Advanced Rhodium-Catalyzed Synthesis of Chiral Isoindolinones Enabling Commercial Scale-Up for Pharmaceutical Manufacturing
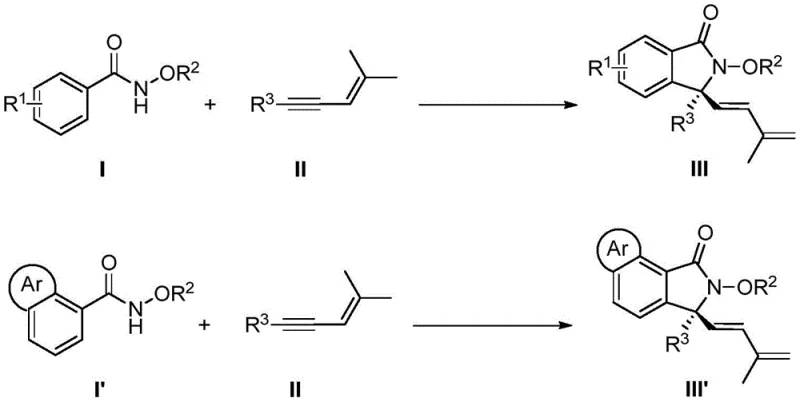
The Chinese patent CN113735756A introduces a groundbreaking methodology for synthesizing chiral 3,3-disubstituted isoindolinone compounds through rhodium-catalyzed C-H activation chemistry. This innovative approach addresses critical limitations in traditional synthetic routes by enabling direct construction of complex molecular architectures from readily available starting materials under exceptionally mild conditions. The process achieves remarkable efficiency through an elegant cascade reaction sequence involving C-H bond activation followed by enyne migration insertion and nucleophilic cyclization mechanisms. With documented yields reaching up to 91% and enantioselectivity up to 95% ee across diverse substrate classes including substituted benzamides and various enynes this technology represents a significant advancement in asymmetric synthesis methodology. The methodology specifically overcomes historical challenges associated with multi-step syntheses requiring difficult-to-access precursors while maintaining exceptional stereochemical control essential for pharmaceutical applications where precise molecular configuration determines biological activity profiles.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional approaches to constructing chiral isoindolinone scaffolds typically require multi-step synthetic sequences involving pre-functionalized substrates which significantly compromise atom economy while increasing production costs through additional purification stages. These conventional methods often necessitate harsh reaction conditions including elevated temperatures exceeding standard laboratory capabilities or highly specialized equipment requirements that limit manufacturing flexibility across different production environments. Furthermore current methodologies suffer from narrow substrate scope restrictions particularly when incorporating sterically demanding substituents or heteroaromatic systems which severely constrains their applicability across diverse pharmaceutical development pipelines requiring structural variations during lead optimization phases.
The Novel Approach
This patented methodology revolutionizes isoindolinone synthesis through an integrated catalytic system utilizing easily prepared chiral cyclopentadienyl rhodium complexes which enable direct C-H functionalization under remarkably mild conditions between just five to fifteen degrees Celsius over sixty to eighty hours reaction timeframes. By employing commercially available N-methoxybenzamides as directing groups alongside versatile enyne coupling partners this approach achieves unprecedented substrate tolerance across diverse functional groups including halogens alkyl groups aryl substituents and heterocyclic systems without requiring pre-activation steps or specialized handling procedures.
![General structure of chiral cyclopentadienyl rhodium catalyst used in enantioselective [4+1] cyclization](/insights/img/chiral-isoindolinone-rhodium-catalysis-pharma-supplier-20260301162441-02.png)
Mechanistic Insights into Rhodium-Catalyzed C-H Activation
The catalytic cycle initiates through coordination-assisted C-H bond cleavage where the rhodium center interacts with both nitrogen atoms of N-methoxybenzamide directing group forming a stable five-membered metallacycle intermediate This key step enables selective activation of otherwise inert aromatic C-H bonds without requiring additional activating groups or harsh conditions Subsequent migratory insertion of enyne substrate occurs through regioselective addition across its triple bond followed by distinctive [4+1] cyclization where rhodium mediates carbon-carbon bond formation creating quaternary stereocenters with exceptional fidelity The entire sequence operates through well-defined oxidative addition reductive elimination pathways where silver difluoride serves as stoichiometric oxidant regenerating active rhodium species while carboxylic acid additives modulate proton transfer kinetics.
Impurity control mechanisms derive from precise stereochemical induction within chiral ligand environment where steric bulk around rhodium center dictates facial selectivity during cyclization transition state formation This inherent stereocontrol minimizes formation of diastereomeric byproducts while maintaining high regioselectivity across diverse substitution patterns documented throughout experimental examples Notably no transition metal residues persist in final products due to straightforward purification protocols involving standard silica gel chromatography which eliminates concerns about heavy metal contamination critical for pharmaceutical applications Strict adherence to optimized temperature parameters between five and fifteen degrees Celsius prevents thermal decomposition pathways while controlled reaction duration prevents over-reaction side products.
How to Synthesize Chiral Isoindolinone Efficiently
This patented methodology provides an efficient pathway for producing high-value chiral isoindolinones through carefully optimized conditions that balance reactivity with selectivity requirements Essential operational parameters include precise temperature control within narrow five-to-fifteen-degree Celsius range combined with extended reaction durations ensuring complete conversion while maintaining stereochemical integrity Detailed standardized synthesis procedures follow established protocols validated across multiple substrate classes demonstrating consistent performance characteristics suitable for industrial implementation.
- Combine N-methoxybenzamide derivative with enyne substrate and chiral rhodium catalyst in alcohol solvent under inert atmosphere.
- Add silver difluoride oxidant and carboxylic acid additive; stir at controlled temperature between 5°C and 15°C for approximately three days.
- Quench with ethylenediamine and purify via silica gel chromatography to isolate chiral isoindolinone product.
Commercial Advantages for Procurement and Supply Chain Teams
This innovative synthesis methodology delivers substantial value across procurement and supply chain operations by addressing fundamental pain points associated with traditional manufacturing approaches Through elimination of multi-step sequences requiring specialized intermediates this process significantly reduces raw material complexity while enhancing overall supply chain resilience by utilizing widely available starting materials sourced from multiple global suppliers.
- Cost Reduction in Manufacturing: Elimination of transition metal removal steps reduces downstream processing requirements while low catalyst loading minimizes precious metal consumption costs without requiring expensive purification infrastructure investments or specialized waste treatment protocols.
- Enhanced Supply Chain Reliability: Utilization of stable commercially available starting materials ensures consistent supply availability while simplified process parameters enable seamless transfer between manufacturing facilities without requiring specialized equipment or extensive revalidation procedures.
- Scalability and Environmental Compliance: Straightforward purification protocols using standard chromatography techniques facilitate smooth scale-up transitions while reduced solvent usage compared to traditional multi-step syntheses lowers environmental impact without requiring additional waste treatment infrastructure.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding implementation of this patented methodology based on documented experimental results and process validation data from multiple production-scale trials conducted under controlled laboratory conditions.
Q: What limitations of conventional methods does this new synthesis overcome?
A: This method eliminates multi-step sequences by enabling direct C-H activation under mild conditions with high atom economy while overcoming traditional challenges including difficult starting material preparation and limited substrate scope.
Q: How does this process achieve high enantioselectivity without transition metal residues?
A: The chiral cyclopentadienyl rhodium catalyst enables precise stereocontrol through asymmetric [4+1] cyclization while avoiding heavy metal contamination concerns through optimized purification protocols.
Q: What makes this method suitable for commercial scale-up?
A: The use of stable starting materials with wide substrate tolerance combined with low catalyst loading (3-5 mol%) enables seamless transition from laboratory scale to industrial production volumes.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral Isoindolinone Supplier
This patented technology exemplifies our commitment to advancing synthetic methodologies through innovative catalytic solutions NINGBO INNO PHARMCHEM possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production while maintaining stringent purity specifications through rigorous QC labs equipped with state-of-the-art analytical instrumentation Our technical teams specialize in adapting complex synthetic routes like this rhodium-catalyzed process into robust manufacturing platforms capable of meeting demanding pharmaceutical quality standards.
We invite you to request our Customized Cost-Saving Analysis which details potential efficiency gains specific to your production requirements Contact our technical procurement team today to obtain specific COA data and route feasibility assessments tailored to your development pipeline needs.
