Advanced Synthesis of S-3-(4-aminophenyl)piperidine for Scalable PARP Inhibitor Production
The pharmaceutical industry's relentless pursuit of potent oncology therapeutics has placed significant demand on the supply chain for high-purity chiral intermediates, specifically for the synthesis of Poly (ADP-ribose) polymerase (PARP) inhibitors. Patent CN109134351B, published in early 2022, introduces a groundbreaking synthetic methodology for S-3-(4-aminophenyl)piperidine, a critical building block for drugs like Niraparib. This technology addresses long-standing bottlenecks in optical purity and raw material costs, offering a robust pathway for commercial-scale manufacturing. By leveraging a strategic sequence of Suzuki coupling, selective reductions, and chiral resolution, the process achieves an enantiomeric excess exceeding 99%, setting a new benchmark for quality in this sector.
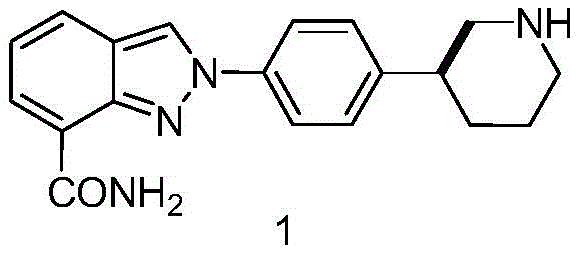
For R&D directors evaluating process viability, the structural integrity and impurity profile of the intermediate are paramount. The patented route utilizes 3-pyridineboronic acid and 4-bromonitrobenzene as starting materials, initiating a Suzuki-Miyaura cross-coupling reaction. Unlike traditional methods that might struggle with regioselectivity or require harsh conditions, this approach employs optimized palladium catalysis, such as Pd(dppf)Cl2·DCM, ensuring high conversion rates under mild thermal conditions ranging from 60°C to 100°C. The subsequent transformation involves a chemoselective reduction of the nitro group using iron powder and hydrochloric acid, a method chosen for its operational safety and ease of workup compared to catalytic hydrogenation of nitro groups which can sometimes lead to over-reduction or safety hazards.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior art, such as the route described in J. Med. Chem. (2009), relied heavily on 4-iodonitrobenzene as a coupling partner. While chemically feasible, the use of iodo-substituted aromatics imposes a severe economic burden due to the high market price of iodine-containing reagents compared to their bromine counterparts. Furthermore, the historical reliance on direct chiral column chromatography for separation presented a formidable barrier to industrial scale-up. Chromatographic separation, while effective on a gram scale, is notoriously difficult to translate to tonnage production due to solvent consumption, low throughput, and high operational expenditures. Most critically, these legacy methods often capped at an enantiomeric excess of approximately 82%, which is insufficient for modern regulatory standards requiring near-perfect optical purity to minimize off-target biological effects.
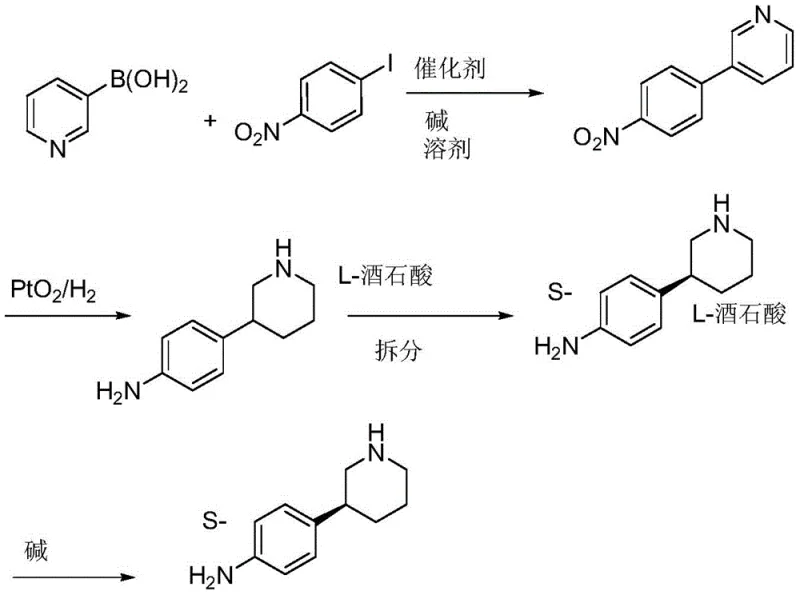
The Novel Approach
The innovative strategy outlined in CN109134351B circumvents these issues through a clever 'protection-reduction-resolution' manifold. By introducing an acylation step using acetic anhydride prior to the reduction of the pyridine ring, the process modifies the electronic and steric environment of the molecule. This acetylated intermediate serves as a superior substrate for chiral resolution using L-camphorsulfonic acid. The acetyl group acts as a crucial recognition handle, allowing the resolving agent to differentiate between enantiomers with high fidelity. Following resolution, a simple hydrolysis step removes the protecting group, yielding the free amine with exceptional optical purity. This sequence eliminates the need for preparative chiral HPLC, replacing it with a scalable crystallization-based resolution that is inherently more suitable for large-volume manufacturing.
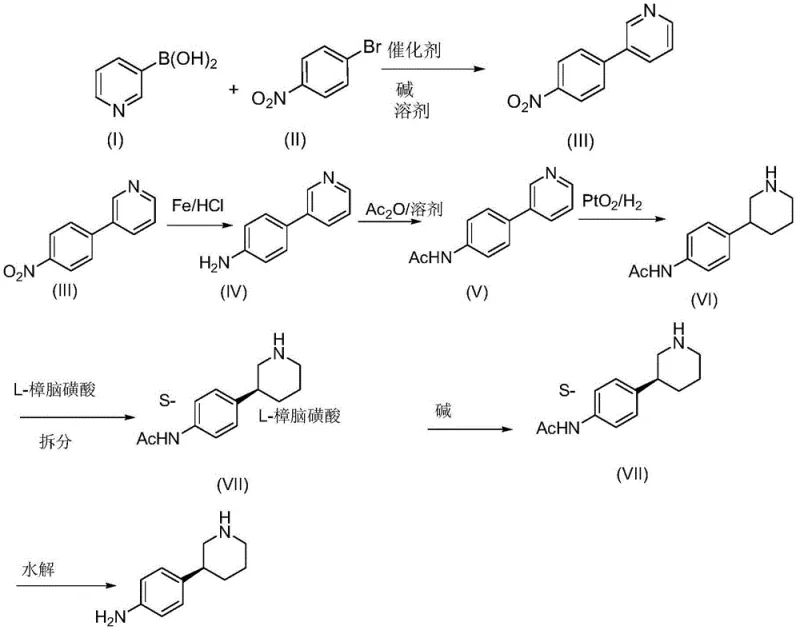
Mechanistic Insights into Suzuki Coupling and Chiral Resolution
The core of this synthesis lies in the precise orchestration of functional group transformations. The initial Suzuki coupling is facilitated by a base, typically potassium carbonate or sodium carbonate, which activates the boronic acid for transmetallation with the palladium catalyst. The choice of solvent systems, such as THF/water or dioxane/water mixtures, is critical to solubilize both the organic halides and the inorganic base, ensuring a homogeneous reaction environment that maximizes yield. Following the coupling, the reduction of the nitro group to an aniline derivative is executed using iron powder in acidic media. This heterogeneous reduction is highly selective for the nitro group, leaving the pyridine ring intact, which is essential for the subsequent steps.
The mechanistic advantage becomes most apparent during the chiral resolution phase. The racemic 3-(4-acetamidophenyl)piperidine is treated with L-camphorsulfonic acid in a solvent system like isopropanol/water. The diastereomeric salts formed have distinct solubility profiles; the desired S-enantiomer salt crystallizes preferentially upon cooling. The presence of the acetamido group enhances the rigidity and hydrogen-bonding capability of the molecule, stabilizing the crystal lattice of the desired diastereomer. This thermodynamic control allows for the isolation of the S-enantiomer with an ee value reaching up to 99.7% after recrystallization, a level of purity that is difficult to achieve via kinetic resolution or enzymatic methods alone.
How to Synthesize S-3-(4-aminophenyl)piperidine Efficiently
The synthesis protocol detailed in the patent provides a clear roadmap for laboratory and pilot-scale production, emphasizing reproducibility and safety. The process begins with the rigorous exclusion of oxygen during the coupling phase to prevent catalyst deactivation, followed by controlled addition of reagents to manage exotherms during the reduction steps.
- Perform Suzuki coupling between 3-pyridineboronic acid and 4-bromonitrobenzene using a palladium catalyst to form 3-(4-nitrophenyl)pyridine.
- Reduce the nitro group using iron powder and hydrochloric acid, followed by acylation with acetic anhydride to protect the amine.
- Hydrogenate the pyridine ring using PtO2, resolve the racemate with L-camphorsulfonic acid, and hydrolyze to obtain the final chiral amine.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition from iodo-based to bromo-based chemistry represents a significant opportunity for cost optimization. The substitution of 4-iodonitrobenzene with 4-bromonitrobenzene drastically reduces the raw material bill of materials, as brominated aromatics are commodity chemicals with stable global supply chains. Furthermore, the elimination of chiral chromatography removes a major bottleneck in production throughput. Crystallization-based resolution is not only faster but also consumes significantly less solvent, aligning with green chemistry principles and reducing waste disposal costs. This streamlined workflow ensures a more reliable supply of high-purity intermediates, mitigating the risk of production delays associated with complex purification techniques.
- Cost Reduction in Manufacturing: The replacement of expensive iodine-containing starting materials with cost-effective bromine analogs results in substantial savings on raw material procurement. Additionally, the shift from chromatographic separation to crystallization-based resolution significantly lowers operational expenses related to solvent usage and equipment time. The use of iron powder for nitro reduction is also economically favorable compared to precious metal catalysts often required for similar transformations, further driving down the overall production cost per kilogram.
- Enhanced Supply Chain Reliability: By utilizing widely available commodity chemicals like 3-pyridineboronic acid and 4-bromonitrobenzene, the supply chain becomes more resilient to market fluctuations. The robustness of the Suzuki coupling and iron reduction steps ensures consistent batch-to-batch quality, reducing the likelihood of failed batches that can disrupt downstream API synthesis. The scalability of the resolution step guarantees that supply can be ramped up quickly to meet clinical or commercial demand without the need for specialized chromatographic infrastructure.
- Scalability and Environmental Compliance: The process is designed with industrial scalability in mind, avoiding hazardous reagents and high-pressure conditions where possible. The aqueous workups and crystallization steps generate less hazardous waste compared to silica gel chromatography, simplifying environmental compliance and waste treatment. The ability to recycle solvents like ethanol and isopropanol further enhances the environmental profile of the manufacturing process, making it attractive for facilities aiming to reduce their carbon footprint.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and benefits of this patented synthesis route. These insights are derived directly from the experimental data and claims within the patent documentation, providing clarity on process capabilities.
Q: What is the enantiomeric excess (ee) achieved by this new synthesis method?
A: The patented method achieves an enantiomeric excess of greater than 99%, significantly surpassing the 82% ee limit of conventional literature methods.
Q: Why is 4-bromonitrobenzene preferred over 4-iodonitrobenzene in this process?
A: 4-Bromonitrobenzene is substantially more cost-effective than the iodo-analog used in older routes, driving down raw material costs without compromising reaction efficiency.
Q: How does the acylation step improve chiral resolution?
A: Acylation protects the aromatic amine and modifies the molecular recognition site, enhancing the interaction with L-camphorsulfonic acid for superior chiral separation.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable S-3-(4-aminophenyl)piperidine Supplier
At NINGBO INNO PHARMCHEM, we understand that the successful commercialization of oncology drugs like Niraparib depends on the consistent availability of high-quality chiral intermediates. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and reliability. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch of S-3-(4-aminophenyl)piperidine meets the >99% ee standard required for modern pharmaceutical applications.
We invite you to collaborate with us to leverage this advanced synthetic technology for your pipeline. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our optimized manufacturing process can enhance your supply chain efficiency and reduce overall project costs.
