Advanced Palladium-Catalyzed Carbonylation for Scalable 2,3-Dihydroquinolone Manufacturing
Advanced Palladium-Catalyzed Carbonylation for Scalable 2,3-Dihydroquinolone Manufacturing
The development of efficient synthetic routes for nitrogen-containing heterocycles remains a cornerstone of modern medicinal chemistry, particularly for scaffolds exhibiting potent biological activity. Patent CN112239456B discloses a groundbreaking preparation method for substituted 2,3-dihydroquinolone compounds, utilizing a transition metal palladium-catalyzed carbonylation reaction. This technology addresses critical challenges in the synthesis of these valuable intermediates, which serve as key structural motifs in various bioactive molecules, including antitumor agents and analgesics. As illustrated in the biological context, compounds featuring the 2,3-dihydroquinolone skeleton, such as the antitumor agents A and B and the analgesic agent C shown below, demonstrate significant therapeutic potential against human cancer cells and pain pathways.
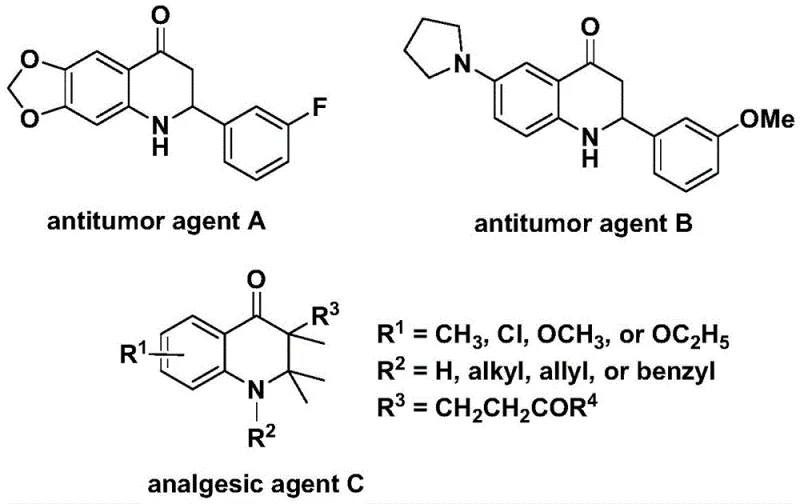
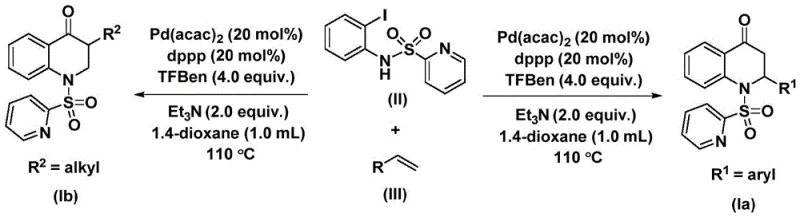
The significance of this patent lies not only in the biological relevance of the target molecules but also in the methodological innovation it introduces. Traditional synthetic approaches often suffer from harsh reaction conditions, limited substrate scope, or the requirement for hazardous reagents. In contrast, the disclosed method employs N-pyridine sulfonyl-o-iodoaniline and olefins as readily available starting materials, reacting them in the presence of a palladium catalyst and a solid carbon monoxide substitute. This approach facilitates the direct and efficient construction of the carbonyl-containing six-membered heterocyclic ring, offering a streamlined pathway that is highly amenable to industrial application and large-scale production.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 2,3-dihydroquinolone skeletons has relied on methodologies that present substantial operational and safety hurdles for process chemists and manufacturing teams. Conventional carbonylation reactions frequently necessitate the use of carbon monoxide gas, which poses severe safety risks due to its high toxicity and flammability, requiring specialized high-pressure equipment and rigorous safety protocols that inflate capital expenditure. Furthermore, many existing literature reports describe synthetic routes with poor atom economy or those that require multi-step sequences to install the carbonyl functionality, leading to cumulative yield losses and increased waste generation. The limited tolerance for diverse functional groups in older methods often restricts the chemical space accessible to medicinal chemists, forcing them to compromise on molecular design or engage in tedious protection-deprotection strategies that delay project timelines.
The Novel Approach
The novel approach detailed in patent CN112239456B represents a paradigm shift by utilizing a solid carbon monoxide surrogate, specifically 1,3,5-trimesic acid phenol ester (TFBen), thereby circumventing the dangers associated with gaseous CO. This method enables the direct coupling of N-pyridine sulfonyl-o-iodoaniline with a wide variety of olefins under relatively mild thermal conditions. As depicted in the general reaction scheme below, the process integrates the carbonylation and cyclization steps into a single pot, significantly reducing processing time and solvent consumption. The use of a palladium catalyst system comprising bis(acetylacetone)palladium and a diphosphine ligand ensures high catalytic turnover and excellent regioselectivity, allowing for the precise installation of aryl or alkyl groups at the 2- and 3-positions of the quinolone ring.
Mechanistic Insights into Palladium-Catalyzed Carbonylative Cyclization
To fully appreciate the robustness of this synthetic methodology, one must examine the underlying catalytic cycle that drives the transformation. The reaction initiates with the oxidative addition of the palladium(0) species into the carbon-iodine bond of the N-pyridine sulfonyl-o-iodoaniline substrate, generating a reactive aryl-palladium intermediate. This step is critical as it activates the aromatic ring for subsequent functionalization. Following this activation, the carbon monoxide molecule, which is released in situ from the thermal decomposition of the TFBen additive, inserts into the palladium-carbon bond. This insertion forms an acyl-palladium intermediate, effectively building the carbonyl carbon of the final lactam structure directly into the growing molecular framework without the need for external gas feeds.
Subsequently, the olefin substrate coordinates to the palladium center and undergoes migratory insertion into the acyl-palladium bond. This step determines the regiochemistry of the alkyl or aryl substituent at the 3-position of the dihydroquinolone ring. The specific structures of the starting materials, such as the N-pyridine sulfonyl-o-iodoaniline shown in formula (II) and the diverse olefins represented by formula (III), play a pivotal role in the efficiency of this coordination. Finally, an intramolecular nucleophilic attack by the nitrogen atom followed by reductive elimination releases the substituted 2,3-dihydroquinolone product and regenerates the active palladium catalyst, closing the catalytic loop. This mechanistic elegance ensures that the reaction proceeds with high fidelity, minimizing the formation of side products and simplifying the downstream purification process.


How to Synthesize Substituted 2,3-Dihydroquinolone Efficiently
The practical implementation of this chemistry is designed for ease of execution in both laboratory and pilot plant settings. The protocol involves charging a reaction vessel with the palladium catalyst, ligand, base, CO surrogate, and substrates in an aprotic organic solvent such as 1,4-dioxane. The mixture is then heated to promote the cascade of reactions described in the mechanistic section. For R&D teams looking to replicate or adapt this chemistry, the standardized procedure outlined in the patent provides a reliable baseline for optimization. The detailed standardized synthesis steps are provided in the guide below to ensure reproducibility and safety during scale-up operations.
- Charge a reaction vessel with bis(acetylacetone)palladium, dppp ligand, triethylamine, TFBen (CO source), N-pyridine sulfonyl-o-iodoaniline, and the desired olefin in 1,4-dioxane.
- Heat the reaction mixture to 110 °C and stir continuously for 48 hours to ensure complete conversion of the starting materials.
- Upon completion, filter the mixture, adsorb onto silica gel, and purify via column chromatography to isolate the target 2,3-dihydroquinolone product.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this palladium-catalyzed carbonylation route offers compelling advantages for procurement managers and supply chain directors focused on cost efficiency and risk mitigation. The substitution of hazardous carbon monoxide gas with a stable solid surrogate drastically reduces the regulatory burden and infrastructure costs associated with handling toxic gases, thereby enhancing the overall safety profile of the manufacturing facility. This shift not only protects personnel but also minimizes the potential for production stoppages due to safety audits or gas supply disruptions, ensuring a more resilient supply chain for critical pharmaceutical intermediates.
- Cost Reduction in Manufacturing: The elimination of high-pressure gas equipment and the use of commercially available, inexpensive starting materials like olefins and iodoanilines contribute to significant operational cost savings. By consolidating multiple synthetic steps into a single carbonylative cyclization, the process reduces solvent usage, labor hours, and waste disposal costs, leading to a more economical production model for high-purity pharmaceutical intermediates.
- Enhanced Supply Chain Reliability: The broad substrate compatibility of this method means that manufacturers can source a wide range of olefins and aniline derivatives from standard chemical suppliers without needing custom synthesis for every new analog. This flexibility allows for rapid adaptation to changing market demands or raw material availability, reducing lead times for high-purity substituted quinolones and ensuring continuous supply for downstream drug development programs.
- Scalability and Environmental Compliance: The reaction conditions, utilizing moderate temperatures and standard organic solvents, are inherently scalable from gram to multi-kilogram batches without requiring exotic engineering solutions. Furthermore, the high atom efficiency and reduced waste generation align with green chemistry principles, facilitating easier compliance with increasingly stringent environmental regulations and supporting sustainable manufacturing practices in the fine chemical industry.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and scope of this patented technology. These answers are derived directly from the experimental data and specifications provided in the patent documentation to assist technical teams in evaluating the feasibility of this route for their specific projects.
Q: What is the primary advantage of using TFBen over carbon monoxide gas?
A: Using 1,3,5-trimethoxyphenyl carbonate (TFBen) as a solid carbon monoxide surrogate eliminates the need for handling hazardous high-pressure CO gas cylinders, significantly improving operational safety and simplifying reactor requirements for industrial scale-up.
Q: What types of substituents are compatible with this palladium-catalyzed method?
A: The method exhibits excellent substrate compatibility, tolerating various functional groups including methyl, tert-butyl, methoxy, fluorine, and chlorine on the aryl ring, as well as linear, branched alkyl, and silyl groups on the olefin component.
Q: What are the typical reaction yields for this synthesis?
A: Experimental data indicates robust performance with isolated yields ranging typically from 60% to 88%, depending on the specific steric and electronic nature of the olefin and aniline substrates employed.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Substituted 2,3-Dihydroquinolone Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic importance of robust synthetic methodologies like the one described in CN112239456B for the development of next-generation therapeutics. Our team of expert process chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that promising laboratory discoveries can be seamlessly transitioned into reliable commercial supplies. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of substituted 2,3-dihydroquinolone intermediates meets the exacting standards required by global pharmaceutical partners.
We invite you to collaborate with us to leverage this advanced carbonylation technology for your drug discovery pipelines. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are prepared to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our manufacturing capabilities can accelerate your project timelines while optimizing your overall cost structure.
