Advanced Synthesis of 2-Mercapto-1-Alkyl Imidazole: A Breakthrough in Safety and Scalability for Electronic Intermediates
The landscape of fine chemical manufacturing is constantly evolving, driven by the dual demands of higher purity standards and stricter safety protocols, particularly within the electronic chemicals sector. Patent CN107522661B, published in early 2020, introduces a transformative preparation method for 2-mercapto-1-alkyl imidazole, a critical intermediate valued for its unique heterocyclic structure and reactivity. This innovation addresses long-standing inefficiencies in heterocyclic synthesis by replacing hazardous reagents with a more controlled, environmentally benign pathway. For R&D directors and procurement specialists alike, this patent represents a pivotal shift away from toxic thiocyanate routes toward a safer, lithiation-based strategy that ensures consistent product quality. The methodology not only enhances the reproducibility of the synthesis but also aligns perfectly with the rigorous specifications required for semiconductor and pharmaceutical applications. By leveraging this advanced chemistry, manufacturers can secure a more reliable supply chain for high-value azacycle derivatives.
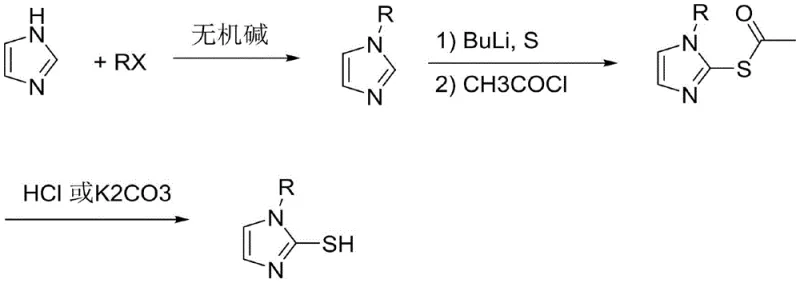
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of mercaptoimidazoles has been plagued by significant safety and environmental drawbacks that complicate large-scale manufacturing. One prevalent conventional method involves the reaction of halogenated acetaldehyde dimethyl acetal with substituted primary amines, followed by treatment with potassium thiocyanate. This route is fundamentally flawed due to the generation of hydrogen thiocyanate (HSCN), a highly toxic by-product that decomposes during the process, creating severe handling hazards and requiring complex waste treatment infrastructure. Furthermore, alternative literature methods utilizing N-alkyl imidazole and n-butyl lithium often employ excessive amounts of sulfur powder, typically greater than one equivalent. This surplus of elemental sulfur creates a precarious environment where spontaneous combustion and oxidation are genuine risks, leading to uneven reaction yields and potential safety incidents in the plant. These legacy processes fail to meet modern green chemistry standards, imposing hidden costs related to safety management and regulatory compliance that erode profit margins.
The Novel Approach
In stark contrast, the novel approach detailed in the patent data revolutionizes the synthesis by introducing a precise, three-step sequence that prioritizes safety and efficiency without compromising yield. The process begins with a robust alkylation of imidazole using alkyl halides in the presence of inorganic bases, achieving conversion rates exceeding 95 percent when optimized bases like cesium carbonate are selected. Crucially, the subsequent lithiation step employs a strictly controlled stoichiometric ratio of sulfur powder, specifically between 0.9 and 0.95 equivalents, which effectively eliminates the danger of excess sulfur accumulation. The unstable thiolate intermediate is immediately captured by acetyl chloride to form a stable 2-acetylthio-1-alkyl imidazole, preventing degradation and ensuring high reproducibility. This strategic modification not only mitigates the risk of spontaneous combustion but also streamlines the purification process, resulting in a final product with exceptional purity suitable for demanding electronic applications.
Mechanistic Insights into Lithiation-Thiolation and Acetyl Protection
The core of this synthetic breakthrough lies in the meticulous control of the lithiation-thiolation mechanism, which dictates both the safety profile and the chemical yield of the final product. The process initiates by dissolving the 1-alkyl imidazole precursor in an ether solvent, such as tetrahydrofuran or 2-methyltetrahydrofuran, and cooling the mixture to cryogenic temperatures around -15°C. At this stage, the addition of 1 to 1.1 equivalents of n-butyl lithium facilitates the selective deprotonation at the C2 position of the imidazole ring, generating a highly reactive nucleophilic species. Unlike traditional methods that might allow this intermediate to react indiscriminately, the patented protocol introduces a sub-stoichiometric amount of elemental sulfur (0.9-0.95 equivalents) under reflux conditions. This precise limitation ensures that the sulfur is fully consumed in the formation of the thiolate anion, leaving no residual oxidant to cause side reactions or safety hazards. The immediate follow-up addition of acetyl chloride serves as a critical trapping agent, converting the sensitive thiolate into a robust thioester derivative that can be isolated and stored safely before the final deprotection step.
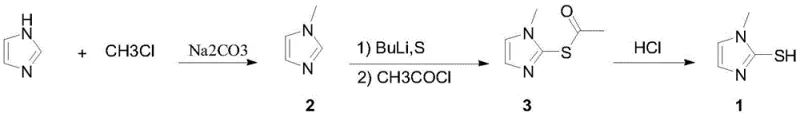
Furthermore, the versatility of this mechanism is demonstrated by its compatibility with various alkyl substituents, ranging from simple methyl and ethyl groups to more sterically hindered cyclopropyl rings. For instance, when synthesizing 2-mercapto-1-cyclopropylimidazole, the use of cesium carbonate in the initial alkylation step proves essential, driving conversion rates above 95 percent where other bases like sodium carbonate fail significantly. The subsequent lithiation and sulfur insertion proceed smoothly, maintaining the integrity of the cyclopropyl ring despite the harsh basic conditions. The final deprotection step, whether catalyzed by hydrogen chloride gas or anhydrous potassium carbonate in an alcohol solvent, cleanly removes the acetyl protecting group to reveal the free thiol functionality. This mechanistic robustness ensures that impurity profiles remain minimal, with gas chromatography data consistently showing purities above 98.5 percent, a critical metric for customers in the semiconductor industry who cannot tolerate trace metal or organic contaminants.
How to Synthesize 2-Mercapto-1-Alkyl Imidazole Efficiently
Implementing this synthesis route requires careful attention to reaction conditions, particularly temperature control and reagent stoichiometry, to maximize the benefits of the patented method. The process is designed to be operationally simple yet chemically precise, allowing for seamless transition from laboratory benchtop to pilot plant scales. Operators must ensure that the lithiation step is conducted under inert atmosphere conditions to prevent moisture interference, while the subsequent sulfur addition requires vigilant monitoring to maintain the optimal 0.9-0.95 equivalent ratio. The detailed standardized synthesis steps below outline the exact parameters for temperature, solvent selection, and workup procedures necessary to achieve the reported high yields and purity levels. Adhering to these guidelines ensures that the safety advantages of avoiding excess sulfur are fully realized while maintaining the economic efficiency of the process.
- Alkylation of imidazole with alkyl halides using inorganic bases like cesium carbonate to form 1-alkyl imidazole.
- Low-temperature lithiation followed by reaction with controlled sulfur equivalents and immediate acetyl chloride capture.
- Deprotection of the acetyl group using catalytic HCl or potassium carbonate in alcohol solvents to yield the final thiol product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented synthesis route offers profound strategic advantages that extend far beyond simple chemical transformation. The elimination of toxic by-products like HSCN drastically simplifies the waste treatment workflow, reducing the burden on environmental compliance teams and lowering the overall cost of disposal. By removing the need for expensive and hazardous thiocyanate reagents, the raw material costs are significantly reduced, allowing for more competitive pricing structures in the final market. Moreover, the enhanced safety profile, characterized by the avoidance of spontaneous combustion risks associated with excess sulfur powder, translates directly into lower insurance premiums and reduced downtime for safety inspections. This reliability ensures a continuous supply of critical intermediates, mitigating the risk of production stoppages that can plague less stable chemical processes.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven by the substitution of high-cost, hazardous reagents with inexpensive, commodity-grade inorganic bases and alkyl halides. By optimizing the sulfur usage to sub-stoichiometric levels, the process minimizes raw material waste and eliminates the need for complex scavenging steps to remove excess sulfur residues. This streamlined approach reduces the consumption of solvents and energy during the workup phase, leading to substantial operational expenditure savings. Additionally, the high yield and reproducibility of the reaction mean that fewer batches are rejected due to quality failures, further enhancing the overall cost-efficiency of the manufacturing campaign.
- Enhanced Supply Chain Reliability: The robustness of the synthesis pathway ensures a stable and predictable supply of 2-mercapto-1-alkyl imidazole derivatives, which is crucial for maintaining uninterrupted production lines in downstream industries. The use of readily available starting materials such as imidazole and common alkyl halides reduces dependency on niche suppliers who might face their own supply constraints. Furthermore, the scalability of the process, demonstrated by successful runs in high-pressure reactors, guarantees that volume demands can be met without compromising on quality or lead times. This reliability makes the supplier a preferred partner for long-term contracts where consistency is paramount.
- Scalability and Environmental Compliance: From an environmental perspective, this method represents a significant leap forward in green chemistry, aligning with global trends towards sustainable manufacturing practices. The absence of heavy metal catalysts and toxic gas evolution simplifies the permitting process for new production facilities and eases the regulatory burden on existing plants. The ability to scale this process from kilogram to multi-ton quantities without encountering the safety bottlenecks of traditional sulfur chemistry allows for rapid capacity expansion to meet market growth. This combination of environmental stewardship and industrial scalability positions the technology as a future-proof solution for the evolving needs of the electronic and pharmaceutical sectors.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation and benefits of this advanced synthesis technology. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation, offering clarity on how this method outperforms legacy techniques. Understanding these details is essential for technical teams evaluating the feasibility of integrating this route into their existing manufacturing portfolios. The answers highlight the specific operational improvements and safety enhancements that define the value proposition of this innovative approach.
Q: How does this new method improve safety compared to traditional sulfur powder methods?
A: Traditional methods often use excess sulfur powder which poses spontaneous combustion risks. This patent utilizes a controlled stoichiometric ratio of 0.9-0.95 equivalents of sulfur and immediately captures the intermediate with acetyl chloride, significantly mitigating oxidation and safety hazards.
Q: What are the purity levels achievable with this synthesis route?
A: The described process consistently achieves GC purity levels exceeding 98.5%, with specific examples demonstrating purities up to 99.2%, making it highly suitable for sensitive electronic chemical applications.
Q: Can this process be scaled for industrial production?
A: Yes, the method is designed for large-scale production, utilizing standard inorganic bases and avoiding toxic by-products like HSCN, which simplifies waste treatment and facilitates commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Mercapto-1-Alkyl Imidazole Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to safer, more efficient chemical processes is vital for maintaining competitiveness in the global market. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of patents like CN107522661B are fully realized in practical, industrial settings. We are committed to delivering products that meet stringent purity specifications, supported by our rigorous QC labs that utilize state-of-the-art analytical instrumentation to verify every batch. Our capability to handle complex heterocyclic syntheses allows us to offer a level of quality assurance that few competitors can match, making us the ideal partner for your critical supply chain needs.
We invite you to engage with our technical procurement team to discuss how this advanced synthesis route can be tailored to your specific project requirements. By requesting a Customized Cost-Saving Analysis, you can gain a clear understanding of the economic impact of switching to this superior method. We encourage you to contact us today to obtain specific COA data and route feasibility assessments that will demonstrate our commitment to excellence and our ability to support your long-term growth objectives in the electronic chemicals sector.
