-1,3-dibenzyltetrahydrofuroimidazole-2,4-dione, a pivotal chiral lactone intermediate in the total synthesis of biotin. This innovation addresses long-standing challenges in stereoselectivity and cost-efficiency by utilizing a novel chiral alcohol auxiliary that enables high-yield desymmetrization of meso-cyclic anhydrides. For R&D directors and procurement strategists, this technology represents a significant leap forward, offering a route that combines mild reaction conditions with exceptional stereocontrol, thereby ensuring a reliable supply of high-purity pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
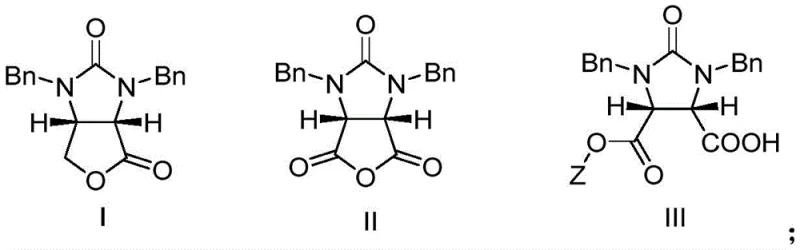
The Limitations of Conventional Methods
Historically, the synthesis of chiral biotin intermediates has been plagued by inefficiencies inherent in early resolution and asymmetric synthesis techniques. Traditional methods, such as those disclosed in US Patent 3876656, relied on complex chiral auxiliaries derived from chloramphenicol by-products, which resulted in convoluted multi-step sequences with poor atom economy. Furthermore, alternative approaches utilizing lithium aluminum hydride reductions, as seen in Japanese Patent JP05320168, often suffered from safety hazards associated with handling strong reducing agents on a large scale, alongside moderate yields that hovered around 74%. These legacy processes frequently required expensive resolving agents that could not be effectively recovered, leading to substantial waste generation and inflated production costs that hindered industrial scalability.
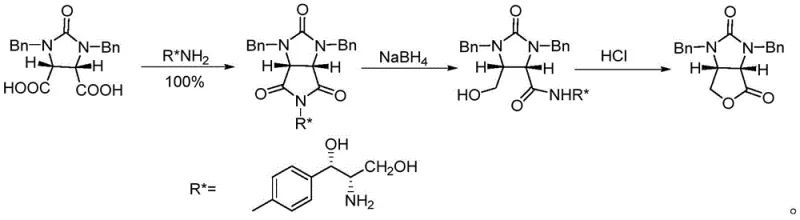
The Novel Approach
In stark contrast, the methodology outlined in CN110804062B streamlines the synthesis into a concise three-step sequence: selective alcoholysis, reduction, and cyclization. By employing a specifically designed chiral alcohol, (1R,2S)-1-phenyl-2-(1-pyrrolidinyl)-1-propanol, the process achieves direct kinetic resolution of the meso-anhydride starting material. This novel route eliminates the need for hazardous reagents like lithium aluminum hydride, substituting them with safer, more manageable reducing agents such as sodium borohydride. The result is a process that not only simplifies operational complexity but also dramatically improves the overall yield and stereoselectivity, making it an ideal candidate for cost reduction in biotin manufacturing and ensuring a stable supply chain for downstream API producers.
Mechanistic Insights into Chiral Alcohol-Mediated Desymmetrization
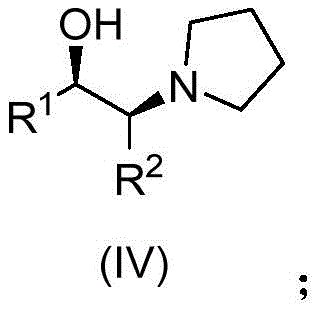
The core innovation of this technology lies in the unique structural properties of the chiral alcohol auxiliary used in the initial alcoholysis step. Unlike simple chiral alcohols that rely solely on steric bulk for induction, the auxiliary employed here, depicted as Formula (IV), incorporates a pyrrolidine ring adjacent to the chiral center. This structural feature serves a dual purpose: it provides the necessary steric environment to differentiate between the enantiotopic carbonyl groups of the meso-anhydride, and the tertiary amine nitrogen within the pyrrolidine ring acts as an intramolecular base. This intrinsic basicity catalyzes the acylation reaction without the need for external alkaline additives, which often complicate workup procedures and introduce impurities.
Following the formation of the chiral monoester intermediate (Formula III), the subsequent reduction and cyclization steps are meticulously controlled to preserve the established stereochemistry. The reduction of the ester moiety using sodium borohydride proceeds under mild conditions to generate a hydroxy-acid precursor, which spontaneously or acid-catalytically cyclizes to form the fused furan-imidazole ring system. The mechanism ensures that the chiral information installed in the first step is faithfully transferred to the final lactone product, resulting in high enantiomeric excess (ee) values. This precise control over the reaction pathway minimizes the formation of diastereomeric impurities, a critical factor for meeting the stringent purity specifications required for pharmaceutical grade intermediates.
How to Synthesize (3aS,6aR)-1,3-dibenzyltetrahydrofuroimidazole-2,4-dione Efficiently
The synthesis protocol described in the patent offers a straightforward operational framework suitable for pilot and commercial scale operations. The process begins with the activation of the cyclic anhydride in an organic solvent such as toluene or dichloromethane, followed by the addition of the chiral alcohol and a carbodiimide dehydrating agent. Detailed standard operating procedures regarding stoichiometry, temperature control during the exothermic alcoholysis, and the specific workup methods for isolating the intermediate are critical for success. For a comprehensive, step-by-step technical guide including exact molar ratios and quenching protocols, please refer to the standardized synthesis instructions provided below.
- Perform selective alcoholysis of cyclic anhydride (II) with chiral alcohol (IV) using DCC or EDCI as a dehydrating agent to form monoester (III).
- Reduce the ester group of compound (III) using sodium borohydride (NaBH4) in tetrahydrofuran to generate the hydroxy intermediate.
- Execute acid-catalyzed cyclization by refluxing the reduced intermediate in hydrochloric acid to obtain the final chiral lactone (I) and recover the chiral alcohol.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthetic route offers tangible benefits that extend beyond mere chemical elegance. The primary advantage lies in the economic efficiency driven by the recoverability of the chiral auxiliary. In traditional resolution processes, chiral reagents are often consumed or lost, representing a significant sunk cost. However, this patented method allows for the chiral alcohol to be recovered from the aqueous mother liquor after the final cyclization step via simple extraction and neutralization. This closed-loop capability significantly reduces the raw material cost per kilogram of the final product, enhancing the overall margin profile for manufacturers.
- Cost Reduction in Manufacturing: The elimination of expensive and hazardous reagents like lithium aluminum hydride in favor of sodium borohydride drastically lowers reagent costs and safety compliance expenditures. Furthermore, the high diastereoselectivity of the initial alcoholysis step minimizes the loss of starting material to unwanted isomers, thereby improving the effective yield of the process. The ability to recycle the chiral alcohol auxiliary means that the effective cost of this key reagent is amortized over multiple batches, leading to substantial long-term savings in the cost of goods sold (COGS) without compromising on quality.
- Enhanced Supply Chain Reliability: The starting materials for this synthesis, including the cyclic anhydride and the specific chiral alcohol, are derived from readily available commodity chemicals, reducing dependency on scarce or geopolitically sensitive precursors. The robustness of the reaction conditions, which tolerate a range of temperatures and solvents, ensures consistent batch-to-batch performance even when scaling up from laboratory to multi-ton production. This reliability mitigates the risk of supply disruptions, ensuring that downstream biotin manufacturers can maintain continuous production schedules to meet global market demand.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, this route is superior to prior art methods. The avoidance of heavy metal catalysts and the use of milder reducing agents simplify waste treatment protocols and reduce the burden on effluent processing facilities. The high selectivity of the reaction reduces the generation of complex organic waste streams, aligning with modern green chemistry principles. This makes the process highly scalable, allowing for seamless transition from kilogram-scale development to hundred-ton annual commercial production while maintaining strict adherence to environmental regulations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and claims presented in the patent documentation, providing clarity on the feasibility and performance of the method. Understanding these details is crucial for technical teams evaluating the integration of this process into existing manufacturing lines.
Q: What is the key advantage of the chiral alcohol used in this synthesis?
A: The specific chiral alcohol, (1R,2S)-1-phenyl-2-(1-pyrrolidinyl)-1-propanol, features a cyclic tertiary amine structure that acts as both a stereocontrol element and an intrinsic basic catalyst, eliminating the need for external alkaline additives and facilitating high diastereoselectivity.
Q: Is the chiral auxiliary recoverable in this process?
A: Yes, a significant economic advantage of this method is the recoverability of the chiral alcohol. After the final cyclization step, the aqueous phase can be neutralized and extracted to recover the chiral alcohol in high yield (approximately 90% in optimized examples) for reuse.
Q: What represents the typical purity profile of the final lactone?
A: The process described in patent CN110804062B consistently achieves high optical purity, with experimental examples demonstrating enantiomeric excess (ee) values reaching up to 99% and chemical yields exceeding 90% under optimized conditions.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable (3aS,6aR)-1,3-dibenzyltetrahydrofuroimidazole-2,4-dione Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of high-quality intermediates in the pharmaceutical value chain. As a specialized CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and consistency. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, including chiral HPLC analysis to guarantee the optical integrity of every batch of (3aS,6aR)-1,3-dibenzyltetrahydrofuroimidazole-2,4-dione we produce.
We invite you to collaborate with us to leverage this advanced synthetic technology for your biotin projects. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements. Please contact us today to request specific COA data and route feasibility assessments, and let us demonstrate how our expertise can optimize your supply chain and drive down your manufacturing costs.
