Advanced Cobalt-Catalyzed Three-Component Synthesis for High-Purity Pharmaceutical Intermediates
The landscape of organic synthesis for complex molecular scaffolds is continuously evolving, driven by the need for more efficient and cost-effective methodologies. A significant breakthrough in this domain is documented in Chinese Patent CN114456089B, which discloses a novel Lewis acid-promoted three-component reaction method involving olefins, oxime esters, and amines. This technology represents a paradigm shift in the construction of amino-nitrile frameworks, which are ubiquitous motifs in medicinal chemistry and agrochemical development. By leveraging a cobalt salt catalyst, this invention successfully achieves the simultaneous difunctionalization of olefins, introducing both amine and cyano groups in a single synthetic operation. This approach not only streamlines the synthetic route but also addresses critical limitations associated with traditional methods, such as the dependence on costly photocatalysts, harsh oxidative conditions, or multi-step sequences that erode overall yield and atom economy.
For research and development teams focused on process optimization, the ability to install these two valuable functional groups concurrently offers a strategic advantage in library synthesis and lead optimization. The cyano group serves as a versatile handle for further transformation into carboxylic acids, amines, or tetrazoles, while the amine functionality provides essential hydrogen-bonding capabilities for biological target engagement. Consequently, mastering this cobalt-catalyzed protocol allows chemists to access diverse chemical space with greater speed and precision, ultimately accelerating the drug discovery timeline.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of molecules containing both amino and cyano functionalities has often required sequential steps or the use of specialized reagents that pose significant challenges for large-scale manufacturing. Traditional difunctionalization strategies frequently rely on precious metal photocatalysts, such as iridium or ruthenium complexes, which are not only prohibitively expensive but also raise concerns regarding residual metal contamination in final pharmaceutical products. Furthermore, many existing protocols necessitate the use of strong oxidants or peroxides to drive the radical generation steps, introducing safety hazards and complicating waste disposal procedures. These factors collectively contribute to higher production costs and extended processing times, creating bottlenecks for procurement and supply chain managers who are tasked with ensuring consistent and economical material flow.
The Novel Approach
In stark contrast to these conventional limitations, the methodology described in patent CN114456089B utilizes an earth-abundant cobalt salt, specifically cobalt chloride (CoCl2), as a highly effective Lewis acid catalyst. This substitution dramatically lowers the raw material costs while maintaining robust catalytic performance. The reaction proceeds under thermal conditions at 90°C in acetonitrile, eliminating the need for specialized photo-reactor equipment and allowing the process to be conducted in standard glass-lined or stainless-steel reactors. As illustrated in the general reaction scheme below, the convergence of an olefin, an oxime ester acting as the alkyl nitrile source, and an amine nucleophile occurs in a single pot, delivering the target 1,2-amino-nitrile product with high efficiency.
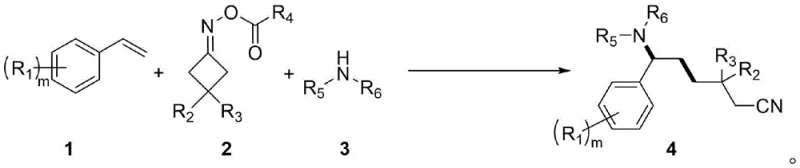
This streamlined approach significantly enhances the step economy of the synthesis, reducing the number of isolation and purification stages required. For a reliable pharmaceutical intermediates supplier, this translates to a more agile manufacturing process capable of responding quickly to market demands. The broad substrate scope demonstrated in the patent, encompassing various substituted styrenes and anilines, further underscores the versatility of this method, making it a powerful tool for cost reduction in pharmaceutical intermediates manufacturing.
Mechanistic Insights into Cobalt-Catalyzed Radical Difunctionalization
Understanding the underlying mechanistic pathway is crucial for R&D directors aiming to adapt this chemistry for specific proprietary targets. The proposed mechanism involves a sophisticated radical relay process mediated by the cobalt catalyst. Initially, the cobalt(II) species facilitates a single-electron transfer to the oxime ester substrate, triggering the homolytic cleavage of the N-O bond. This key activation step generates an iminyl radical intermediate along with a cobalt(III) species and a carboxylate anion. The iminyl radical subsequently undergoes beta-scission to release the critical alkyl cyano radical, which serves as the carbon-centered radical species responsible for the initial functionalization of the olefin.
Once generated, the alkyl cyano radical rapidly adds across the double bond of the styrene derivative, forming a stabilized benzylic radical intermediate. This transient species is then intercepted by the cobalt(III) oxidant generated in the earlier step, resulting in a single-electron oxidation that produces a benzylic carbocation while regenerating the active cobalt(II) catalyst to close the catalytic cycle. Finally, the amine nucleophile attacks this electrophilic carbocation center to forge the new C-N bond, yielding the final amino-nitrile product. The detailed catalytic cycle depicted below highlights the elegant redox neutrality of the system, where the cobalt catalyst shuttles between oxidation states to drive the transformation without the need for external oxidants.
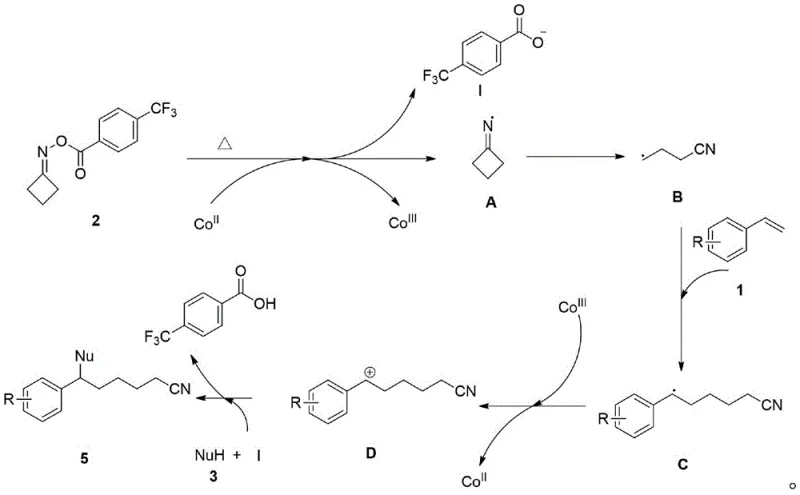
This mechanistic understanding reveals why the reaction exhibits such high functional group tolerance; the radical intermediates are generated and consumed rapidly within the coordination sphere of the catalyst, minimizing side reactions such as homocoupling or polymerization. For technical teams, this implies that high-purity pharmaceutical intermediates can be obtained with minimal impurity profiles, simplifying downstream purification and ensuring compliance with stringent quality standards.
How to Synthesize Amino-Nitrile Derivatives Efficiently
Implementing this cobalt-catalyzed protocol in a laboratory or pilot plant setting requires adherence to specific operational parameters to maximize yield and reproducibility. The process begins with the careful selection of high-quality starting materials, specifically the olefin, the O-acyl oxime, and the amine, which are combined in a molar ratio optimized to drive the equilibrium towards the product. The reaction is typically conducted under an inert atmosphere, such as argon, to prevent the quenching of radical intermediates by oxygen. While the patent outlines a generalized procedure, precise control over temperature and reaction time is essential to balance conversion rates with product stability.
- Charge a reactor with the olefin compound, oxime ester, and amine nucleophile under an inert argon atmosphere.
- Add the cobalt salt catalyst (preferably CoCl2) and organic solvent (preferably acetonitrile) to the mixture.
- Heat the reaction mixture to 90°C and stir for approximately 12 hours, followed by silica gel purification to isolate the target product.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this cobalt-catalyzed methodology offers substantial strategic benefits for procurement managers and supply chain heads looking to optimize their sourcing strategies. The primary driver of value lies in the drastic simplification of the supply chain for raw materials; by replacing expensive photocatalysts and hazardous oxidants with commodity-grade cobalt salts, the direct material costs are significantly reduced. This shift not only lowers the bill of materials but also mitigates the supply risk associated with sourcing specialized reagents that may have limited availability or long lead times. Furthermore, the elimination of light-dependent steps removes the need for capital-intensive photochemical equipment, allowing the reaction to be scaled in existing infrastructure.
- Cost Reduction in Manufacturing: The transition to a thermal cobalt-catalyzed process eliminates the recurring expense of precious metal photocatalysts and the energy costs associated with high-power LED arrays. Additionally, the one-pot nature of the reaction reduces solvent consumption and labor hours related to intermediate isolations. These cumulative efficiencies result in a leaner manufacturing process that delivers substantial cost savings without compromising on the quality of the high-purity pharmaceutical intermediates produced.
- Enhanced Supply Chain Reliability: By utilizing widely available starting materials such as substituted styrenes and anilines, manufacturers can diversify their supplier base and reduce dependency on single-source vendors for exotic reagents. The robustness of the reaction conditions ensures consistent batch-to-batch performance, which is critical for maintaining continuous production schedules. This reliability helps in reducing lead time for high-purity pharmaceutical intermediates, ensuring that downstream drug development projects remain on track.
- Scalability and Environmental Compliance: The absence of peroxide oxidants and the use of standard organic solvents simplify the waste treatment process, aligning with increasingly strict environmental regulations. The commercial scale-up of complex pharmaceutical intermediates is facilitated by the straightforward workup procedure involving silica gel chromatography, which is easily adaptable to industrial flash chromatography or crystallization techniques. This scalability ensures that the technology can meet growing market demands sustainably.
Frequently Asked Questions (FAQ)
To assist technical decision-makers in evaluating the feasibility of this technology for their specific applications, we have compiled a set of frequently asked questions based on the detailed experimental data provided in the patent literature. These insights address common concerns regarding catalyst loading, substrate compatibility, and purification methods. Understanding these nuances is essential for integrating this novel synthetic route into existing process development workflows and ensuring a smooth technology transfer from lab to plant.
Q: What are the primary advantages of using cobalt salts over photocatalysts in this difunctionalization reaction?
A: The use of cobalt salts eliminates the reliance on expensive photocatalysts and external light sources, significantly reducing equipment complexity and operational costs while maintaining high catalytic activity and selectivity.
Q: Does this reaction methodology tolerate diverse functional groups on the olefin and amine substrates?
A: Yes, the protocol demonstrates excellent functional group tolerance, accommodating various substituents such as halogens, alkoxy groups, and trifluoromethyl groups on both the styrene and aniline components without compromising yield.
Q: Is the reaction scalable for industrial production of pharmaceutical intermediates?
A: The reaction utilizes standard heating conditions (90°C) and common solvents like acetonitrile, avoiding hazardous oxidants or specialized photo-reactors, which facilitates straightforward scale-up from laboratory to commercial manufacturing scales.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Amino-Nitrile Derivatives Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced catalytic methodologies like the cobalt-promoted difunctionalization described in CN114456089B. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory discoveries are seamlessly translated into robust industrial processes. Our state-of-the-art facilities are equipped to handle sensitive organometallic chemistry under strict safety protocols, and our rigorous QC labs guarantee that every batch meets stringent purity specifications required by global regulatory bodies.
We invite you to collaborate with us to leverage this cutting-edge technology for your next project. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific molecule, demonstrating how this cobalt-catalyzed route can optimize your budget and timeline. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us help you secure a competitive advantage in the marketplace with high-quality, cost-effective chemical solutions.
