Advanced Diazodifluoromethylation Technology for High-Purity Pharmaceutical Intermediate Manufacturing at Commercial Scale
Patent CN114634482A introduces a groundbreaking advancement in fluorination chemistry through the development of a novel diazodifluoromethyl reagent that enables efficient introduction of difluoromethyl groups into organic molecules under mild conditions without requiring hazardous gases or cryogenic temperatures typically associated with conventional methodologies. This innovation addresses critical limitations in traditional difluoromethylation approaches by operating at ambient temperature with simple workup procedures involving standard column chromatography purification while achieving yields ranging from sixty percent to eighty percent through a streamlined synthetic protocol that maintains exceptional functional group compatibility across diverse molecular frameworks encountered in pharmaceutical synthesis. The reagent's versatility extends across multiple therapeutic areas including cardiovascular drugs such as rosuvastatin calcium tablets and oncology targets like PI3K inhibitors where difluoromethyl moieties enhance binding affinity through weak hydrogen bonding interactions with biological receptors due to their unique electronic properties that improve drug-receptor binding kinetics essential for therapeutic efficacy.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional approaches for introducing difluoromethyl groups into organic molecules suffer from significant drawbacks including poor functional group tolerance and demanding reaction conditions that hinder their industrial applicability across diverse molecular architectures commonly encountered in pharmaceutical manufacturing environments where complex structural elements must remain intact throughout synthesis sequences. Methods utilizing HCF₂Cl₂ gas present serious safety hazards due to its toxicity and volatility while requiring specialized handling equipment that increases capital expenditure and operational complexity through additional safety protocols and containment systems necessary for regulatory compliance in GMP facilities worldwide. Alternative techniques involving low temperatures down to minus forty degrees Celsius necessitate expensive cryogenic infrastructure and energy-intensive cooling systems that substantially elevate production costs while limiting scalability potential due to thermal management challenges during scale-up operations from laboratory to commercial production volumes.
The Novel Approach
The patented methodology overcomes these challenges through an innovative one-step synthesis that operates under ambient air conditions without requiring specialized equipment or hazardous materials by employing commercially available hypervalent iodine reagents combined with trimethylsilyl trifluoromethanesulfonic ketone and synthetic substrate Ps-DFA at room temperature for three to five hours achieving yields between sixty percent and eighty percent with exceptional functional group compatibility across diverse molecular frameworks encountered in complex pharmaceutical intermediate synthesis where sensitive structural elements must be preserved throughout processing sequences.
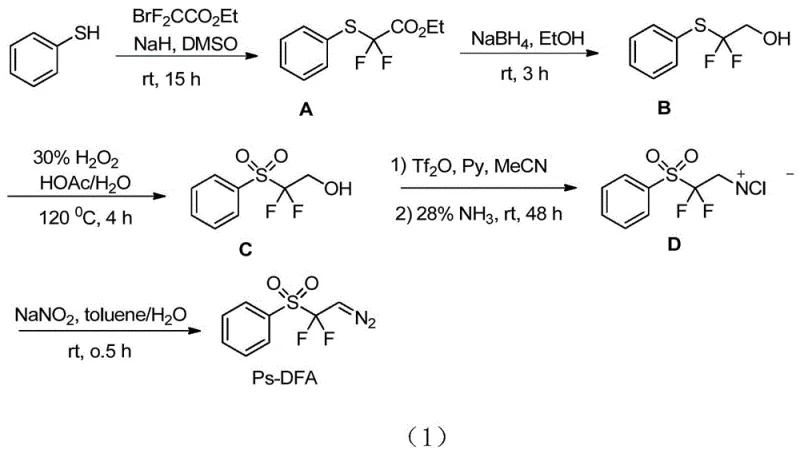
Mechanistic Insights into Diazo Difluoromethylation
The reaction mechanism proceeds through a concerted process where hypervalent iodine species activate trimethylsilyl trifluoromethanesulfonic ketone to generate an electrophilic intermediate facilitating nucleophilic attack by Ps-DFA's diazo component without forming radical intermediates typically associated with difluoromethylation chemistry thereby preventing undesired side reactions that compromise product purity in conventional approaches requiring transition metal catalysts which introduce contamination risks necessitating additional purification steps that increase production costs while reducing overall yield efficiency across multiple processing stages required for pharmaceutical intermediate manufacturing.
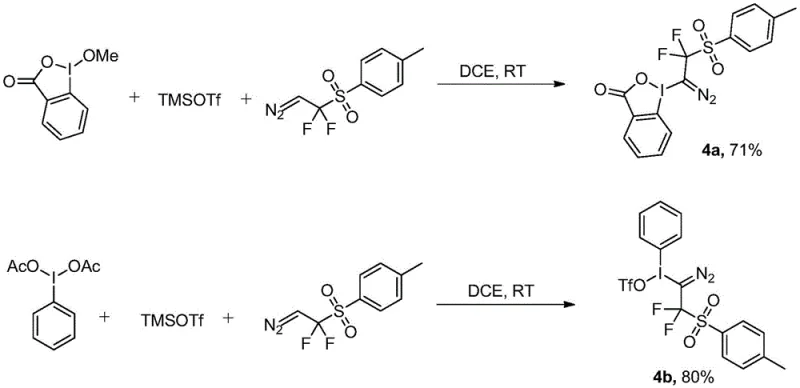
The process exhibits excellent impurity control characteristics due to its high selectivity and minimal byproduct formation during transformation from starting materials to final product where absence of transition metals eliminates potential catalytic impurities while room temperature operation prevents thermal degradation pathways generating unwanted side products common in traditional high-energy processes requiring cryogenic conditions; column chromatography purification using petroleum ether/ethyl acetate mixtures effectively separates minor impurities without specialized techniques like preparative HPLC ensuring consistent high-purity material meeting pharmaceutical industry standards without additional processing steps compromising yield during scale-up operations from laboratory validation to commercial production volumes.
How to Synthesize Diazodifluoromethyl Reagent Efficiently
This innovative synthetic route represents a significant advancement over conventional difluoromethylation methodologies by providing a streamlined pathway operating under ambient conditions without specialized equipment or hazardous materials leveraging commercially available starting materials and standard laboratory techniques to produce high-purity diazodifluoromethyl reagents suitable for pharmaceutical intermediate manufacturing applications where stringent quality specifications are mandatory; detailed standardized operating procedures have been developed based on extensive process optimization studies ensuring consistent results across different production scales while maintaining excellent yield and purity profiles; the following step-by-step guide provides essential technical parameters for successful implementation of this technology in industrial settings.
- Combine commercially available hypervalent iodine reagent such as iodobenzene acetate or methoxybenziodoxolone with trimethylsilyl trifluoromethanesulfonic ketone and synthetic substrate Ps-DFA in a molar ratio of one-to-one-to-two point five using dichloromethane or acetonitrile as solvent.
- Stir the reaction mixture at room temperature under ambient air conditions for three to five hours to achieve complete conversion without requiring cryogenic equipment or hazardous gas handling.
- Purify the resulting crude product through standard column chromatography using petroleum ether and ethyl acetate as eluent to isolate pure diazodifluoromethyl reagent derivatives meeting pharmaceutical purity standards.
Commercial Advantages for Procurement and Supply Chain Teams
This technology delivers substantial value across procurement and supply chain functions by addressing critical pain points associated with traditional difluoromethylation processes while enhancing overall manufacturing efficiency through elimination of hazardous gas handling requirements reducing safety compliance burdens and associated operational costs while simplifying facility requirements globally; utilization of commercially available stable starting materials ensures consistent supply availability avoiding dependencies on specialized gas suppliers; robust room temperature processing demonstrates excellent reproducibility across different manufacturing sites without requiring specialized equipment calibration beyond standard organic synthesis protocols significantly lowering energy consumption profiles compared to conventional approaches needing extensive cooling infrastructure during both reaction execution and intermediate storage phases.
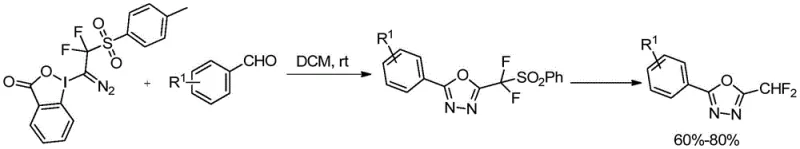
- Cost Reduction in Manufacturing: Elimination of transition metal catalysts removes expensive purification steps required to eliminate metal residues from final products while avoiding costly cryogenic equipment investments; simplified workup procedures using standard column chromatography instead of specialized techniques substantially reduce processing time without compromising quality standards essential for pharmaceutical applications where purity specifications must be strictly maintained throughout production sequences.
- Enhanced Supply Chain Reliability: Utilization of commercially available starting materials with stable shelf lives ensures consistent supply availability eliminating dependence on specialized gas suppliers; robust room temperature process demonstrates excellent reproducibility across different manufacturing sites without requiring specialized equipment calibration or operator training beyond standard organic synthesis protocols maintaining consistent quality profiles regardless of production location.
- Scalability and Environmental Compliance: Straightforward scale-up pathway maintains consistent yield and purity profiles from laboratory validation to commercial production volumes while generating minimal waste streams; elimination of hazardous gases and transition metals significantly improves environmental footprint compared to conventional methods meeting increasingly stringent regulatory requirements for sustainable manufacturing practices in pharmaceutical industry supply chains worldwide.
Frequently Asked Questions (FAQ)
The following questions address common technical concerns regarding implementation based on extensive process development experience across multiple production scales; these insights reflect practical considerations encountered during scale-up activities while maintaining adherence to regulatory requirements for pharmaceutical intermediate manufacturing processes where consistent quality profiles must be maintained throughout all production phases regardless of batch size or manufacturing location.
Q: What are the key advantages over conventional difluoromethylation methods?
A: The patented process eliminates hazardous HCF₂Cl₂ gas requirements and cryogenic conditions while operating at room temperature with simplified workup procedures that significantly reduce manufacturing complexity without compromising product purity essential for pharmaceutical applications.
Q: How does this technology improve supply chain reliability?
A: Utilization of commercially available stable starting materials combined with robust room temperature processing ensures consistent supply availability while avoiding specialized equipment dependencies that could disrupt production continuity across global manufacturing sites.
Q: Can this methodology support large-scale pharmaceutical intermediate production?
A: Yes, the straightforward scale-up pathway maintains consistent yield and purity profiles from laboratory to commercial production scales while generating minimal waste streams through its inherently selective reaction mechanism.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Diazodifluoromethyl Reagent Supplier
Our company possesses extensive experience scaling diverse pathways from one hundred kilograms to one hundred metric tons annual commercial production while maintaining stringent purity specifications through rigorous QC labs equipped with state-of-the-art analytical instrumentation capable of detecting impurities at parts-per-billion levels; as a leading CDMO specialist in complex fluorination chemistry we have successfully implemented similar innovative methodologies across multiple therapeutic areas including cardiovascular drugs where precise difluoromethyl group incorporation proved critical for achieving desired pharmacological profiles while meeting regulatory approval requirements worldwide through robust quality management systems validated across global manufacturing networks.
We invite you to request a Customized Cost-Saving Analysis from our technical procurement team who will provide specific COA data and route feasibility assessments tailored to your unique manufacturing requirements; our experts stand ready to collaborate on optimizing this technology for your specific API intermediate production needs while ensuring seamless integration into existing supply chain frameworks maintaining consistent quality profiles throughout all production phases.
