Advanced Metal-Free Synthesis of Quinoline Oxides for High-Purity Pharmaceutical Intermediates
The landscape of organic synthesis for heterocyclic compounds is undergoing a significant transformation, driven by the urgent need for greener, more cost-effective methodologies that do not compromise on purity or yield. A pivotal advancement in this domain is detailed in patent CN110156683B, which introduces a robust, metal-free synthetic route for producing quinoline oxides. These compounds are critical scaffolds in medicinal chemistry, often serving as key intermediates for developing biologically active agents with diverse therapeutic potentials. The traditional reliance on precious metal catalysts has long been a bottleneck for both R&D budgets and supply chain stability, but this new approach leverages the oxidative power of sodium persulfate to achieve exceptional results. By shifting away from complex catalytic systems, this technology offers a streamlined pathway that aligns perfectly with modern principles of sustainable chemistry and industrial efficiency. For procurement leaders and technical directors alike, understanding the nuances of this innovation is essential for securing a competitive edge in the global market for high-purity pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the preparation of quinoline oxides has relied heavily on protocols that necessitate the use of expensive transition metal catalysts, often in conjunction with harsh oxidizing agents and elevated temperatures. As noted in prior art such as Adv. Synth. Catal., 2019, 361, these conventional routes typically involve reacting quinoline 1-oxides with hydrazine derivatives in the presence of metallic species that are not only costly but also pose significant environmental and safety hazards. The presence of residual metals in the final product is a major concern for pharmaceutical applications, requiring rigorous and expensive purification steps to meet stringent regulatory standards for heavy metal content. Furthermore, the sensitivity of these metal-catalyzed reactions often demands inert atmospheres and anhydrous conditions, adding layers of operational complexity and equipment costs that hinder scalability. The unsatisfactory yields frequently observed in these older methods further exacerbate the economic inefficiency, leading to higher waste generation and increased overall production costs for fine chemical manufacturers.
The Novel Approach
In stark contrast, the methodology disclosed in CN110156683B presents a paradigm shift by completely eliminating the need for metal catalysts, relying instead on the potent oxidative capability of sodium persulfate. This novel approach utilizes readily available quinoline 1-oxide compounds and semicarbazide derivatives as starting materials, which react efficiently under mild thermal conditions ranging from 80°C to 100°C. The simplicity of the reaction system is remarkable; it proceeds smoothly under ambient air conditions without the need for specialized inert gas protection, drastically reducing operational overheads. The use of common solvents such as water, acetonitrile, or DMSO further enhances the practicality of this method, offering flexibility in process optimization. By achieving high yields through a direct oxidative coupling mechanism, this technology provides an optimal alternative solution that addresses the economic and environmental drawbacks of previous techniques, making it an ideal candidate for reliable agrochemical intermediate supplier networks and pharmaceutical supply chains alike.
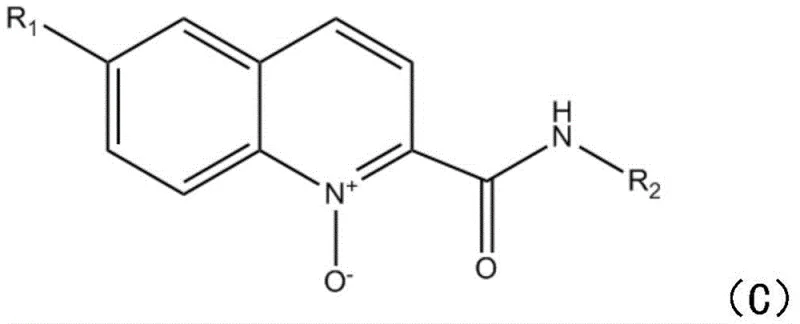
Mechanistic Insights into Sodium Persulfate-Mediated Radical Oxidation
The core of this technological breakthrough lies in the unique radical mechanism facilitated by sodium persulfate, which acts as a single-electron oxidant to generate reactive acyl radicals from the semicarbazide precursors. Upon thermal activation, the persulfate anion undergoes homolytic cleavage to produce sulfate radical anions, which then abstract hydrogen atoms or interact with the semicarbazide to form the crucial acyl radical species. These electrophilic radicals subsequently attack the electron-deficient positions of the quinoline 1-oxide ring system, initiating a cascade of cyclization and rearrangement steps that ultimately lead to the formation of the stable quinoline oxide framework. This radical pathway is highly selective, minimizing the formation of unwanted by-products that typically plague metal-catalyzed cross-coupling reactions. The absence of metal coordination complexes simplifies the electronic landscape of the reaction, allowing for a cleaner transformation that is easier to control and monitor. Understanding this mechanism is vital for R&D teams aiming to replicate or modify the process for specific substrate variations, as it highlights the importance of maintaining precise stoichiometric ratios to ensure complete radical generation and consumption.
From an impurity control perspective, the metal-free nature of this synthesis offers distinct advantages in managing the杂质 profile of the final API intermediate. Without the introduction of transition metals, the risk of metal-catalyzed side reactions, such as over-oxidation or polymerization, is significantly reduced, leading to a cleaner crude reaction mixture. The primary impurities are likely to be unreacted starting materials or simple hydrolysis products, which are generally easier to separate via standard chromatographic techniques compared to metal-ligand complexes. The patent specifies the use of silica gel column chromatography with ethyl acetate and petroleum ether gradients, a standard purification method that is easily scalable to preparative HPLC or crystallization processes in a manufacturing setting. This predictability in impurity behavior allows quality control laboratories to establish robust specifications more rapidly, accelerating the timeline from process development to commercial validation. Consequently, the mechanistic clarity translates directly into enhanced process reliability and reduced risk of batch failures during scale-up operations.
How to Synthesize 2-Carbamoylquinoline 1-oxide Efficiently
Implementing this synthesis in a laboratory or pilot plant setting requires careful attention to the stoichiometric balance and thermal parameters defined in the patent to maximize efficiency. The process begins with the precise weighing of quinoline 1-oxide derivatives and semicarbazide compounds, which are then introduced into a reaction vessel along with two equivalents of sodium persulfate relative to the substrate. The choice of solvent—whether water for green chemistry applications or acetonitrile/DMSO for solubility reasons—should be dictated by the specific substitution pattern of the starting materials to ensure a homogeneous reaction mixture. Once the reagents are combined, the system is sealed and heated to a temperature between 80°C and 100°C, where it is maintained for a period ranging from 2 to 10 hours depending on the reactivity of the specific substrates involved. Detailed standardized synthesis steps see the guide below.
- Combine quinoline 1-oxide compounds and semicarbazide compounds in a reactor with sodium persulfate and a suitable solvent such as water, acetonitrile, or DMSO.
- Seal the reactor and maintain the reaction mixture at a temperature between 80°C and 100°C under air conditions for a duration of 2 to 10 hours to ensure complete conversion.
- Upon completion, purify the crude reaction mixture using silica gel column chromatography with an ethyl acetate and petroleum ether gradient to isolate the target quinoline oxide product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this metal-free synthesis route represents a strategic opportunity to optimize cost structures and enhance supply security. The elimination of precious metal catalysts removes a volatile cost component from the bill of materials, as prices for metals like palladium or copper can fluctuate wildly based on geopolitical factors and mining outputs. Furthermore, the reliance on sodium persulfate, a commodity chemical produced in massive quantities for various industries, ensures a stable and abundant supply of the key oxidant, mitigating the risk of raw material shortages that could disrupt production schedules. The simplified reaction conditions, which do not require inert atmospheres or specialized high-pressure equipment, lower the barrier to entry for contract manufacturing organizations, thereby expanding the pool of potential suppliers and fostering a more competitive sourcing environment. These factors collectively contribute to a more resilient supply chain capable of withstanding market shocks while maintaining consistent delivery performance for critical pharmaceutical intermediates.
- Cost Reduction in Manufacturing: The most immediate financial benefit stems from the complete removal of expensive transition metal catalysts, which traditionally account for a significant portion of raw material costs in heterocyclic synthesis. By substituting these with inexpensive sodium persulfate, the direct material cost is drastically simplified, allowing for substantial cost savings that can be passed down the value chain or reinvested into R&D. Additionally, the downstream processing costs are reduced because there is no need for specialized metal scavenging resins or extensive washing protocols to remove trace metal contaminants, which streamlines the purification workflow. This economic efficiency makes the process highly attractive for cost reduction in pharmaceutical intermediates manufacturing, particularly for high-volume products where margin compression is a constant challenge.
- Enhanced Supply Chain Reliability: The use of widely available, non-hazardous reagents like sodium persulfate and common organic solvents ensures that the supply chain is not dependent on niche vendors or restricted chemicals. This ubiquity of raw materials means that procurement teams can source inputs from multiple geographic regions, reducing the risk of supply disruptions caused by local regulatory changes or logistical bottlenecks. The robustness of the reaction conditions, which tolerate air and moisture better than sensitive metal-catalyzed systems, also reduces the likelihood of batch failures due to environmental factors, ensuring a more predictable output volume. This reliability is crucial for reducing lead time for high-purity pharmaceutical intermediates, enabling manufacturers to respond more agilely to fluctuating market demands.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, this method offers a cleaner profile that aligns with increasingly strict global regulations on industrial emissions and waste disposal. The absence of heavy metals simplifies wastewater treatment processes, as there is no need for complex precipitation or ion-exchange steps to meet discharge limits for toxic metals. The reaction's ability to proceed in water or benign solvents further supports green chemistry initiatives, potentially qualifying the process for environmental incentives or preferred supplier status with eco-conscious clients. The commercial scale-up of complex pharmaceutical intermediates is thus facilitated by a process that is not only chemically efficient but also environmentally sustainable, future-proofing the manufacturing operation against tightening regulatory frameworks.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel synthesis technology, based on the detailed specifications provided in the patent documentation. These insights are designed to clarify the operational feasibility and strategic benefits for stakeholders evaluating this method for their production pipelines. Understanding these details is key to making informed decisions about process adoption and supplier qualification.
Q: Does this synthesis method require expensive transition metal catalysts?
A: No, the patented method (CN110156683B) specifically eliminates the need for expensive metal catalysts, utilizing cheap and readily available sodium persulfate as the sole oxidant, which significantly reduces raw material costs and simplifies downstream purification.
Q: What yields can be expected from this metal-free protocol?
A: The process demonstrates excellent efficiency, with reported isolated yields reaching as high as 97% for various substrates, including those with chloro, fluoro, and methyl substituents, ensuring high material throughput for production.
Q: Is this process suitable for large-scale commercial manufacturing?
A: Yes, the reaction operates under mild conditions (80-100°C) in air without stringent inert atmosphere requirements, and uses common solvents like water or acetonitrile, making it highly amenable to scale-up from laboratory to industrial tonnage production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Quinoline Oxides Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this metal-free synthesis technology and are fully equipped to leverage it for your specific project needs. As a seasoned CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless and efficient. Our state-of-the-art facilities are designed to handle the specific thermal and safety requirements of oxidative reactions, while our rigorous QC labs enforce stringent purity specifications to guarantee that every batch meets the highest international standards. We understand that consistency is paramount in the pharmaceutical industry, and our commitment to quality assurance ensures that the quinoline oxides we deliver are free from the impurities that often plague less optimized synthetic routes.
We invite you to engage with our technical procurement team to discuss how this innovative process can be tailored to your specific molecular targets. By requesting a Customized Cost-Saving Analysis, you can gain a clear understanding of the economic advantages this method offers over your current supply chain. We encourage you to reach out for specific COA data and route feasibility assessments, allowing us to demonstrate concretely how our expertise in advanced organic synthesis can drive value and efficiency for your organization. Let us be your partner in navigating the complexities of modern chemical manufacturing.
