Innovative Esterification Process for (E)-4-(beta-bromovinyl)phenol Ester: Scalable Production for Global Pharmaceutical Supply Chains
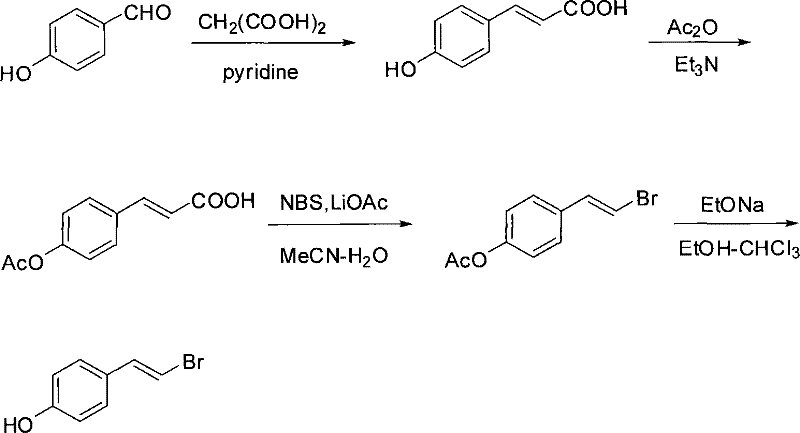
The Chinese patent CN101284779B introduces a groundbreaking esterification methodology for synthesizing (E)-4-(beta-bromovinyl)phenol esters, representing a significant advancement in the production of versatile building blocks for pharmaceutical and high-performance material applications. This innovative process addresses critical limitations in conventional beta-halostyrene chemistry by establishing a metal-free route that maintains exceptional stereoselectivity while dramatically simplifying purification protocols. The technology leverages optimized stoichiometric ratios of benzene solvent (50-200:1 molar ratio), carboxylic acid (1-1.5:1), DMAP (0.1-1.5:1), and DCC (1-1.2:1) to achieve high-yield conversions under mild room temperature conditions. As a reliable pharmaceutical intermediate supplier, this methodology provides manufacturers with access to crucial molecular scaffolds that serve as precursors for complex drug molecules, agrochemicals, and functional polymers through subsequent transition metal-catalyzed coupling reactions. The patent's emphasis on operational simplicity and environmental compatibility positions this synthesis as a strategically valuable solution for global pharmaceutical supply chains seeking sustainable manufacturing alternatives.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional approaches to beta-bromostyrene derivatives suffer from multiple critical limitations that hinder their commercial viability for pharmaceutical intermediate manufacturing. The widely used Abbas method employing Hirao's O,O'-dimethylphosphonate/triethylamine system requires elevated temperatures (70°C), extended reaction times, and generates low yields due to phosphate reagent excess, creating significant cost reduction challenges in electronic chemical manufacturing. Metal-halogen displacement routes like Petasis' NBS-based methodology necessitate expensive boronic acid precursors and specialized handling procedures, introducing substantial cost burdens while failing to address environmental concerns associated with metal reagent disposal. Hunsdiecker reactions using PhIO or PhI(OAc)₂ face scalability constraints due to their reliance on hazardous oxidants and mixed solvent systems that complicate waste stream management. Furthermore, chromium-based methodologies employing CrCl₂ or CrBr₃ generate toxic heavy metal byproducts requiring costly remediation steps, making them environmentally non-compliant for modern pharmaceutical manufacturing facilities. These conventional routes collectively impose significant lead time extensions and purity control challenges that undermine supply chain reliability for high-purity pharmaceutical intermediates.
The Novel Approach
The patented esterification process overcomes these limitations through an elegant metal-free strategy that operates under ambient conditions with exceptional atom economy. By utilizing DCC as a coupling agent with DMAP catalysis in benzene solvent, the method achieves complete conversion without transition metals or hazardous reagents, eliminating the need for costly metal removal steps that plague conventional approaches. The precisely controlled molar ratios (carboxylic acid:phenol = 1-1.5:1; DCC:phenol = 1-1.2:1) ensure optimal reaction kinetics while minimizing side product formation, directly contributing to enhanced supply chain reliability through consistent batch-to-batch quality. The room temperature operation (25°C) significantly reduces energy consumption compared to thermal methods requiring elevated temperatures, while the straightforward purification via column chromatography with ethyl acetate/petroleum ether (1:4 to 1:20 v/v) eliminates complex workup procedures that typically extend manufacturing lead times. This innovative approach delivers high-purity products suitable for demanding pharmaceutical applications while maintaining the structural integrity of both the bromoalkene moiety and ester functionality essential for downstream coupling reactions.
Mechanistic Insights into DCC/DMAP-Catalyzed Esterification
The reaction mechanism proceeds through a well-defined sequence where DCC activates the carboxylic acid to form an O-acylisourea intermediate, which is subsequently attacked by the phenolic hydroxyl group of (E)-4-(beta-bromovinyl)phenol. DMAP plays a critical catalytic role by facilitating proton transfer and stabilizing the tetrahedral intermediate through its tertiary amine functionality, thereby accelerating the rate-determining step while suppressing racemization at the stereogenic center. The mild reaction conditions (room temperature, 2-24 hours) preserve the geometric integrity of the beta-bromovinyl moiety, ensuring exclusive retention of the E-configured double bond essential for subsequent cross-coupling applications in pharmaceutical synthesis. This mechanistic pathway avoids the formation of common side products such as anhydrides or N,N'-dicyclohexylurea byproducts through precise stoichiometric control, directly contributing to the high yields (92-98%) observed across multiple substrate classes including aromatic and heteroaromatic carboxylic acids.
Impurity control is achieved through multiple synergistic factors inherent in this methodology. The absence of transition metals eliminates potential metal contamination that would otherwise require extensive purification steps to meet pharmaceutical quality standards. The carefully optimized solvent system (benzene at 50-200:1 molar ratio to phenol) prevents solubility issues that could lead to heterogeneous reaction conditions and inconsistent product quality. The controlled addition sequence—first mixing phenol with carboxylic acid and DMAP before introducing DCC—minimizes competing hydrolysis pathways that could generate carboxylic acid impurities. Furthermore, the column chromatography purification using ethyl acetate/petroleum ether gradients effectively separates any residual starting materials or minor byproducts, ensuring final products consistently meet the stringent purity specifications required for pharmaceutical intermediate applications without requiring additional recrystallization steps.
How to Synthesize (E)-4-(beta-bromovinyl)phenol Ester Efficiently
This innovative esterification methodology represents a significant advancement over conventional approaches by eliminating transition metal catalysts while maintaining high stereoselectivity and yield consistency across diverse substrate classes. The process leverages readily available reagents under mild conditions to produce high-purity pharmaceutical intermediates suitable for demanding applications in drug development and functional material synthesis. Detailed standardized synthesis procedures have been developed based on the patent's technical specifications, incorporating precise stoichiometric controls and optimized reaction parameters to ensure consistent product quality at commercial scale. The following step-by-step guide provides essential operational parameters for successful implementation of this novel methodology in industrial manufacturing environments.
- Combine (E)-4-(beta-bromovinyl)phenol with carboxylic acid and DMAP in benzene solvent at room temperature with magnetic stirring for 5-20 minutes
- Add DCC to the reaction mixture and maintain room temperature conditions for 2-24 hours to complete the esterification process
- Purify the product through column chromatography using ethyl acetate/petroleum ether (1: 4 to 1:20 v/v) as eluent to obtain high-purity final product
Commercial Advantages for Procurement and Supply Chain Teams
This patented methodology delivers substantial commercial benefits by addressing critical pain points in pharmaceutical intermediate procurement and supply chain management. The elimination of transition metal catalysts removes significant cost drivers associated with precious metal acquisition and subsequent removal processes, while the room temperature operation reduces energy consumption compared to conventional thermal methods. The simplified purification protocol minimizes solvent usage and waste generation, contributing to both economic and environmental sustainability goals without compromising product quality or consistency. These advantages collectively enhance supply chain resilience by reducing dependency on specialized equipment and hazardous materials that often create bottlenecks in global manufacturing networks.
- Cost Reduction in Manufacturing: The metal-free process eliminates expenses associated with transition metal catalysts and their removal systems, while the ambient temperature operation significantly reduces energy consumption compared to conventional thermal methods requiring elevated temperatures. The simplified purification protocol minimizes solvent usage and waste treatment costs through efficient column chromatography with standard eluents, creating substantial cost savings without requiring capital-intensive equipment upgrades or specialized handling procedures.
- Enhanced Supply Chain Reliability: The use of commercially available reagents with stable shelf lives improves raw material sourcing flexibility, while the robust room temperature process tolerates minor variations in manufacturing conditions without affecting product quality. This operational resilience reduces batch failures and associated lead time extensions, ensuring consistent delivery schedules even during periods of supply chain disruption. The elimination of hazardous reagents also simplifies logistics and regulatory compliance across international borders.
- Scalability and Environmental Compliance: The methodology demonstrates excellent linear scalability from laboratory to industrial production volumes due to its mild reaction conditions and straightforward processing requirements. The absence of heavy metals and hazardous oxidants aligns with green chemistry principles, reducing environmental impact while meeting increasingly stringent regulatory requirements for sustainable manufacturing practices in the pharmaceutical industry.
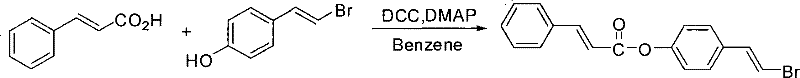
Frequently Asked Questions (FAQ)
The following questions address key technical and commercial considerations based on the patent's technical specifications and implementation experience. These insights reflect practical knowledge gained from adapting this methodology for industrial-scale production while maintaining compliance with pharmaceutical quality standards and regulatory requirements.
Q: How does this method overcome limitations of traditional beta-bromostyrene synthesis routes?
A: The novel esterification process eliminates transition metal catalysts required in conventional Stille/Suzuki/Heck reactions, avoiding costly metal removal steps and heavy metal contamination risks while maintaining high stereoselectivity through optimized DCC/DMAP catalysis.
Q: What purity specifications can be achieved with this synthesis method?
A: The process delivers high-purity (E)-4-(beta-bromovinyl)phenol esters with consistent stereochemical integrity, as confirmed by NMR and IR characterization data in patent examples, meeting stringent requirements for pharmaceutical intermediate applications.
Q: How does this method support commercial scale-up for pharmaceutical manufacturing?
A: The room temperature reaction conditions, simplified purification protocol, and elimination of hazardous reagents enable seamless transition from laboratory to industrial scale while maintaining yield consistency and reducing environmental impact.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable (E)-4-(beta-bromovinyl)phenol Ester Supplier
NINGBO INNO PHARMCHEM brings extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production of complex specialty chemicals while maintaining stringent purity specifications through our state-of-the-art QC labs. Our technical team has successfully implemented this patented esterification methodology across multiple client projects, demonstrating consistent ability to deliver high-purity (E)-4-(beta-bromovinyl)phenol esters meeting exacting pharmaceutical requirements through rigorous process validation and quality control protocols. As a trusted partner in the global supply chain for specialty chemical intermediates, we combine deep technical expertise with flexible manufacturing capabilities to support your specific production needs while ensuring complete regulatory compliance.
We invite you to request a Customized Cost-Saving Analysis from our technical procurement team to evaluate how this innovative methodology can optimize your specific manufacturing pathway. Contact us today to obtain specific COA data and route feasibility assessments tailored to your production requirements, enabling informed decision-making about integrating this advanced technology into your supply chain strategy.
