Advanced Manufacturing of UV-1577 Intermediates via Solvent-Free Friedel-Crafts Alkylation
The chemical industry is currently witnessing a significant paradigm shift in the synthesis of high-performance ultraviolet absorbers, driven by the urgent need for greener, more efficient manufacturing protocols. A pivotal development in this sector is documented in Chinese Patent CN116023343A, which discloses a revolutionary preparation method for the UV-1577 intermediate, specifically 2-chloro-4,6-diphenyl-1,3,5-triazine. This innovation addresses critical bottlenecks in traditional synthetic routes by eliminating hazardous auxiliary solvents and drastically optimizing reaction kinetics. For R&D directors and procurement specialists seeking a reliable UV absorber intermediate supplier, this technology represents a substantial leap forward in process intensification. By utilizing raw material benzene simultaneously as a reactant and a solvent, the method not only simplifies the operational workflow but also significantly enhances the economic viability of producing high-purity OLED material precursors and polymer additives. The strategic elimination of chlorinated solvents aligns perfectly with modern environmental compliance standards, offering a robust solution for cost reduction in polymer additive manufacturing while maintaining exceptional product quality.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 2-chloro-4,6-diphenyl-1,3,5-triazine has been plagued by inefficient multi-step processes that impose heavy burdens on both operational costs and environmental safety. One prevalent conventional technique involves a cyclization method where urea reacts with benzonitrile under organic base catalysis to form a hydroxy-intermediate, which subsequently requires chlorination with thionyl chloride. As illustrated in the reaction pathway below, this approach is inherently flawed due to its excessive step count and mediocre overall yield, which typically hovers around only 71%. Furthermore, the reliance on thionyl chloride introduces severe handling hazards and generates corrosive waste streams that complicate downstream processing. Another traditional route employs Grignard reagents, necessitating the use of magnesium powder and halogenated benzenes. This method is particularly notorious for its harsh reaction conditions and the inherent risk of explosion caused by peroxide formation during solvent recovery. With yields stagnating at approximately 72% and requiring rigorous anhydrous conditions, these legacy methods fail to meet the demands of modern commercial scale-up of complex polymer additives.
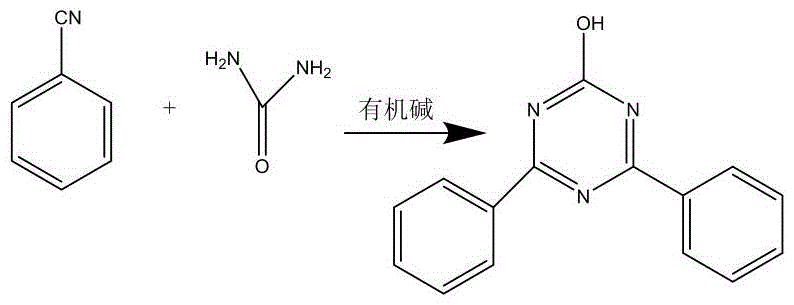
The Novel Approach
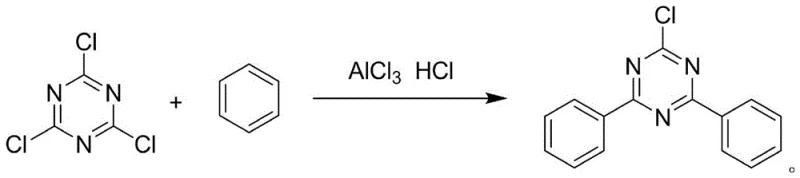
In stark contrast to these cumbersome legacy protocols, the novel approach detailed in the patent utilizes a direct Friedel-Crafts alkylation strategy that fundamentally redefines the reaction system architecture. By selecting aluminum trichloride as the primary catalyst and employing concentrated hydrochloric acid or hydrogen chloride gas as a co-catalyst, the process achieves a remarkable transformation efficiency. The most groundbreaking aspect of this methodology is the complete removal of auxiliary solvents such as o-dichlorobenzene or chlorobenzene, which were previously deemed necessary to provide a suitable reaction medium. Instead, the raw material benzene serves a dual function, acting as both the substrate and the solvent, which dramatically reduces the volume of waste generated and lowers raw material procurement costs. This streamlined design not only shortens the Friedel-Crafts reaction time significantly but also improves the production environment by minimizing volatile organic compound emissions. For supply chain heads focused on reducing lead time for high-purity UV absorber intermediates, this single-step optimization offers a compelling value proposition that translates directly into enhanced market responsiveness and operational agility.
Mechanistic Insights into FeCl3-Catalyzed Cyclization
The core of this technological breakthrough lies in the precise manipulation of electrophilic aromatic substitution dynamics within the triazine ring system. The reaction mechanism proceeds through the activation of cyanuric chloride by the Lewis acid catalyst, aluminum trichloride, which generates a highly reactive electrophilic species capable of attacking the electron-rich benzene ring. Unlike previous iterations of alkylation that suffered from poor selectivity at elevated temperatures, this novel process leverages a tightly controlled temperature window between 10°C and 50°C to govern the reaction trajectory. This thermal regulation is critical for suppressing side reactions and ensuring that the substitution occurs selectively at the desired positions on the triazine core. The addition of hydrogen chloride gas or concentrated hydrochloric acid further stabilizes the reaction intermediate, facilitating the departure of the chloride leaving group and driving the equilibrium towards the formation of the 2-chloro-4,6-diphenyl-1,3,5-triazine product. By optimizing the mass ratio of benzene to cyanuric chloride to between 1:6 and 1:15, the system maintains an ideal concentration gradient that maximizes collision frequency without promoting oligomerization or degradation.
Impurity control in this synthesis is achieved through a combination of stoichiometric precision and kinetic management, ensuring that the final product meets stringent purity specifications required for advanced applications. The patent data indicates that by maintaining the reaction time between 5 hours and 30 hours depending on the specific temperature setpoint, the formation of mono-substituted or tri-substituted byproducts is effectively minimized. For instance, operating at 18°C for 10 hours has been shown to yield products with liquid phase purity exceeding 98%, demonstrating the robustness of the impurity suppression mechanism. This level of control is vital for R&D teams who require consistent batch-to-batch reproducibility when developing new polymer formulations. The absence of extraneous solvents also means that there are fewer opportunities for solvent-derived impurities to become entrapped in the crystal lattice of the final product, thereby simplifying the purification process. Consequently, the resulting intermediate exhibits superior thermal stability and compatibility, making it an ideal candidate for high-performance UV absorber applications where long-term durability is paramount.
How to Synthesize 2-Chloro-4,6-diphenyl-1,3,5-triazine Efficiently
Implementing this advanced synthesis route requires adherence to specific operational parameters to fully realize the benefits of the solvent-free design. The process begins with the careful charging of benzene, cyanuric chloride, and aluminum trichloride into a reaction vessel equipped with agitation and temperature control systems. It is imperative to maintain a nitrogen atmosphere throughout the procedure to prevent moisture ingress, which could deactivate the catalyst and compromise yield. The reaction mixture is then subjected to a gradual temperature ramp, holding within the optimal range of 10°C to 50°C for a duration determined by the specific kinetic profile desired. Upon completion, the reaction is quenched by pouring into water, followed by the recovery of unreacted benzene through distillation for reuse, reinforcing the circular economy aspects of the process. The detailed standardized synthesis steps see the guide below for a comprehensive breakdown of the operational protocol.
- Charge benzene, cyanuric chloride, and aluminum trichloride into a reactor under nitrogen protection.
- Control the reaction temperature between 10°C and 50°C for a duration of 5 to 30 hours.
- Quench the reaction in water, recover benzene via distillation, and filter the solid product.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this novel synthesis method offers profound advantages that extend far beyond simple chemical yield improvements, addressing critical pain points in global supply chain management. By eliminating the need for expensive and environmentally taxing solvents like o-dichlorobenzene, manufacturers can achieve substantial cost savings in raw material procurement and waste disposal fees. This reduction in auxiliary chemical consumption directly translates to a lower cost of goods sold, allowing for more competitive pricing strategies in the global market for specialty chemicals. Furthermore, the simplification of the process flow removes the necessity for specialized equipment such as high-power ultrasonic devices, which were previously required to reduce catalyst loading in older methods. This equipment simplification reduces capital expenditure requirements and minimizes maintenance downtime, thereby enhancing the overall reliability of the production line. For procurement managers, this means a more stable supply source with reduced risk of production interruptions due to equipment failure or regulatory changes regarding solvent usage.
- Cost Reduction in Manufacturing: The elimination of auxiliary solvents and the reduction in catalyst usage create a leaner manufacturing process that significantly lowers operational expenditures. Without the need to purchase, store, and dispose of large volumes of chlorinated solvents, the financial burden on the production facility is drastically reduced. Additionally, the ability to recover and recycle benzene directly from the reaction mixture further amplifies these savings, creating a closed-loop system that maximizes resource efficiency. This economic efficiency allows suppliers to offer more attractive pricing structures without compromising on quality, providing a distinct competitive edge in price-sensitive markets.
- Enhanced Supply Chain Reliability: The robustness of this synthetic route ensures a consistent and reliable supply of high-purity intermediates, which is crucial for downstream manufacturers operating on tight schedules. The simplified process reduces the number of potential failure points, minimizing the likelihood of batch failures that could disrupt the supply chain. Moreover, the use of readily available raw materials such as benzene and cyanuric chloride mitigates the risk of supply shortages associated with niche reagents. This reliability fosters stronger partnerships between suppliers and clients, as it guarantees the continuity of production for critical end-products like UV-stabilized polymers and coatings.
- Scalability and Environmental Compliance: The process is inherently designed for scalability, utilizing standard reaction vessels and avoiding complex device modifications that often hinder scale-up efforts. This ease of translation from laboratory to pilot to full commercial scale ensures that demand surges can be met without significant lead times for new equipment installation. From an environmental standpoint, the reduction in wastewater generation and the avoidance of toxic solvents align with increasingly stringent global environmental regulations. This compliance not only protects the manufacturer from potential fines and legal issues but also enhances the brand reputation as a sustainable and responsible chemical producer.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation and benefits of this patented synthesis technology. These insights are derived directly from the experimental data and comparative analysis presented in the patent documentation, providing a clear understanding of the process capabilities. Understanding these details is essential for stakeholders evaluating the feasibility of integrating this intermediate into their existing supply chains or product formulations. The answers reflect the consensus of technical experts who have reviewed the efficacy of the solvent-free alkylation method against traditional benchmarks.
Q: What are the primary advantages of this new alkylation method over traditional cyclization?
A: The novel method eliminates the need for toxic solvents like o-dichlorobenzene and reduces reaction steps, resulting in yields exceeding 96% compared to roughly 71% for cyclization methods.
Q: How does the process ensure high selectivity without ultrasonic assistance?
A: By strictly controlling the reaction temperature between 10°C and 50°C and utilizing benzene as the solvent, the process achieves high triazine selectivity without the energy waste and noise pollution of ultrasonic devices.
Q: Is this synthesis route suitable for large-scale industrial production?
A: Yes, the process uses standard equipment (three-neck flasks with stirring and condensation) and avoids complex device modifications, making it highly scalable and easy to implement in existing facilities.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Chloro-4,6-diphenyl-1,3,5-triazine Supplier
As the demand for high-performance UV absorbers continues to grow across the polymer and coating industries, securing a partnership with a technically proficient manufacturer is more critical than ever. NINGBO INNO PHARMCHEM stands at the forefront of this sector, leveraging extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production to deliver consistent quality. Our commitment to excellence is underpinned by stringent purity specifications and rigorous QC labs that ensure every batch of 2-chloro-4,6-diphenyl-1,3,5-triazine meets the highest international standards. We understand that the integrity of your final product depends on the quality of the intermediates you source, which is why we invest heavily in advanced analytical capabilities and process optimization technologies. By choosing us as your partner, you gain access to a supply chain that is not only robust and reliable but also deeply attuned to the nuances of complex chemical synthesis.
We invite you to engage with our technical procurement team to discuss how our capabilities can support your specific project requirements. Whether you need a Customized Cost-Saving Analysis for your current sourcing strategy or require specific COA data to validate our product against your internal standards, we are ready to assist. Our team is prepared to provide detailed route feasibility assessments that can help you optimize your manufacturing processes and reduce time-to-market for your innovative products. Contact us today to explore how our advanced synthesis technologies can drive value and efficiency in your operations.
