Scalable Enzymatic Manufacturing of Topiroxostat Intermediates for Global Pharma Supply Chains
Scalable Enzymatic Manufacturing of Topiroxostat Intermediates for Global Pharma Supply Chains
The pharmaceutical industry is currently witnessing a significant paradigm shift towards greener, more sustainable synthetic methodologies, particularly for high-volume anti-gout agents like Topiroxostat. A pivotal advancement in this domain is detailed in patent CN115572747B, which discloses a novel preparation method that fundamentally reimagines the construction of the pyridyl-triazole scaffold. Unlike legacy processes that rely on hazardous stoichiometric reagents, this innovation leverages biocatalysis to achieve regioselective chlorination, followed by a streamlined cyclization and cyanation sequence. For R&D directors and procurement strategists, this patent represents a critical opportunity to optimize the supply chain for this high-purity API intermediate. By replacing toxic cyanation steps with an enzymatic approach, the technology not only enhances operator safety but also drastically simplifies the purification workflow, offering a compelling value proposition for manufacturers seeking to reduce their environmental footprint while maintaining rigorous quality standards.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
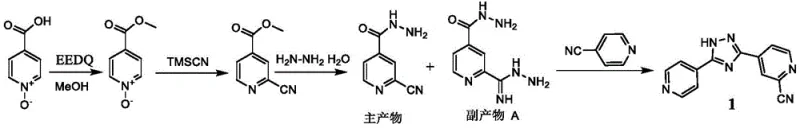
Historically, the synthesis of Topiroxostat has been plagued by significant safety and efficiency bottlenecks, primarily stemming from the reliance on extremely toxic cyanating agents. As illustrated in earlier patents such as CN1561340 and CN1826335 filed by Fuji Pharmaceutical, the conventional routes typically necessitate the use of trimethylsilyl cyanide (TMSCN) for introducing the cyano group. This reagent poses severe handling risks due to its volatility and toxicity, requiring specialized containment infrastructure and generating substantial hazardous waste streams that complicate regulatory compliance. Furthermore, these traditional pathways often suffer from poor atom economy and low selectivity, inevitably generating difficult-to-remove byproducts that mandate the use of column chromatography for purification. The reliance on chromatographic separation is a major impediment to commercial scale-up, as it is inherently batch-limited, solvent-intensive, and economically unviable for multi-ton production, thereby inflating the cost of goods sold and extending lead times for the final active pharmaceutical ingredient.
The Novel Approach
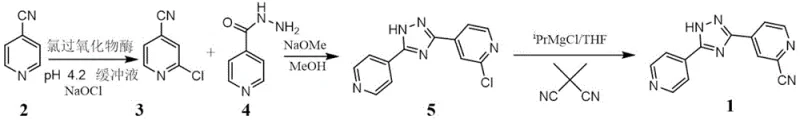
In stark contrast to these cumbersome legacy methods, the methodology disclosed in CN115572747B introduces a disruptive three-step sequence that prioritizes safety and scalability from the outset. The core innovation lies in the first step, where 4-cyanopyridine is subjected to enzymatic chlorination using chloroperoxidase and sodium hypochlorite. This biocatalytic transformation occurs under mild aqueous conditions, specifically at a controlled pH of 4.2 and temperatures ranging from 0 to 40°C, achieving remarkable regioselectivity for the 2-position without the need for protecting groups. This eliminates the hazardous cyanation step from the beginning of the synthesis, replacing it with a benign oxidative process. Subsequent steps involve a base-mediated cyclization with isonicotinyl hydrazide and a final Grignard-mediated substitution using dimethyl malononitrile. This strategic reordering of synthetic logic ensures that the most hazardous reagents are either eliminated or used in a controlled manner late in the sequence, while the bulk of the molecular complexity is built using safe, commodity-grade starting materials, resulting in a total yield that significantly outperforms traditional multi-step syntheses.
Mechanistic Insights into Enzymatic Chlorination and Grignard Substitution
The mechanistic elegance of this new route is anchored in the specificity of the chloroperoxidase enzyme during the initial functionalization of the pyridine ring. In Step S1, the enzyme facilitates the transfer of chlorine from sodium hypochlorite to the 2-position of 4-cyanopyridine with high fidelity, yielding 2-chloro-4-cyanopyridine with purity levels exceeding 98%. This enzymatic precision is crucial because it avoids the formation of polychlorinated impurities that are common in electrophilic aromatic substitution using harsh Lewis acids. The subsequent cyclization in Step S2 utilizes sodium methoxide in methanol to promote the condensation between the chloro-intermediate and isonicotinyl hydrazide, forming the central 1,2,4-triazole ring. The final transformation in Step S3 is a nucleophilic aromatic substitution where the chlorine atom on the triazole scaffold is displaced by a cyano group derived from dimethyl malononitrile, activated by isopropyl magnesium chloride. This Grignard-mediated cyanation is highly efficient, avoiding the direct use of gaseous HCN or solid metal cyanides, and proceeds smoothly in tetrahydrofuran at temperatures between -10 and 40°C, ensuring that the sensitive triazole core remains intact while the critical nitrile functionality is installed with high conversion rates.
From an impurity control perspective, this pathway offers distinct advantages by minimizing the generation of structural analogs that are difficult to purge. The high selectivity of the enzymatic step ensures that the starting material for the cyclization is exceptionally clean, which cascades through the rest of the synthesis to reduce the burden on the final crystallization. In traditional routes, side reactions during the cyanation step often lead to hydrolysis products or over-cyanated species that co-elute with the product. However, by utilizing dimethyl malononitrile as the cyanide source in the presence of a Grignard reagent, the reaction kinetics favor the desired substitution over hydrolysis. Furthermore, the workup procedures described, involving simple pH adjustments and solvent extractions followed by crystallization, are highly effective at removing inorganic salts and organic byproducts. This results in a final product with purity specifications consistently above 99%, meeting the stringent requirements for pharmaceutical intermediates without the need for preparative HPLC or extensive recrystallization cycles.
How to Synthesize Topiroxostat Efficiently
The implementation of this synthesis requires precise control over reaction parameters to maximize the benefits of the enzymatic and organometallic steps. The process begins with the preparation of a phosphate buffer system to maintain the optimal pH for chloroperoxidase activity, followed by the careful addition of oxidant to prevent enzyme deactivation. The subsequent cyclization requires anhydrous conditions to ensure complete conversion of the hydrazide, while the final Grignard step demands strict temperature control to manage the exotherm and prevent reagent decomposition. For a detailed breakdown of the specific molar ratios, solvent volumes, and isolation techniques required to replicate this high-yielding process, please refer to the standardized synthesis guide below.
- Step 1: Enzymatic Chlorination. React 4-cyanopyridine with sodium hypochlorite using chloroperoxidase in a phosphate buffer (pH 4.2) at 0-40°C to obtain 2-chloro-4-cyanopyridine with >98% purity.
- Step 2: Triazole Cyclization. Condense the chloro-intermediate with isonicotinyl hydrazide in methanol using sodium methoxide as a base at 20-80°C to form the chlorotriazole scaffold.
- Step 3: Cyanation via Grignard. React the chlorotriazole compound with dimethyl malononitrile in the presence of isopropyl magnesium chloride in THF at -10 to 40°C to yield final Topiroxostat.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this enzymatic route translates into tangible improvements in cost structure and supply reliability. The most significant economic driver is the elimination of trimethylsilyl cyanide (TMSCN), a reagent that commands a high market price due to its specialized handling requirements and limited supplier base. By substituting this with commodity chemicals like sodium hypochlorite and dimethyl malononitrile, the raw material costs are drastically reduced. Additionally, the removal of column chromatography from the purification train represents a massive reduction in processing time and solvent consumption. Chromatography is a major bottleneck in fine chemical manufacturing, often limiting batch sizes and requiring expensive silica gel and vast quantities of organic solvents. Replacing this with crystallization and filtration allows for the use of standard stainless steel reactors and centrifuges, facilitating a seamless transition from pilot scale to commercial production without the need for capital-intensive equipment upgrades.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven by the simplification of the unit operations and the use of inexpensive reagents. The enzymatic step operates in an aqueous buffer, reducing the demand for organic solvents in the early stages of synthesis. Furthermore, the high selectivity of the biocatalyst minimizes the formation of waste byproducts, which lowers the costs associated with waste treatment and disposal. The overall yield improvement means that less starting material is required to produce the same amount of final product, directly enhancing the gross margin for manufacturers. The avoidance of toxic reagents also reduces the regulatory burden and insurance costs associated with handling hazardous materials, contributing to a leaner and more cost-effective operational model.
- Enhanced Supply Chain Reliability: Supply chain resilience is significantly bolstered by the reliance on widely available starting materials. 4-cyanopyridine and isonicotinyl hydrazide are commodity chemicals produced by multiple global suppliers, mitigating the risk of single-source dependency. In contrast, specialized reagents like TMSCN or specific protecting group precursors often have volatile supply chains prone to disruption. The robustness of the enzymatic process also means that production is less susceptible to variations in raw material quality, as the enzyme provides a consistent level of selectivity. This stability ensures predictable lead times and consistent delivery schedules, which is critical for pharmaceutical companies managing just-in-time inventory for API production.
- Scalability and Environmental Compliance: The environmental profile of this synthesis aligns perfectly with modern green chemistry principles, making it easier to secure regulatory approvals and maintain social license to operate. The process generates significantly less hazardous waste, particularly by avoiding cyanide salts and silicon-containing byproducts that require special incineration. The mild reaction conditions (0-40°C) reduce energy consumption for heating and cooling compared to processes requiring cryogenic temperatures or high-pressure reflux. This lower energy intensity, combined with the ability to recycle solvents like methanol and THF, results in a substantially lower carbon footprint. For large-scale manufacturers, this translates to easier compliance with increasingly strict environmental regulations and a stronger sustainability narrative for their end customers.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented Topiroxostat synthesis. These insights are derived directly from the experimental data and process descriptions found in the patent literature, providing a clear understanding of the technology's capabilities and limitations. Understanding these details is essential for technical teams evaluating the feasibility of technology transfer and for commercial teams assessing the long-term viability of the supply chain.
Q: How does the enzymatic chlorination step improve safety compared to traditional methods?
A: Traditional methods often utilize highly toxic reagents like trimethylsilyl cyanide (TMSCN) or require harsh chlorination conditions. The patented enzymatic process uses chloroperoxidase and sodium hypochlorite under mild aqueous conditions (pH 4.2, 0-40°C), eliminating the need for hazardous cyanating agents in the early stages and significantly reducing environmental disposal costs.
Q: What is the expected purity profile of Topiroxostat produced via this route?
A: The process demonstrates exceptional selectivity, particularly in the enzymatic step which yields 2-chloro-4-cyanopyridine with purity exceeding 98%. The final API intermediate achieves purity levels between 99.1% and 99.4% after crystallization, minimizing the burden on downstream purification and ensuring compliance with stringent pharmacopeial standards.
Q: Is this synthesis route suitable for large-scale commercial production?
A: Yes, the route is designed for industrial scalability. It avoids column chromatography, relying instead on crystallization and filtration for purification. The reaction conditions are mild (mostly below 80°C), and the starting materials like 4-cyanopyridine are commodity chemicals, ensuring a robust and cost-effective supply chain for multi-ton manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Topiroxostat Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this enzymatic synthesis route for the global gout medication market. As a leading CDMO partner, we possess the technical expertise and infrastructure to rapidly adopt and optimize this green chemistry pathway for our clients. Our facilities are equipped with state-of-the-art biocatalysis suites and multipurpose reactors capable of handling sensitive organometallic reactions, ensuring that the transition from lab scale to commercial production is seamless. We bring extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, guaranteeing that your supply needs are met with consistency and precision. Our commitment to quality is underpinned by stringent purity specifications and rigorous QC labs that utilize advanced analytical techniques to verify the identity and purity of every batch, ensuring full compliance with international pharmacopeial standards.
We invite pharmaceutical partners to collaborate with us to leverage this innovative technology for their Topiroxostat supply chains. By partnering with our technical procurement team, you can access a Customized Cost-Saving Analysis that quantifies the specific economic benefits of switching to this enzymatic route for your volume requirements. We encourage you to contact us today to request specific COA data from our pilot runs and to discuss route feasibility assessments tailored to your project timelines. Together, we can drive down costs and enhance the sustainability of your anti-gout drug portfolio.
