Advanced Platinum Luminescent Compounds for High-Efficiency OLED Display Manufacturing
Introduction to Next-Generation OLED Emitters
The rapid evolution of the organic light-emitting diode (OLED) industry demands materials that balance high performance with economic viability, a challenge addressed directly by the technological breakthroughs detailed in patent CN112028943A. This patent introduces a novel platinum luminescent compound based on pyridoimidazole derivatives, specifically designed to overcome the cost barriers associated with traditional iridium-based phosphors. As the market shifts towards more sustainable and cost-effective display technologies, the development of bivalent platinum complexes represents a critical pivot point for manufacturers seeking to optimize their supply chains without compromising on luminous efficiency. The compound, characterized by the molecular formula C60H66N6O6Pt2, demonstrates exceptional photophysical properties including a solid powder photoluminescence quantum efficiency of 14.38% at 293K and a remarkable lifetime of 14.65μs. These metrics position the material as a highly competitive candidate for next-generation lighting and display applications, offering a robust alternative to expensive rare-earth metals while maintaining the rigorous performance standards required by top-tier electronics manufacturers.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the OLED industry has relied heavily on iridium (Ir) complexes to achieve high internal quantum efficiency through effective harvesting of triplet excitons via strong spin-orbit coupling. However, the reliance on iridium presents significant strategic vulnerabilities for global supply chains, primarily due to the metal's status as a scarce and expensive rare earth element. The extraction and purification of iridium involve complex, energy-intensive processes that not only drive up raw material costs but also introduce substantial environmental burdens through heavy metal pollution. Furthermore, the volatility of precious metal markets can lead to unpredictable pricing fluctuations, making long-term cost forecasting difficult for large-scale display panel producers. Traditional synthesis routes for these high-performance emitters often require harsh reaction conditions and specialized catalysts that complicate purification protocols, leading to lower overall yields and higher waste generation. These factors collectively create a bottleneck for the mass adoption of OLED technology in cost-sensitive consumer electronics sectors, necessitating a shift towards more abundant and economically stable metal centers.
The Novel Approach
The innovative methodology presented in the patent data circumvents these traditional bottlenecks by utilizing platinum (Pt) as the central metal atom, which offers a far more favorable cost-to-performance ratio compared to iridium. The synthesis strategy employs a modular approach, constructing a specialized pyridoimidazole ligand system that effectively stabilizes the platinum center in a planar quadrilateral geometry, crucial for maintaining high phosphorescence efficiency. By leveraging common and recyclable solvents such as ethylene glycol and ethanol, the process significantly reduces the environmental footprint and operational complexity typically associated with organometallic synthesis. The reaction conditions are notably mild, with key steps occurring at temperatures ranging from 60°C to 130°C, which enhances safety profiles and reduces energy consumption during manufacturing. This novel approach not only lowers the barrier to entry for producing high-quality luminescent materials but also ensures a more stable and predictable supply chain for downstream OLED device fabricators looking to reduce their dependency on volatile rare earth markets.
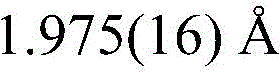
Mechanistic Insights into Pt(II) Cyclometalation and Coordination
The core of this technology lies in the precise engineering of the coordination environment around the platinum center, which dictates the electronic transitions responsible for light emission. In the described complex, the central Pt(II) atom adopts a typical planar quadrilateral structure, coordinated by carbon and nitrogen atoms from the pyridoimidazole ligand and two oxygen atoms from the auxiliary 2,2,6,6-tetramethyl-3,5-heptanedione (dpm) ligand. This specific geometric arrangement facilitates strong spin-orbit coupling (SOC), enabling rapid intersystem crossing (ISC) from singlet to triplet states, which is the fundamental mechanism allowing for the harvesting of triplet excitons in phosphorescent OLEDs. The bond lengths within the complex, such as the Pt-O bonds measuring approximately 2.111 Å and 1.975 Å, and the Pt-N and Pt-C bonds around 1.975 Å and 1.981 Å respectively, indicate a tight and stable coordination sphere that minimizes non-radiative decay pathways. This structural rigidity is essential for maintaining high quantum efficiency in the solid state, where molecular motion can otherwise quench excited states.
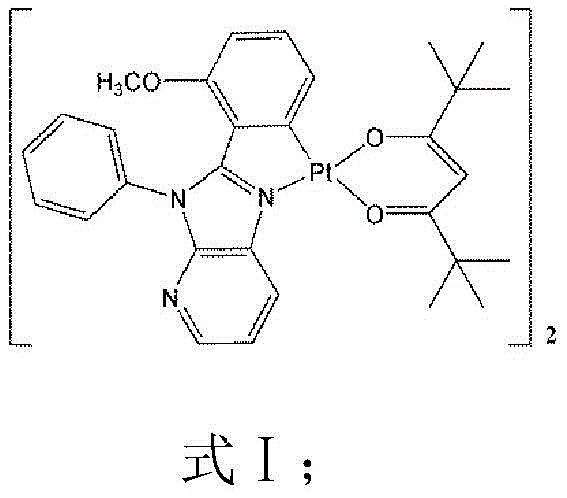
Furthermore, the electronic nature of the pyridoimidazole ligand plays a pivotal role in tuning the emission color and efficiency of the final material. The conjugation within the ligand system supports intraligand charge transfer (ILCT) transitions, observed as absorption bands in the 320nm to 350nm range, while the interaction between the metal d-orbitals and ligand π*-orbitals facilitates metal-to-ligand charge transfer (MLCT). The presence of the methoxy group on the phenyl ring of the ligand further modulates the electron density, fine-tuning the HOMO-LUMO gap to achieve the desired green-yellow emission peaks at 515nm and 546nm. Understanding these mechanistic details is vital for R&D teams aiming to replicate or modify the synthesis for specific color coordinates, as slight variations in ligand substitution can drastically alter the photophysical profile. The robustness of this coordination chemistry ensures that the material maintains its integrity under the electrical stress conditions found in operating OLED devices, promising longer device lifetimes and consistent color performance over time.
How to Synthesize Pyridoimidazole Platinum Complex Efficiently
The synthesis of this high-value electronic chemical involves a streamlined six-step process that begins with the construction of the organic ligand framework followed by the metallation and final coordination steps. The initial phases focus on building the pyridoimidazole core through nucleophilic substitution and reduction reactions, utilizing readily available starting materials like 2-chloro-3-nitropyridine and aniline. Subsequent oxidation and condensation steps refine the ligand structure to ensure optimal binding affinity for the platinum center. The final stages involve the coordination of the ligand with potassium tetrachloroplatinate to form a chloro-bridged dimer, which is then converted into the monomeric target compound using the beta-diketonate auxiliary ligand. This logical progression allows for easy monitoring of reaction progress and purification at each stage, ensuring high purity levels essential for electronic applications. For a detailed breakdown of the specific reaction parameters, stoichiometry, and workup procedures required to achieve reproducible results, please refer to the standardized synthesis guide below.
- Synthesize the pyridoimidazole ligand precursor through condensation and oxidation steps using 2-chloro-3-nitropyridine and aniline derivatives.
- Coordinate the ligand with potassium tetrachloroplatinate in an ethylene glycol-water mixture at 80°C to form the chloro-bridged dimer intermediate.
- React the intermediate with 2,2,6,6-tetramethylheptanedione and sodium carbonate at 130°C to finalize the planar quadrilateral platinum complex structure.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, the adoption of this platinum-based luminescent compound offers distinct strategic advantages over traditional iridium counterparts, primarily driven by raw material availability and process simplicity. The shift from scarce rare earth metals to platinum, which is relatively more abundant and price-stable, mitigates the risk of supply disruptions and cost spikes that frequently plague the electronics sector. Additionally, the synthesis protocol described in the patent utilizes common industrial solvents such as ethylene glycol, ethanol, and dichloromethane, all of which are easily sourced and can be efficiently recovered and recycled through standard distillation processes. This solvent recovery capability not only reduces the volume of hazardous waste requiring disposal but also lowers the overall operational expenditure associated with raw material consumption. The mild reaction temperatures and atmospheric pressure conditions further simplify the engineering requirements for production facilities, allowing for the use of standard glass-lined or stainless steel reactors without the need for specialized high-pressure equipment.
- Cost Reduction in Manufacturing: The substitution of expensive iridium with platinum fundamentally alters the cost structure of the emitter material, providing a direct reduction in the bill of materials for OLED panel production. By eliminating the need for costly rare earth precursors and reducing the complexity of the purification steps through the use of robust crystallization techniques, manufacturers can achieve significant savings in both direct material costs and processing overhead. The ability to recycle solvents like ethylene glycol and water mixtures further compounds these savings, creating a leaner and more cost-efficient production cycle that enhances overall profit margins without sacrificing product quality.
- Enhanced Supply Chain Reliability: Relying on platinum and common organic building blocks diversifies the supply base, reducing dependency on geographically concentrated sources of rare earth elements that are often subject to trade restrictions or geopolitical instability. The synthetic route utilizes commodity chemicals that are widely available from multiple global suppliers, ensuring continuity of supply even during market fluctuations. This resilience is critical for maintaining consistent production schedules for high-volume display manufacturing, where any interruption in material flow can result in substantial downstream losses and missed market opportunities.
- Scalability and Environmental Compliance: The process is inherently scalable due to its reliance on homogeneous solution-phase reactions that translate well from laboratory to pilot and commercial scales. The use of mild conditions and recyclable solvents aligns with increasingly stringent environmental regulations regarding volatile organic compound (VOC) emissions and heavy metal waste. By minimizing the generation of hazardous byproducts and facilitating solvent reuse, this technology supports sustainable manufacturing practices, helping companies meet their corporate social responsibility goals and comply with international environmental standards such as RoHS and REACH.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this platinum luminescent compound in industrial settings. These answers are derived directly from the experimental data and structural analysis provided in the patent documentation, ensuring accuracy and relevance for technical decision-makers. Understanding these specifics is crucial for evaluating the feasibility of integrating this material into existing OLED fabrication lines and for assessing its long-term viability as a standard emitter. We encourage stakeholders to review these points carefully to gain a comprehensive understanding of the material's capabilities and limitations.
Q: What are the photoluminescence properties of this platinum complex?
A: The compound exhibits a maximum excitation wavelength of 342nm and dual emission peaks at 515nm and 546nm, with a solid-state quantum efficiency of 14.38% and a lifetime of 14.65μs.
Q: Why is platinum preferred over iridium for this application?
A: Platinum offers a significantly lower cost profile compared to rare earth iridium while maintaining strong spin-orbit coupling capabilities necessary for efficient phosphorescence in OLED devices.
Q: Is the synthesis process scalable for industrial production?
A: Yes, the process utilizes mild reaction conditions (60-130°C) and common solvents like ethylene glycol and ethanol which can be recovered and reused, facilitating easier scale-up and waste management.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Platinum Luminescent Compound Supplier
As a leader in the fine chemical sector, NINGBO INNO PHARMCHEM is uniquely positioned to support the commercialization of advanced OLED materials like the pyridoimidazole-based platinum complex described in CN112028943A. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to mass manufacturing is seamless and efficient. We maintain stringent purity specifications and operate rigorous QC labs equipped with state-of-the-art analytical instrumentation to guarantee that every batch meets the exacting standards required for electronic grade materials. Our commitment to quality and consistency makes us the ideal partner for display manufacturers seeking to secure a stable supply of high-performance luminescent compounds.
We invite you to engage with our technical procurement team to discuss how we can tailor our production capabilities to your specific volume and purity requirements. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into how switching to our platinum-based emitters can optimize your overall manufacturing costs. We are ready to provide specific COA data and route feasibility assessments to demonstrate our capability to deliver reliable, high-quality materials that drive innovation in your display products.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
