Advanced Resolution Technology for High-Purity S-Citalopram Intermediate S-Diol Manufacturing
The pharmaceutical industry's relentless pursuit of enantiomerically pure active pharmaceutical ingredients (APIs) has placed significant emphasis on the efficient production of S-Citalopram, the potent S-enantiomer of the antidepressant citalopram. Patent CN102190600A introduces a transformative preparation method for the key chiral intermediate, S-diol (I), addressing critical bottlenecks in scalability and purity that have historically hindered cost-effective manufacturing. This technology leverages a sophisticated crystallization strategy using a specific mixed solvent system of ethyl acetate and ethanol or acetonitrile to resolve the racemic precursor (II). By optimizing the thermodynamic conditions of crystallization, this method achieves exceptional optical purity and yield, positioning it as a cornerstone technology for reliable pharmaceutical intermediates supplier networks aiming to support large-scale generic and branded drug production.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the resolution of racemic 4-(4-dimethylamino-1-p-fluorophenyl-1-hydroxyl butyl)-3-(hydroxymethyl) cyanobenzene has been fraught with inefficiencies that compromise both economic viability and product quality. Early methodologies, such as those described in U.S. Pat 4943590, relied on solvents like Virahol which proved unsuitable for the rigorous demands of industrialized production due to poor crystal morphology and difficult filtration characteristics. Subsequent attempts documented in Chinese patent application CN 101265215 and PCT application WO2006106531 utilized mixed solvents containing halohydrocarbons or acetone; however, these processes suffered from a phenomenon known as 'impurity wrapping,' where the precipitating diastereomeric salts entrapped significant amounts of the unwanted enantiomer and excess resolving agent. This entrapment necessitated energy-intensive and yield-reducing recrystallization steps to achieve acceptable optical purity. Furthermore, methods relying on seed-induced crystallization, as seen in CN101386583, often resulted in inconsistent yields and variable purity profiles, creating unacceptable risks for cost reduction in API manufacturing where batch-to-batch consistency is paramount.
The Novel Approach
The innovative methodology disclosed in CN102190600A fundamentally re-engineers the crystallization environment to overcome these legacy defects by employing a binary solvent system centered on ethyl acetate. By dissolving the racemic substrate in ethyl acetate and introducing the resolving agent, D-(+) di-p-toluoyl-tartaric acid, via an ethanol or acetonitrile solution, the process creates a supersaturation gradient that favors the selective nucleation of the desired S-configured diastereomeric salt. This specific solvent combination ensures that the solubility of the target salt drops precipitously upon cooling while maintaining the unwanted enantiomer in solution, effectively preventing the co-crystallization of impurities. The result is the formation of even-grained, robust crystals that are easily filtered and washed, eliminating the need for repetitive purification cycles. This approach not only simplifies the operational workflow but also drastically enhances the overall mass balance of the synthesis, making it especially suitable for industrialized large-scale production where throughput and waste minimization are critical metrics.
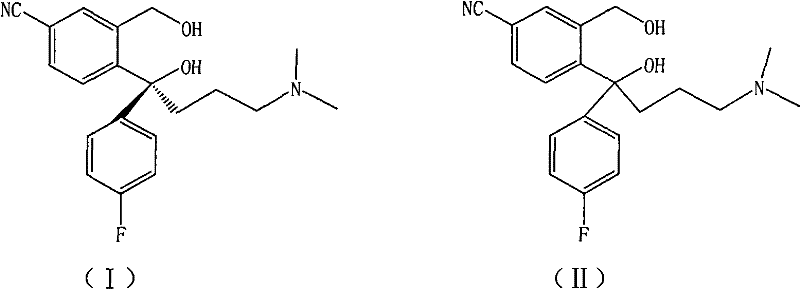
Mechanistic Insights into Diastereomeric Salt Crystallization
The core of this technological advancement lies in the precise manipulation of solubility parameters and nucleation kinetics within the ethyl acetate-based solvent matrix. When the racemic substrate is fully solvated in ethyl acetate at elevated temperatures (approximately 55°C), the system exists in a high-energy state where intermolecular interactions are minimized. Upon the dropwise addition of the chiral resolving agent dissolved in a miscible anti-solvent like ethanol, the local concentration of the diastereomeric pair increases rapidly. The specific ratio of ethyl acetate to ethanol (optimized between 1:4 and 1:5) is critical; it creates a thermodynamic window where the S-configured salt reaches supersaturation first, initiating spontaneous nucleation. Unlike chaotic precipitation, this controlled nucleation allows for orderly crystal lattice growth, where the chiral recognition between the substrate and the D-(+) di-p-toluoyl-tartaric acid is maximized, effectively excluding the R-enantiomer from the growing crystal face.

Furthermore, the mechanism inherently controls the impurity profile by leveraging the differential solubility of the diastereomeric salts at lower temperatures. As the reaction mixture is slowly cooled from 50°C down to 0°C, the solubility of the target S-salt decreases significantly, driving high recovery rates without dragging down the mother liquor's impurities. The slow cooling profile prevents 'oiling out' or amorphous precipitation, which are common causes of enantiomeric contamination in faster processes. The resulting crystal mass exhibits superior physical properties, allowing for efficient washing with cold ethyl acetate to remove surface-adhered mother liquor containing the R-enantiomer. This mechanistic precision ensures that the final free base, obtained after liberation with ammoniacal liquor, possesses the stringent purity specifications required for downstream cyclization into the final API, thereby securing the quality of the entire synthetic route.
How to Synthesize S-Diol Efficiently
To implement this high-efficiency resolution process, operators must adhere to strict control over solvent ratios and thermal gradients to replicate the patent's success. The procedure begins with the complete dissolution of the racemic precursor in ethyl acetate, followed by the controlled introduction of the resolving agent solution to initiate the chiral separation. Maintaining the specified temperature profile during the crystallization phase is essential to ensure the formation of the correct crystal polymorph and to maximize optical purity. While the general workflow is robust, detailed attention to the specific stoichiometry and cooling rates described in the patent examples is necessary to achieve the reported yields and purity levels. The detailed standardized synthesis steps see the guide below for exact operational parameters.
- Dissolve the racemic substrate in ethyl acetate at elevated temperature (approx. 55°C) to ensure complete solvation.
- Prepare a solution of the resolving agent, D-(+) di-p-toluoyl-tartaric acid, in either ethanol or acetonitrile and add it dropwise to the substrate solution.
- Control the cooling profile carefully, stirring at 50°C then slowly cooling to 0°C to induce crystallization of the desired diastereomeric salt.
- Filter the precipitated crystals under reduced pressure, wash with ethyl acetate, and liberate the free S-diol base using ammoniacal liquor and extraction.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this resolution technology translates directly into enhanced operational stability and significant cost optimization opportunities. By replacing hazardous and expensive halohydrocarbon solvents with commodity chemicals like ethyl acetate and ethanol, the process reduces raw material costs and simplifies regulatory compliance regarding solvent residues and environmental emissions. The elimination of multiple recrystallization steps, which were previously required to fix the 'impurity wrapping' issues of older methods, leads to a substantial reduction in processing time and utility consumption. This streamlined workflow not only lowers the cost of goods sold (COGS) but also increases the effective capacity of existing manufacturing assets, allowing for greater output without capital expenditure on new equipment.
- Cost Reduction in Manufacturing: The shift to a greener solvent system eliminates the need for costly solvent recovery systems associated with chlorinated hydrocarbons, while the high single-pass yield reduces the volume of material that needs to be reprocessed. By avoiding the loss of product inherent in repetitive recrystallization loops, the overall material efficiency is drastically improved, leading to tangible savings in raw material procurement. Additionally, the simplified workup procedure reduces labor hours and energy usage for heating and cooling cycles, contributing to a leaner manufacturing cost structure that enhances competitiveness in the global market.
- Enhanced Supply Chain Reliability: The reliance on widely available, non-controlled solvents such as ethyl acetate and ethanol mitigates the risk of supply disruptions that can occur with specialized or regulated chemicals. The robustness of the crystallization process ensures consistent batch quality, reducing the likelihood of production delays caused by out-of-specification results that require investigation and rework. This predictability allows supply chain planners to maintain tighter inventory controls and shorter lead times for high-purity pharmaceutical intermediates, ensuring a steady flow of materials to downstream API synthesis units without the volatility associated with less stable chemical processes.
- Scalability and Environmental Compliance: The formation of even-grained crystals with excellent filtration characteristics makes this process highly scalable from pilot plant to commercial tonnage without the engineering challenges posed by slimy or fine precipitates. The use of environmentally benign solvents aligns with increasingly strict global environmental regulations, reducing the burden of waste treatment and disposal costs associated with hazardous waste streams. This alignment with green chemistry principles not only future-proofs the manufacturing site against regulatory tightening but also enhances the corporate sustainability profile, which is increasingly valued by international pharmaceutical partners and stakeholders.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this resolution technology, derived directly from the experimental data and claims of the patent. Understanding these nuances is vital for R&D teams evaluating the feasibility of technology transfer and for procurement specialists assessing the long-term viability of the supply source. The answers provided reflect the specific advantages of the ethyl acetate-based system over traditional methods, highlighting its suitability for modern pharmaceutical manufacturing standards.
Q: What is the critical solvent system for high-yield resolution?
A: The patent specifies a mixed solvent system comprising ethyl acetate combined with either ethanol or acetonitrile. Specifically, a volume ratio of ethyl acetate to the alcohol/nitrile component of approximately 1:4 to 1:5 is optimal for maximizing crystal quality and optical purity.
Q: What optical purity can be achieved with this method?
A: Experimental data from the patent indicates that this resolution method consistently achieves chiral purity levels exceeding 98%, with specific embodiments reporting values up to 99.0% as measured by HPLC, significantly reducing the need for multiple recrystallization cycles.
Q: Why is ethyl acetate preferred over halohydrocarbons?
A: Ethyl acetate offers superior safety and environmental profiles compared to halohydrocarbons. Furthermore, it facilitates better crystal habit formation, preventing the 'wrapping' of impurities and enantiomers that often plagues older solvent systems, thereby streamlining the filtration process.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable S-Citalopram Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from laboratory innovation to commercial reality requires more than just a patent; it demands deep process engineering expertise and unwavering commitment to quality. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this resolution method are fully realized in a GMP-compliant manufacturing environment. We maintain stringent purity specifications and operate rigorous QC labs equipped with advanced chiral HPLC capabilities to guarantee that every batch of S-diol meets the exacting standards required for antidepressant API synthesis, providing our partners with absolute confidence in their supply chain.
We invite forward-thinking pharmaceutical companies to collaborate with us to leverage this advanced technology for their next-generation antidepressant portfolios. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are prepared to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our optimized manufacturing process can drive value and efficiency for your organization.
