Advanced Synthesis of Difluorophenyl-Modified Iridium Complexes for Next-Generation OLED Applications
Advanced Synthesis of Difluorophenyl-Modified Iridium Complexes for Next-Generation OLED Applications
The rapid evolution of organic light-emitting diode (OLED) technology demands phosphorescent materials that not only exhibit high quantum efficiency but also maintain stability in solid-state devices. A significant breakthrough in this domain is documented in patent CN116396336A, which discloses a sophisticated preparation method for difluorophenyl-modified aggregation-induced luminescent iridium complexes. This intellectual property introduces two distinct cationic iridium(III) complexes, designated as Ir1 and Ir2, which overcome the traditional limitations of aggregation-caused quenching (ACQ) by leveraging aggregation-induced emission (AIE) mechanisms. For R&D directors and procurement specialists in the electronic chemicals sector, this patent represents a pivotal shift towards more efficient emitters that perform exceptionally well in high-concentration environments typical of OLED emissive layers. The strategic incorporation of electron-withdrawing trifluoromethyl and difluorophenyl groups fine-tunes the electronic properties, resulting in materials with superior photophysical characteristics suitable for next-generation display technologies.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the development of phosphorescent emitters for OLED applications has been plagued by the phenomenon known as Aggregation-Caused Quenching (ACQ). In conventional fluorescent and phosphorescent molecules, strong intermolecular interactions in the solid state or at high concentrations often lead to non-radiative decay pathways, drastically reducing emission efficiency. This poses a severe challenge for device fabrication, where emitters must be densely packed within the host matrix to ensure effective charge transport and energy transfer. Traditional synthesis routes for iridium complexes often rely on neutral ligands that do not sufficiently suppress these detrimental pi-pi stacking interactions. Furthermore, many existing synthetic protocols involve harsh reaction conditions, expensive noble metal catalysts in excessive amounts, or multi-step purifications that generate significant chemical waste. These factors collectively increase the cost of goods sold (COGS) and complicate the supply chain for high-purity electronic chemicals, making mass production economically challenging for many manufacturers seeking reliable OLED material suppliers.
The Novel Approach
The methodology outlined in CN116396336A offers a transformative solution by engineering iridium complexes that exhibit Aggregation-Induced Emission (AIE) properties. Unlike their ACQ-prone counterparts, these difluorophenyl-modified complexes remain weakly emissive in dilute solutions but become highly luminescent when aggregated or in the solid state. This is achieved through a modular design strategy that combines a specific cyclometalated ligand, 2-(4-(4-(2,4-difluorophenyl)phenyl)phenyl)-5-trifluoromethylpyridine, with distinct N^N auxiliary ligands. The introduction of bulky fluorinated groups creates steric hindrance that restricts intramolecular rotation and prevents close pi-pi stacking, thereby opening radiative decay channels. Experimental data from the patent indicates that the emission intensity ratio (I/I0) in acetonitrile/water mixtures exceeds 10, with Ir2 reaching a remarkable ratio of 10.92. This novel approach not only enhances device efficiency but also simplifies the formulation process for OLED manufacturers, providing a clear pathway for cost reduction in electronic chemical manufacturing through improved material performance and reduced loading requirements.
Mechanistic Insights into Suzuki Coupling and Cyclometalation
The synthetic architecture of these high-performance materials relies on a robust sequence of palladium-catalyzed cross-coupling reactions followed by precise coordination chemistry. The construction of the cyclometalated ligand begins with a Suzuki-Miyaura coupling between 2-bromo-5-trifluoromethylpyridine and 4-bromophenylboronic acid. This step is critically optimized to proceed under aerobic conditions at 80°C using potassium carbonate as the base and palladium acetate as the catalyst, yielding the bromo-functionalized intermediate with a 50% isolated yield. Subsequently, a second Suzuki coupling connects this intermediate with 2,4-difluorophenylboronic acid under inert nitrogen atmosphere at 70°C, utilizing tetrakis(triphenylphosphine)palladium to achieve a high yield of 79%. This two-step ligand synthesis ensures the precise placement of electron-withdrawing fluorine atoms, which are essential for lowering the LUMO energy levels and stabilizing the excited state. The final coordination involves reacting the ligand with IrCl3·3H2O in a mixture of ethylene glycol monoethyl ether and water at 120°C to form a chloro-bridged dimer, which is then cleaved by the auxiliary ligands to form the final cationic monomers.
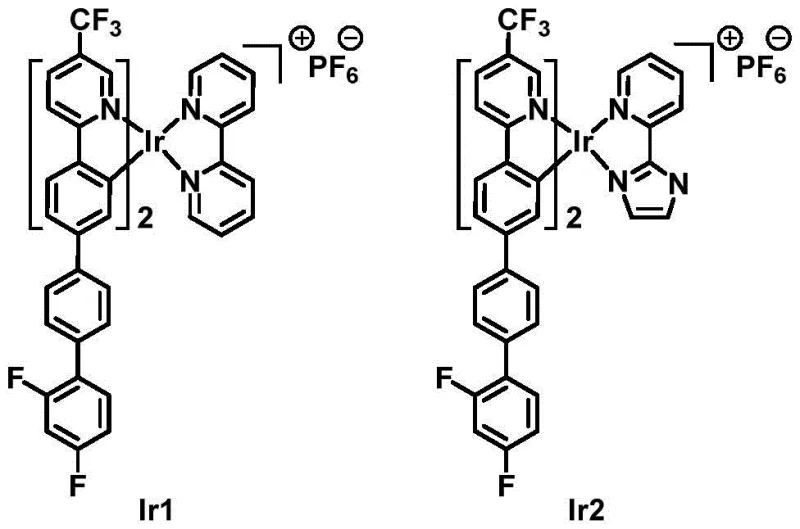
The structural diversity of the final products is achieved by varying the N^N auxiliary ligand, which profoundly influences the photophysical properties and packing arrangements of the complexes. As illustrated in the molecular diagrams, Ir1 incorporates a 2,2'-bipyridine auxiliary ligand, while Ir2 utilizes a 2-(2-imidazolyl)pyridine moiety. This variation allows for fine-tuning of the emission color and lifetime without altering the core cyclometalated framework. The presence of the hexafluorophosphate (PF6-) counterion, introduced via metathesis with saturated KPF6 solution, ensures the formation of stable ionic salts that are soluble in common organic processing solvents. The rigorous control over reaction stoichiometry, such as using 2.5 equivalents of the cyclometal ligand relative to iridium chloride, drives the equilibrium towards the desired bis-cyclometalated species, minimizing the formation of homoleptic or mono-cyclometalated impurities. This level of mechanistic control is vital for achieving the stringent purity specifications required in the semiconductor and display industries.
How to Synthesize Difluorophenyl-Modified Iridium Complexes Efficiently
The synthesis of these advanced phosphorescent materials follows a streamlined three-stage protocol that balances high yield with operational simplicity, making it highly attractive for contract development and manufacturing organizations (CDMOs). The process initiates with the construction of the functionalized pyridine backbone, proceeds through the assembly of the terphenyl-like cyclometalating ligand, and concludes with the metallation and anion exchange steps. Each stage has been optimized to minimize side reactions and facilitate purification via standard column chromatography. For technical teams looking to replicate or scale this chemistry, the detailed standardized synthesis steps are provided in the guide below, ensuring reproducibility and adherence to the quality benchmarks set forth in the patent literature.
- Perform Suzuki cross-coupling of 2-bromo-5-trifluoromethylpyridine with 4-bromophenylboronic acid using Pd(OAc)2 catalyst at 80°C.
- Execute a second Suzuki coupling with 2,4-difluorophenylboronic acid under nitrogen protection at 70°C to form the cyclometal ligand.
- Coordinate the ligand with IrCl3·3H2O to form a chloro-bridged dimer, then react with auxiliary ligands (bipyridine or imidazolyl-pyridine) and KPF6.
Commercial Advantages for Procurement and Supply Chain Teams
From a supply chain and procurement perspective, the synthetic route described in CN116396336A offers substantial advantages over legacy methods for producing phosphorescent dopants. The reliance on Suzuki cross-coupling, a well-established and widely available industrial reaction, ensures that the necessary catalytic systems and reagents are readily accessible from multiple global vendors, reducing the risk of single-source bottlenecks. Furthermore, the use of common solvents such as ethanol, tetrahydrofuran, and ethylene glycol monoethyl ether eliminates the need for specialized or hazardous solvent handling infrastructure, thereby lowering capital expenditure for production facilities. The modular nature of the synthesis allows for the decoupled production of the cyclometal ligand and the auxiliary ligands, enabling inventory flexibility and just-in-time manufacturing strategies that are crucial for maintaining lean supply chains in the volatile electronics market.
- Cost Reduction in Manufacturing: The elimination of exotic reagents and the use of air-stable conditions for the initial coupling step significantly lower the operational costs associated with inert gas consumption and specialized equipment. By achieving high yields in the ligand synthesis stage (up to 79%), the overall material throughput is maximized, reducing the cost per gram of the final active pharmaceutical ingredient (API) equivalent in the electronic sector. Additionally, the straightforward purification via column chromatography using dichloromethane and petroleum ether avoids the need for expensive preparative HPLC or sublimation processes in the early stages, driving down the total cost of ownership for the manufacturing process.
- Enhanced Supply Chain Reliability: The starting materials, including various bromopyridines and phenylboronic acids, are commodity chemicals produced at multi-ton scales globally, ensuring long-term supply continuity. The robustness of the reaction conditions, which tolerate moderate temperatures (70-120°C) and standard pressures, means that production can be easily transferred between different manufacturing sites without extensive re-validation. This geographic flexibility is a key asset for supply chain heads aiming to diversify their supplier base and mitigate risks associated with regional disruptions or trade tariffs affecting the flow of high-purity OLED materials.
- Scalability and Environmental Compliance: The process generates manageable waste streams primarily consisting of inorganic salts and spent organic solvents, which can be treated using standard industrial waste management protocols. The absence of highly toxic heavy metals other than the product itself (iridium) simplifies environmental compliance and worker safety measures. Moreover, the high atom economy of the Suzuki coupling steps aligns with green chemistry principles, enhancing the sustainability profile of the final product. This is increasingly important for multinational corporations striving to meet corporate social responsibility (CSR) goals and reduce the carbon footprint of their electronic component sourcing.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these difluorophenyl-modified iridium complexes. These insights are derived directly from the experimental data and claims within the patent specification, providing a reliable foundation for decision-making. Understanding these nuances is essential for stakeholders evaluating the feasibility of integrating these materials into existing OLED production lines or developing new device architectures that leverage aggregation-induced emission.
Q: What distinguishes these iridium complexes from traditional phosphorescent materials?
A: Unlike traditional fluorophores that suffer from Aggregation-Caused Quenching (ACQ), these complexes exhibit Aggregation-Induced Emission (AIE), showing significantly enhanced luminescence in aggregated states or solid films, which is critical for OLED efficiency.
Q: What are the key yield metrics for the synthetic route described in CN116396336A?
A: The patent reports a 50% yield for the first intermediate, 79% for the final cyclometal ligand, and overall yields of 54% for Ir1 and 68% for Ir2, demonstrating a robust and viable pathway for production.
Q: Can this synthesis be scaled for industrial OLED material manufacturing?
A: Yes, the process utilizes standard Suzuki coupling conditions and common solvents like ethylene glycol monoethyl ether, avoiding exotic reagents, which facilitates commercial scale-up and supply chain reliability.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Difluorophenyl Iridium Complex Supplier
The technological potential of the synthesis route described in CN116396336A is immense, offering a clear path to high-efficiency, solid-state luminescent materials that solve the perennial problem of efficiency roll-off in OLEDs. At NINGBO INNO PHARMCHEM, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive consistent quality regardless of order volume. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, including trace metal analysis and isotopic purity, which are critical for the performance and longevity of electronic devices. We understand that the transition from bench-scale synthesis to industrial manufacturing requires meticulous attention to detail, and our team is dedicated to bridging that gap seamlessly.
We invite procurement leaders and R&D directors to engage with us for a Customized Cost-Saving Analysis tailored to your specific volume requirements and purity needs. By partnering with our technical procurement team, you can access specific COA data and comprehensive route feasibility assessments that will help you optimize your supply chain for maximum efficiency and cost-effectiveness. Whether you require custom synthesis of the Ir1 or Ir2 analogues or scale-up of the intermediate ligands, our expertise in organometallic chemistry positions us as the ideal partner for your next-generation display material projects.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
