Advanced Synthesis of O-Aminophenol Derivatives for Commercial Scale-Up and High-Purity Applications
Advanced Synthesis of O-Aminophenol Derivatives for Commercial Scale-Up and High-Purity Applications
The pharmaceutical and agrochemical industries continuously demand efficient routes to complex structural motifs, particularly o-aminophenol derivatives, which serve as critical building blocks for kinase inhibitors and other biologically active compounds. Patent CN108148005B introduces a groundbreaking methodology that leverages transition metal-catalyzed C-H bond functionalization to streamline the production of these valuable intermediates. This technology addresses long-standing challenges in organic synthesis by utilizing a transient directing group strategy that significantly simplifies the overall process flow. By employing 2-chloro-5-nitropyrimidine as a robust guiding group, the invention enables precise ortho-functionalization under relatively mild conditions compared to classical approaches. The strategic implementation of palladium catalysis combined with hypervalent iodine oxidants allows for high regioselectivity, ensuring that the desired structural isomers are produced with minimal byproduct formation. This level of control is paramount for R&D directors seeking to optimize impurity profiles in early-stage drug development. Furthermore, the protocol demonstrates exceptional functional group tolerance, accommodating various substituents such as halogens, alkyl groups, and electron-withdrawing moieties without compromising reaction efficiency.
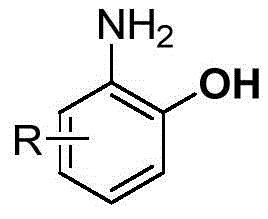
The versatility of this synthetic route extends beyond simple substrate scope, offering a scalable pathway that aligns with modern green chemistry principles. Traditional methods often rely on stoichiometric amounts of hazardous reagents or require extreme temperatures that pose safety risks in a manufacturing environment. In contrast, the disclosed method operates at moderate temperatures and utilizes solvents like acetonitrile and tetrahydrofuran, which are standard in industrial settings. The ability to perform these transformations with high atom economy and reduced waste generation makes this patent highly attractive for procurement managers focused on sustainability and cost efficiency. Moreover, the final deprotection step utilizes hydrazine hydrate at room temperature, eliminating the need for energy-intensive heating or dangerous metal reductions. This innovation not only enhances operational safety but also reduces the capital expenditure associated with specialized high-pressure or high-temperature reactor systems. As global regulatory standards tighten around chemical manufacturing, adopting such inherently safer and cleaner technologies becomes a strategic imperative for supply chain heads aiming to ensure long-term continuity and compliance.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of ortho-functionalized phenols has been plagued by significant technical hurdles that hinder efficient commercial production. Conventional strategies frequently depend on the use of strong bases, active metals like sodium or potassium, and harsh reaction conditions that are difficult to control on a large scale. These aggressive environments often lead to poor selectivity, resulting in complex mixtures of regioisomers that are costly and time-consuming to separate. The requirement for cryogenic temperatures or prolonged heating cycles further exacerbates energy consumption and increases the risk of thermal runaway incidents in plant operations. Additionally, the removal of traditional directing groups often necessitates multiple purification steps, including extensive chromatography or recrystallization, which drastically lowers the overall yield and throughput. For procurement teams, these inefficiencies translate into higher raw material costs and extended lead times, making the final API intermediates less competitive in the global market. The reliance on sensitive reagents that degrade upon exposure to air or moisture also complicates logistics and storage, adding another layer of complexity to the supply chain management of these critical chemicals.
The Novel Approach
The methodology outlined in patent CN108148005B represents a paradigm shift by introducing a mild and highly selective catalytic cycle that overcomes the drawbacks of legacy processes. By utilizing a pyrimidine-based directing group, the reaction achieves precise C-H activation at the ortho-position without the need for pre-functionalized substrates, thereby reducing the number of synthetic steps required. The use of palladium acetate as a catalyst in conjunction with iodobenzene diacetate as a terminal oxidant facilitates a smooth transformation that proceeds with high conversion rates. Crucially, the conditions are compatible with a wide array of functional groups, allowing chemists to introduce diverse substituents early in the synthesis without protecting group manipulation. This streamlining of the synthetic route directly impacts the cost of goods sold by minimizing waste and maximizing the utilization of starting materials. Furthermore, the final cleavage of the directing group is achieved under neutral conditions using hydrazine hydrate, which is a significant improvement over the acidic or basic hydrolysis typically required. This gentle deprotection ensures the integrity of sensitive molecular architectures, making the process ideal for synthesizing complex pharmaceutical intermediates where structural fidelity is non-negotiable.
Mechanistic Insights into Pd-Catalyzed C-H Acetoxylation
At the heart of this innovative synthesis lies a sophisticated catalytic mechanism that orchestrates the selective functionalization of inert C-H bonds. The process initiates with the coordination of the palladium catalyst to the nitrogen atoms of the 2-chloro-5-nitropyrimidine directing group, forming a stable cyclopalladated intermediate. This coordination brings the metal center into close proximity with the ortho-C-H bond, lowering the activation energy required for cleavage. Subsequent oxidation by the hypervalent iodine species generates a high-valent palladium complex, which facilitates the insertion of the acetoxy group onto the aromatic ring. The electronic properties of the nitro group on the pyrimidine ring play a crucial role in modulating the electrophilicity of the directing group, enhancing its ability to coordinate with the metal center while remaining easy to remove later. This delicate balance between stability during the reaction and lability during workup is key to the success of the method. Understanding this mechanistic nuance allows R&D teams to fine-tune reaction parameters such as solvent polarity and catalyst loading to achieve optimal performance for specific substrates.
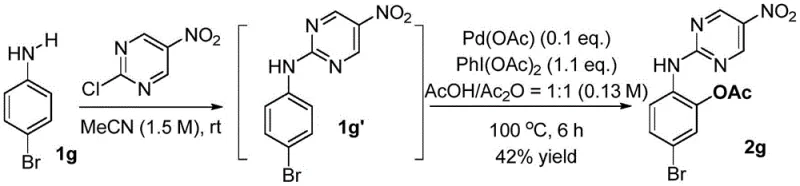
Following the C-H activation step, the removal of the directing group is executed through a nucleophilic substitution mechanism driven by hydrazine hydrate. The hydrazine attacks the electron-deficient pyrimidine ring, displacing the aniline moiety and regenerating the free phenol functionality. This step proceeds rapidly at room temperature in tetrahydrofuran, highlighting the kinetic favorability of the cleavage reaction. The mildness of this condition is particularly beneficial for substrates containing acid- or base-sensitive functionalities that would otherwise decompose under traditional hydrolysis conditions. The entire sequence demonstrates a high degree of chemoselectivity, ensuring that other reactive sites on the molecule remain untouched. For process chemists, this level of predictability reduces the need for extensive troubleshooting during scale-up, accelerating the timeline from laboratory bench to pilot plant. The mechanistic clarity provided by the patent data empowers technical teams to implement robust quality control measures, ensuring that every batch meets the stringent purity specifications required for downstream pharmaceutical applications.

How to Synthesize O-Aminophenol Derivatives Efficiently
Implementing this synthesis route requires careful attention to reaction parameters to maximize yield and purity while maintaining safety standards. The process begins with the preparation of the pyrimidine-aryl amine intermediate, which serves as the foundation for the subsequent C-H activation. Operators must ensure that the acetonitrile solvent is thoroughly dried and degassed to prevent catalyst deactivation, as moisture and oxygen can inhibit the palladium cycle. The reaction is typically conducted under a nitrogen atmosphere to maintain an inert environment, which is standard practice in fine chemical manufacturing. Following the isolation of the intermediate, the C-H activation step involves precise control of temperature and stoichiometry to drive the reaction to completion without generating excessive byproducts. The final deprotection step is straightforward but requires careful quenching and extraction to remove hydrazine residues effectively. Detailed standardized synthesis steps are provided below to guide technical teams in replicating this high-performance protocol.
- React arylamine with 2-chloro-5-nitropyrimidine in acetonitrile to install the directing group.
- Perform Pd-catalyzed C-H activation using PhI(OAc)2 to achieve ortho-acetoxylation.
- Remove the directing group using hydrazine hydrate in THF at room temperature to yield the final product.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this patented technology offers substantial benefits that extend beyond mere technical feasibility. For procurement managers, the ability to utilize readily available starting materials such as substituted anilines and commodity chemicals like 2-chloro-5-nitropyrimidine significantly reduces raw material costs and supply risks. The elimination of exotic or highly regulated reagents simplifies the sourcing process and enhances supply chain resilience against market fluctuations. Moreover, the streamlined nature of the synthesis reduces the number of unit operations required, leading to lower utility consumption and reduced waste disposal costs. This efficiency translates into a more competitive pricing structure for the final intermediates, allowing pharmaceutical companies to optimize their overall production budgets. The robustness of the process also minimizes the risk of batch failures, ensuring a consistent supply of high-quality materials that keep downstream manufacturing schedules on track.
- Cost Reduction in Manufacturing: The process eliminates the need for expensive and hazardous reagents typically associated with traditional ortho-functionalization methods, leading to significant operational savings. By avoiding the use of active metals and harsh conditions, the requirement for specialized corrosion-resistant equipment is reduced, lowering capital expenditure. The high selectivity of the reaction minimizes the formation of difficult-to-remove impurities, which reduces the load on purification units and decreases solvent consumption. These factors collectively contribute to a leaner manufacturing process that maximizes resource utilization and drives down the cost per kilogram of the final product.
- Enhanced Supply Chain Reliability: The reliance on stable and commercially abundant reagents ensures that production is not vulnerable to shortages of niche chemicals. The mild reaction conditions allow for flexible manufacturing schedules, as the process does not require extended cooling or heating periods that tie up reactor capacity. This flexibility enables suppliers to respond more quickly to changes in demand, reducing lead times for customers. Additionally, the safety profile of the process reduces the likelihood of unplanned shutdowns due to safety incidents, ensuring a continuous and reliable flow of materials to the market.
- Scalability and Environmental Compliance: The protocol has been demonstrated to scale effectively from gram to multi-gram quantities without loss of efficiency, indicating strong potential for ton-scale production. The use of common organic solvents and the absence of heavy metal waste streams simplify waste treatment and disposal, aligning with increasingly strict environmental regulations. This compliance reduces the administrative burden and costs associated with environmental permitting and monitoring. The overall green chemistry profile of the method enhances the corporate sustainability image of manufacturers, which is becoming a key differentiator in global supply chains.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and advantageous effects described in the patent documentation. They provide clarity on the operational requirements and potential benefits for stakeholders considering this route for their supply chain. Understanding these details is essential for making informed decisions about process adoption and vendor selection. The information below reflects the current state of the art as disclosed in the intellectual property.
Q: What are the advantages of the pyrimidine directing group in this synthesis?
A: The 2-chloro-5-nitropyrimidine directing group allows for mild introduction and removal conditions, avoiding the harsh active metals and high temperatures typically required in traditional methods.
Q: Is this process suitable for large-scale manufacturing?
A: Yes, the patent demonstrates scalability from gram-level to multi-gram batches with consistent yields, utilizing standard solvents and commercially available catalysts suitable for industrial adaptation.
Q: How does this method impact impurity profiles?
A: The high regioselectivity of the C-H activation step minimizes side reactions, resulting in a cleaner crude product that simplifies downstream purification and ensures high-purity specifications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable O-Aminophenol Derivatives Supplier
NINGBO INNO PHARMCHEM stands at the forefront of chemical innovation, possessing extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our technical team is uniquely qualified to adapt the advanced C-H activation techniques described in patent CN108148005B to meet your specific volume requirements. We maintain stringent purity specifications across all our product lines, supported by rigorous QC labs that ensure every shipment meets the highest industry standards. Our commitment to quality and consistency makes us the preferred partner for multinational corporations seeking a dependable source of complex pharmaceutical intermediates. We understand the critical nature of your supply chain and are dedicated to providing solutions that enhance your operational efficiency.
We invite you to engage with our technical procurement team to discuss how we can support your project goals. Request a Customized Cost-Saving Analysis to evaluate the economic benefits of switching to this advanced synthesis route. Our experts are ready to provide specific COA data and route feasibility assessments tailored to your unique molecular targets. By collaborating with us, you gain access to a wealth of chemical expertise and manufacturing capacity that can accelerate your development timelines. Contact us today to initiate a conversation about optimizing your supply chain with high-performance o-aminophenol derivatives.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
