Advanced Solvent-Free Manufacturing of High-Purity Disubstituted p-Nitrophenylacetonitrile Derivatives for Commercial Scale-Up in Pharma
Patent CN109694335B introduces a groundbreaking solvent-free methodology for synthesizing disubstituted p-nitrophenylacetonitrile derivatives addressing critical limitations in traditional synthetic routes that rely on organic solvents such as alcohols or dimethyl sulfoxide which are typically required as both reaction media and co-catalysts creating substantial economic burdens through procurement recovery and disposal expenses while generating hazardous waste streams conflicting with modern environmental regulations The patent demonstrates an integrated tandem reaction sequence combining Knoevenagel condensation reduction and alkylation steps under solvent-free conditions at temperatures ranging from 80 to 100°C substantially shortening overall reaction time compared to existing methods requiring up to twenty-four hours as documented in Tetrahedron Letters This innovative approach leverages dihydropyridine esters as a non-toxic hydrogen source with high chemoselectivity achieving superior yields exceeding eighty-five percent for most aromatic aldehyde substrates while maintaining excellent functional group tolerance across electron-donating and electron-withdrawing substituents The elimination of solvent usage not only reduces raw material costs but also simplifies downstream purification through column chromatography using petroleum ether and ethyl acetate mixtures making it an ideal solution for pharmaceutical intermediate manufacturing where purity cost-efficiency and regulatory compliance are paramount considerations for global supply chains
The Limitations of Conventional Methods vs The Novel Approach
The Limitations of Conventional Methods
Traditional synthesis routes for mono-substituted p-nitrophenylacetonitrile derivatives necessitate organic solvents like alcohols or dimethyl sulfoxide creating significant economic burdens through procurement recovery disposal expenses while generating hazardous waste streams that conflict with environmental regulations These methods require extended reaction times up to twenty-four hours leading to reduced throughput higher operational costs and increased risk of impurity formation during multi-step isolation procedures The reliance on transition metal catalysts in alternative routes introduces additional challenges related to metal contamination necessitating costly removal steps before products meet regulatory standards for drug substance manufacturing Furthermore conventional approaches suffer from poor atom economy due to intermediate purification requirements which increase material loss potential side reactions and overall process inefficiency particularly problematic when scaling up sensitive pharmaceutical intermediates where batch consistency is critical These cumulative drawbacks—solvent dependency prolonged reaction durations complex purification requirements and potential metal contamination—create substantial barriers to cost-effective sustainable production of these valuable chemical building blocks essential for active pharmaceutical ingredient synthesis
The Novel Approach
The patented method described in CN109694335B overcomes these limitations through an elegant solvent-free tandem reaction system integrating Knoevenagel condensation reduction and alkylation steps into a single streamlined process without intermediate isolation By utilizing dihydropyridine esters as a non-toxic hydrogen source with excellent chemoselectivity under mild thermal conditions eighty to one hundred degrees Celsius this approach eliminates organic solvents entirely while achieving reaction completion within six to nine hours compared to conventional twenty-four-hour timeframe The process employs readily available bases like potassium carbonate or DIEA at optimized molar ratios ensuring high conversion efficiency across diverse aromatic aldehydes including those with electron-donating or electron-withdrawing groups Crucially eliminating solvent usage reduces raw material costs simplifies waste management by avoiding hazardous streams while direct progression from condensation to alkylation minimizes impurity formation pathways This innovative strategy represents a paradigm shift toward sustainable manufacturing practices maintaining high yields typically above eighty-five percent while significantly improving process economics through reduced cycle times lower energy consumption and enhanced operational flexibility essential for modern pharmaceutical supply chains
Mechanistic Insights into Solvent-Free Tandem Synthesis
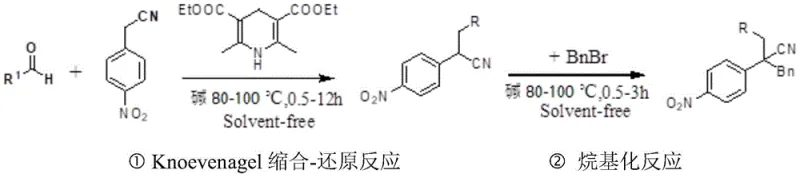
The core innovation lies in seamless integration of Knoevenagel condensation reduction into a single tandem step followed by direct alkylation under solvent-free conditions Initially p-nitrophenylacetonitrile reacts with aromatic aldehydes via base-catalyzed Knoevenagel condensation forming an αβ-unsaturated nitrile intermediate which undergoes immediate reduction by dihydropyridine esters acting as hydride source without isolation generating reduced adduct serving as nucleophile for subsequent alkylation The dihydropyridine ester functions through concerted hydride transfer maintaining high chemoselectivity toward nitroalkene functionality while avoiding over-reduction side reactions common with traditional hydrogenation methods The absence of solvent creates unique microenvironment where elevated reactant concentrations accelerate both condensation reduction steps minimizing decomposition pathways typical in diluted solution-phase reactions This concentration effect also facilitates alkylation where reduced intermediate undergoes nucleophilic substitution with benzyl bromide under basic conditions forming final disubstituted product with excellent regioselectivity while preventing competitive hydrolysis reactions observed in aqueous systems
Impurity control is inherently optimized through integrated approach as elimination of intermediate isolation prevents atmospheric contamination exposure reducing opportunities for side reactions during workup procedures Mild thermal conditions eighty to one hundred degrees Celsius prevent thermal degradation of sensitive functional groups while precise stoichiometric control of bases between zero point two to one point five equivalents ensures complete conversion without excess reagent residues complicating purification Column chromatography using petroleum etherethyl acetate mixtures effectively separates desired product from minor byproducts formed during tandem sequence with gradient system resolving structurally similar impurities from incomplete alkylation or residual starting materials This comprehensive impurity management strategy consistently delivers products meeting stringent pharmaceutical purity requirements above ninety-eight percent by HPLC as evidenced by high yields typically exceeding eighty-five percent across diverse substrate combinations without additional polishing steps ensuring reliable supply chain continuity for critical drug substance manufacturing
How to Synthesize Disubstituted p-Nitrophenylacetonitrile Derivative Efficiently
This patented methodology represents significant advancement in disubstituted p-nitrophenylacetonitrile derivative synthesis through innovative solvent-free tandem approach integrating multiple synthetic steps into single streamlined process eliminating traditional solvent requirements while reducing overall reaction time from approximately twenty-four hours in conventional methods to just six to nine hours under optimized conditions Breakthrough lies in ability to eliminate organic solvents while simultaneously improving yield consistency across diverse aromatic aldehyde substrates with varying electronic properties Detailed standardized synthesis procedures have been developed based on extensive experimental validation ensuring robust performance regardless of substituent effects on aromatic ring following section provides comprehensive step-by-step guide implementing this efficient manufacturing process industrial settings
- Perform Knoevenagel condensation-reduction tandem reaction by mixing p-nitrophenylacetonitrile, aromatic aldehyde, base, and dihydropyridine ester at molar ratio 1: (1-2):(0.2-1.5):(1-2) under solvent-free conditions at 80-100°C for 0.5-12 hours.
- Conduct alkylation reaction by adding base and benzyl bromide to the product from step one at molar ratio of p-nitrophenylacetonitrile to base to BnBr as 1: (1-4):(1-4), heating to 80-100°C for 0.5-3 hours.
- Purify the final product using column chromatography with silica gel and petroleum ether/ethyl acetate mixture (80: 1 to 15:1 v/v) as mobile phase.
Commercial Advantages for Procurement and Supply Chain Teams
This innovative manufacturing process directly addresses critical pain points faced by procurement supply chain professionals in pharmaceutical industry delivering substantial operational improvements without significant capital investment Process eliminates organic solvents creating immediate cost savings through reduced raw material expenditures while lowering hazardous waste disposal costs associated with specialized treatment requirements Furthermore simplified reaction sequence enhances supply chain reliability by reducing dependency on multiple external suppliers for solvents catalysts improving production flexibility through shorter cycle times enabling faster response fluctuating demand patterns The elimination of transition metal catalysts avoids costly purification steps required for metal residue removal streamlining quality control processes essential for regulatory compliance across global markets
- Cost Reduction in Manufacturing: Complete removal of organic solvents significantly reduces raw material costs eliminating associated expenses for recovery systems hazardous waste disposal procedures while avoiding expensive transition metal catalysts requiring additional purification steps resulting in substantial cost savings throughout production lifecycle without compromising quality yield consistency Essential cost optimization aligns perfectly with procurement objectives seeking sustainable manufacturing solutions that deliver economic value without sacrificing performance metrics required by R&D teams
- Enhanced Supply Chain Reliability: Utilization of readily available starting materials like aromatic aldehydes benzyl bromide with simplified reaction conditions not requiring specialized equipment ensures greater supply chain resilience against raw material shortages logistical disruptions Elimination of solvent dependency removes vulnerability fluctuations solvent markets enabling more consistent production scheduling due shorter reaction times simplified workflow management Critical reliability improvement addresses procurement managers' primary concerns about supply continuity particularly when sourcing complex intermediates requiring multi-step syntheses under traditional approaches
- Scalability and Environmental Compliance: Mild thermal conditions eighty to one hundred degrees Celsius straightforward process design facilitate seamless scale-up from laboratory commercial volumes without specialized reactors safety modifications typically needed high-pressure cryogenic processes Environmentally friendly approach aligns global sustainability initiatives minimizing hazardous waste generation reducing carbon footprint through energy-efficient operation moderate temperatures Supporting procurement teams' ESG objectives while meeting stringent environmental regulations across major markets including EU US Japan ensuring long-term supply chain viability
Frequently Asked Questions (FAQ)
The following questions address common technical commercial concerns regarding implementation patented solvent-free synthesis method disubstituted p-nitrophenylacetonitrile derivatives pharmaceutical manufacturing operations Answers derived directly experimental data process validation studies documented patent CN109694335B providing actionable insights procurement R&D teams evaluating commercial viability this innovative technology
Q: How does the solvent-free method enhance environmental sustainability?
A: The elimination of organic solvents in this patented process significantly reduces hazardous waste generation and disposal costs while aligning with green chemistry principles through minimized resource consumption without compromising reaction efficiency or product quality.
Q: What are the purity advantages for pharmaceutical applications?
A: The tandem reaction mechanism minimizes intermediate isolation steps reducing potential impurity introduction pathways while maintaining stringent purity specifications required for pharmaceutical intermediates through optimized stoichiometric control and mild thermal conditions.
Q: How does this process support commercial scale-up?
A: The moderate temperature requirements (80–100°C) and simplified workflow enable seamless transition from laboratory to industrial scale without specialized equipment ensuring reliable supply chain continuity through robust performance across diverse substrate combinations.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Disubstituted p-Nitrophenylacetonitrile Derivative Supplier
This patented technology represents significant advancement sustainable manufacturing practices critical pharmaceutical intermediates offering environmental benefits economic advantages through innovative solvent-free approach NINGBO INNO PHARMCHEM brings extensive experience scaling diverse pathways from one hundred kgs to one hundred MT annual commercial production while maintaining stringent purity specifications required global regulatory authorities Our rigorous QC labs employ advanced analytical techniques including HPLC NMR spectroscopy ensuring consistent product quality across all production scales providing pharmaceutical manufacturers reliable access high-purity intermediates essential drug substance development Supply chain teams benefit from our proven track record delivering complex molecules on time meeting exacting quality standards demanded modern pharmaceutical industry
We invite you request Customized Cost-Saving Analysis tailored specific manufacturing requirements contacting our technical procurement team today They will provide detailed route feasibility assessments along specific COA data demonstrating how this innovative process optimize supply chain meeting all regulatory compliance standards essential procurement decision-making processes
