Advanced Manufacturing of 2,6-Diethyl-4-Methylphenol: A Cost-Effective Route for Herbicide Intermediates
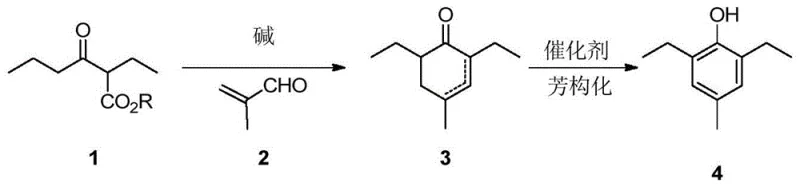
The global demand for high-efficiency herbicides continues to drive innovation in the synthesis of key agrochemical intermediates, specifically targeting molecules like 2,6-diethyl-4-methylphenol, a critical precursor for the herbicide pinoxaden. Patent CN108264449B, published in April 2022, introduces a groundbreaking preparation method that fundamentally shifts the synthetic strategy from modifying existing aromatic rings to constructing them from non-aromatic precursors. This technical breakthrough addresses long-standing inefficiencies in the industry by utilizing a condensation reaction between 2-ethyl-3-oxohexanoate and 2-methylacrolein under alkaline conditions, followed by a catalytic aromatization step. By moving away from the traditional reliance on difficult-to-prepare aniline derivatives or expensive noble metal catalysts, this invention offers a pathway that is not only chemically elegant but also commercially viable for large-scale manufacturing. The strategic pivot to building the aromatic core through cyclization allows for superior control over substitution patterns while drastically simplifying the supply chain requirements for raw materials.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of polysubstituted phenols like 2,6-diethyl-4-methylphenol has been plagued by significant technical and economic hurdles that hinder efficient industrial production. One prevalent method involves the diazotization and hydrolysis of 2,6-diethyl-4-methylaniline, a process notorious for generating substantial quantities of hazardous three wastes and posing severe safety risks due to the instability of diazonium intermediates. Another common approach utilizes the alkylation of p-methylphenol with ethylene gas in the presence of rhodium catalysts; while effective, this route is economically prohibitive due to the extreme cost of rhodium and the necessity for specialized ligands, alongside the inherent dangers of handling high-pressure ethylene gas. Furthermore, multi-step syntheses starting from halogenated phenols often require extensive protection and deprotection strategies, leading to low overall yields, excessive solvent consumption, and the accumulation of heavy metal residues that complicate purification and environmental compliance.
The Novel Approach
In stark contrast to these legacy methods, the novel approach disclosed in the patent employs a completely different synthesis strategy that constructs the aromatic ring de novo from acyclic precursors. By reacting 2-ethyl-3-oxohexanoate with 2-methylacrolein under basic conditions, the process efficiently forms a cyclohexenone intermediate which is subsequently aromatized to the target phenol. This methodology bypasses the need for expensive transition metals like rhodium in the initial carbon-carbon bond formation steps and avoids the safety hazards associated with diazotization or high-pressure hydrogenation. The use of commodity chemicals such as simple esters and aldehydes ensures that raw material sourcing is robust and cost-effective, while the reaction conditions—typically ranging from 60°C to 80°C for condensation—are mild enough to be safely managed in standard chemical reactors without requiring specialized high-pressure equipment.
Mechanistic Insights into Base-Catalyzed Condensation and Aromatization
The core of this synthetic innovation lies in a sophisticated cascade of reactions initiated by a base-catalyzed condensation, likely proceeding through a mechanism analogous to the Robinson annulation. In the first stage, a strong base such as sodium methoxide or sodium hydroxide facilitates the enolization of the 2-ethyl-3-oxohexanoate ester, generating a nucleophilic enolate species. This enolate attacks the electron-deficient double bond of 2-methylacrolein in a Michael addition, establishing the carbon skeleton required for the six-membered ring. Subsequent intramolecular aldol condensation and dehydration steps close the ring to form the 2,6-diethyl-4-methyl-2-cyclohexenone or its 3-ene isomer. The precise control of stoichiometry, with a molar ratio of aldehyde to ester between 1.2:1 and 1.8:1, is critical to minimizing polymerization side reactions of the aldehyde and ensuring high conversion rates of the limiting ester reagent.
Following the formation of the cyclic enone intermediate, the process transitions to an aromatization phase that restores aromaticity to the ring system. This step can be achieved using either an organic base catalyst like DABCO at ambient temperatures or a heterogeneous metal catalyst such as Pd/C under heating. When using DABCO, the mechanism likely involves base-promoted tautomerization and oxidative dehydrogenation facilitated by trace oxygen or disproportionation pathways, yielding the phenol without introducing heavy metal contaminants. Alternatively, the Pd/C catalyzed route operates through a dehydrogenation mechanism at elevated temperatures around 140°C, which drives the equilibrium towards the thermodynamically stable aromatic product with exceptional yields reaching up to 95%. This dual-option catalytic flexibility allows manufacturers to choose between a metal-free process for higher purity specifications or a metal-catalyzed process for maximum throughput.
How to Synthesize 2,6-Diethyl-4-Methylphenol Efficiently
To implement this synthesis effectively, operators must strictly adhere to the optimized reaction parameters regarding temperature, solvent choice, and catalyst loading to maximize yield and minimize impurities. The process begins with the preparation of the reaction mixture in a polar solvent system such as methanol or tetrahydrofuran, where the base is dissolved prior to the addition of the ketone ester substrate. Careful temperature control during the dropwise addition of 2-methylacrolein is essential to manage the exotherm and prevent runaway reactions, with the mixture typically maintained between 60°C and 80°C to ensure complete conversion to the cyclohexenone intermediate. For the subsequent aromatization, the choice of catalyst dictates the workup procedure; using DABCO allows for simple acid washes to remove the organic base, whereas Pd/C requires filtration to recover the catalyst, both methods offering distinct advantages depending on the desired purity profile and equipment availability.
- Perform a condensation reaction between 2-ethyl-3-oxohexanoate and 2-methylacrolein using an alkali catalyst such as sodium methoxide or sodium hydroxide in a solvent like methanol or THF at 60-80°C to form the cyclohexenone intermediate.
- Subject the resulting 2,6-diethyl-4-methyl-2-cyclohexenone intermediate to an aromatization reaction using an organic base catalyst like DABCO or a metal catalyst like Pd/C to yield the final 2,6-diethyl-4-methylphenol product.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, this patented method offers transformative advantages by decoupling production from the volatile markets of precious metals and hazardous gases. The elimination of rhodium catalysts removes a significant cost driver and supply risk, as rhodium prices are subject to extreme fluctuations and geopolitical constraints that can disrupt manufacturing schedules. Furthermore, the reliance on bulk commodity chemicals like ethyl acetoacetate derivatives and methacrolein ensures a stable and diversified supply base, allowing procurement managers to negotiate better terms and secure long-term contracts without fear of single-source bottlenecks. The simplified process flow also translates to reduced operational complexity, meaning lower capital expenditure on specialized high-pressure reactors and safety systems, which ultimately contributes to a more competitive cost structure for the final agrochemical intermediate.
- Cost Reduction in Manufacturing: The most immediate financial benefit arises from the complete removal of expensive noble metal catalysts and complex ligands that characterize traditional alkylation routes. By substituting these high-cost inputs with inexpensive inorganic bases like sodium hydroxide or organic amines like DABCO, the direct material cost per kilogram of product is drastically reduced. Additionally, the shorter synthetic route compared to multi-step protection-deprotection sequences minimizes solvent usage and energy consumption, leading to substantial operational savings that enhance the overall profit margin for manufacturers producing pinoxaden intermediates.
- Enhanced Supply Chain Reliability: Utilizing widely available starting materials such as 2-ethyl-3-oxohexanoate and 2-methylacrolein significantly mitigates the risk of raw material shortages that often plague specialty chemical supply chains. Unlike processes dependent on ethylene gas, which requires dedicated infrastructure and poses logistics challenges, these liquid reagents can be stored and handled using standard tank farms and dosing systems. This flexibility allows for more agile production planning and reduces the lead time associated with sourcing specialized precursors, ensuring consistent delivery schedules for downstream herbicide formulators.
- Scalability and Environmental Compliance: The process is inherently designed for industrial scale-up, featuring reaction conditions that are easily manageable in large batch reactors without the need for exotic engineering controls. The reduction in hazardous waste generation, particularly the avoidance of heavy metal sludge from diazotization or rhodium residues, simplifies wastewater treatment and lowers disposal costs. This alignment with green chemistry principles not only reduces the environmental footprint but also ensures smoother regulatory compliance in jurisdictions with stringent emission standards, facilitating faster approval for commercial production facilities.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel synthesis route for 2,6-diethyl-4-methylphenol. These answers are derived directly from the experimental data and comparative analysis provided in the patent documentation, focusing on yield optimization, catalyst selection, and safety protocols. Understanding these details is crucial for technical teams evaluating the feasibility of adopting this method for their own manufacturing operations or for procurement specialists assessing the quality implications of the new process.
Q: What are the primary advantages of this new synthesis method over traditional rhodium-catalyzed routes?
A: The new method eliminates the need for extremely expensive rhodium metal catalysts and complex ligands required in traditional alkylation processes. It utilizes cheap and readily available raw materials like 2-ethyl-3-oxohexanoate and 2-methylacrolein, significantly reducing raw material costs and supply chain risks associated with precious metals.
Q: How does this process improve safety compared to diazotization methods?
A: Unlike the conventional diazotization of 2,6-diethyl-4-methylaniline which generates large amounts of hazardous waste and poses explosion risks, this condensation-aromatization route operates under milder conditions. It avoids the use of unstable diazonium salts and high-pressure ethylene gas, resulting in a safer operational profile suitable for large-scale industrial production.
Q: What yields can be expected from the aromatization step using different catalysts?
A: The patent data indicates that using Pd/C as a catalyst in N,N-dimethylacetamide at 140°C can achieve yields up to 95%. Alternatively, using an organic base like DABCO at room temperature provides yields around 75-81%, offering a metal-free option that simplifies downstream purification and reduces heavy metal contamination concerns.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2,6-Diethyl-4-Methylphenol Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of robust and scalable synthetic routes in the modern agrochemical supply chain, and we have positioned ourselves as a leader in translating such innovative patents into commercial reality. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial plant is seamless and efficient. We maintain stringent purity specifications and operate rigorous QC labs equipped with advanced analytical instrumentation to guarantee that every batch of 2,6-diethyl-4-methylphenol meets the exacting standards required for herbicide synthesis, providing our partners with absolute confidence in material consistency.
We invite global agrochemical manufacturers and procurement leaders to collaborate with us to leverage this cost-effective technology for their supply chains. By contacting our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements, along with specific COA data and route feasibility assessments. Let us help you optimize your production costs and secure a reliable supply of high-quality intermediates, driving value and efficiency in your herbicide manufacturing operations.
