Advanced Pd-Catalyzed Synthesis of Polysubstituted Benzodihydrofuran Heterocycles for Commercial Scale-Up
The landscape of heterocyclic chemistry is constantly evolving, driven by the relentless demand for novel scaffolds in pharmaceutical and agrochemical applications. A significant breakthrough in this domain is detailed in patent CN111205279B, which discloses a highly efficient preparation method for polysubstituted benzodihydrofuran heterocyclic compounds. These structures are pivotal motifs found in numerous bioactive molecules, serving as core frameworks for drugs targeting various therapeutic areas, including 5-HT4 receptor agonists and IMPDH inhibitors. The patented technology introduces a robust palladium-catalyzed intermolecular ene-yne cross-coupling strategy that fundamentally shifts the paradigm from traditional, often cumbersome synthetic routes to a more streamlined and economically viable process. By leveraging simple and commercially accessible starting materials, this innovation addresses critical bottlenecks in the supply chain of complex fine chemical intermediates, offering a pathway to high-purity products with exceptional structural diversity.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of chroman and dihydrobenzofuran skeletons has relied heavily on transition metal-catalyzed nucleophilic substitutions, hydroalkylations, or intramolecular Heck cyclizations. While these methods have served the industry for decades, they are increasingly viewed as suboptimal for modern manufacturing standards due to several inherent drawbacks. Conventional protocols frequently necessitate harsh reaction conditions, including excessive temperatures that can degrade sensitive functional groups or lead to unwanted side reactions. Furthermore, many established routes suffer from limited substrate applicability, meaning that slight modifications to the molecular structure often require a complete re-optimization of the process, thereby inflating R&D costs and extending development timelines. The reliance on specialized precursors that are difficult to source or synthesize further complicates the supply chain, creating vulnerabilities in the production of high-value pharmaceutical intermediates.
The Novel Approach
In stark contrast to these legacy techniques, the method described in CN111205279B utilizes a sophisticated yet operationally simple palladium-catalyzed cross-coupling reaction between alkynone oxime ethers and o-iodophenyl alkenyl ethers. This innovative route proceeds under remarkably mild conditions, typically requiring temperatures between 50°C and 70°C, which significantly reduces energy consumption and thermal stress on the reaction mixture. The versatility of this approach is exemplified by its broad substrate scope, accommodating a wide array of substituents such as phenyl, naphthyl, and various halogenated or alkoxy groups without compromising yield or purity. As illustrated in the general reaction scheme below, the transformation efficiently constructs the complex benzodihydrofuran core in a single pot, demonstrating high step economy and eliminating the need for multiple isolation steps that traditionally erode overall process efficiency.

Mechanistic Insights into Pd-Catalyzed Oxidative Cyclization
The success of this synthetic methodology lies in the intricate interplay between the palladium catalyst, the copper oxidant, and the specific electronic properties of the substrates. The reaction mechanism initiates with the oxidative addition of the palladium species to the aryl iodide bond of the o-iodophenyl alkenyl ether, generating a reactive aryl-palladium intermediate. This species then undergoes a migratory insertion into the electron-rich olefinic ether moiety, a critical step that sets the stereochemical and regiochemical course of the cyclization. Subsequently, the alkynone oxime ether participates in the catalytic cycle, where the oxime ether functionality acts as an internal oxidant or directing group, facilitating the final ring closure and mass decomposition to release the target heterocycle. This concerted sequence ensures that the reaction proceeds with high atom economy, minimizing the formation of heavy metal waste and maximizing the conversion of valuable starting materials into the desired product.
From a quality control perspective, the mechanistic pathway offers distinct advantages regarding impurity profiles. The use of copper chloride as a co-oxidant and tetrabutylammonium bromide as an additive creates a highly selective environment that suppresses common side reactions such as homocoupling or polymerization of the alkyne components. The tolerance for diverse functional groups, including methoxy, chloro, and alkyl chains, indicates that the catalytic system is robust against electronic variations in the substrate. This resilience is crucial for manufacturing, as it implies that minor fluctuations in raw material quality will not catastrophicly impact the final product specification. The resulting polysubstituted benzodihydrofuran scaffold, shown below with its variable R groups, can be precisely tuned to meet specific biological activity requirements while maintaining a consistent and clean chemical identity suitable for downstream drug development.
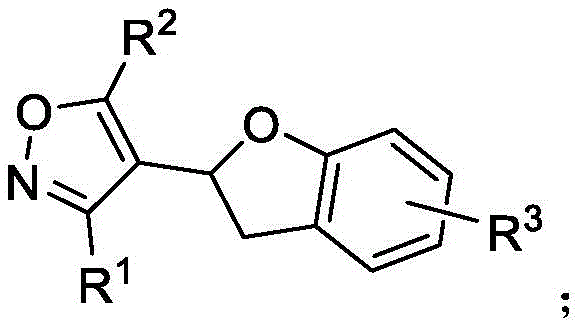
How to Synthesize Polysubstituted Benzodihydrofuran Efficiently
Implementing this synthesis in a production environment requires strict adherence to the optimized parameters defined in the patent to ensure reproducibility and safety. The process begins with the precise charging of the catalytic system, followed by the controlled addition of substrates in an organic solvent medium. The reaction is maintained under agitation for a defined period to allow for complete conversion before proceeding to a standard aqueous workup. This operational simplicity belies the chemical sophistication of the transformation, offering a practical blueprint for chemists aiming to access these valuable heterocycles. For a comprehensive breakdown of the specific molar ratios, temperature controls, and purification techniques required to achieve optimal results, please refer to the standardized synthesis protocol outlined below.
- Charge a reactor with palladium acetate catalyst, copper chloride oxidant, tetrabutylammonium bromide additive, and potassium carbonate base.
- Add alkynone oxime ether and o-iodophenyl alkenyl ether substrates dissolved in tetrahydrofuran (THF) solvent to the reaction mixture.
- Stir the reaction at 50-70°C for 10-16 hours, then cool, extract with ethyl acetate, and purify via chromatography to isolate the target heterocycle.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented synthesis route translates into tangible strategic benefits that extend far beyond the laboratory bench. The shift towards using readily available alkynone oxime ethers and o-iodophenyl alkenyl ethers as feedstocks mitigates the risk of raw material shortages that often plague the production of exotic intermediates. By simplifying the synthetic sequence and reducing the number of unit operations, manufacturers can significantly lower the overall cost of goods sold (COGS) while simultaneously improving throughput. The mild reaction conditions also imply reduced energy overheads and less stringent requirements for reactor metallurgy, allowing for the utilization of existing infrastructure without the need for costly capital expenditures on specialized high-pressure or high-temperature equipment.
- Cost Reduction in Manufacturing: The elimination of harsh reaction conditions and the use of common, inexpensive reagents like potassium carbonate and copper chloride drive down the direct material costs associated with production. Furthermore, the high step economy means fewer solvent exchanges and purification cycles, which drastically reduces solvent consumption and waste disposal fees. This lean manufacturing approach ensures that the final price point for these high-purity pharmaceutical intermediates remains competitive in the global market, providing a clear margin advantage for downstream drug manufacturers.
- Enhanced Supply Chain Reliability: Sourcing stability is a critical concern for any long-term production campaign, and this method addresses it by relying on commodity chemicals rather than bespoke, hard-to-find reagents. The robustness of the catalytic system against functional group variations means that supply chain disruptions for one specific substituted precursor can often be managed by switching to alternative analogues without halting the entire production line. This flexibility ensures a continuous flow of materials, reducing lead times and preventing stock-outs that could delay critical clinical or commercial programs.
- Scalability and Environmental Compliance: The process is inherently designed for scale-up, utilizing tetrahydrofuran (THF) as a solvent, which is well-understood in industrial settings and easily recovered for reuse. The absence of toxic heavy metals in the final product, thanks to efficient purification protocols, simplifies regulatory compliance and reduces the burden on environmental health and safety teams. As the industry moves towards greener chemistry practices, this method's high atom efficiency and mild operating parameters position it as a sustainable choice for the commercial scale-up of complex fine chemicals.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this benzodihydrofuran synthesis technology. These insights are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing a transparent view of the process capabilities. Understanding these details is essential for technical teams evaluating the feasibility of integrating this route into their existing manufacturing portfolios.
Q: What are the advantages of this Pd-catalyzed method over traditional Heck cyclization?
A: Unlike traditional methods that often require harsh temperatures or limited substrate scopes, this novel approach operates under mild conditions (50-70°C) with excellent functional group tolerance and high step economy.
Q: What represents the key starting materials for this synthesis?
A: The process utilizes readily available alkynone oxime ethers and o-iodophenyl alkenyl ethers, which allows for significant diversification of the R1, R2, and R3 substituents on the final benzodihydrofuran scaffold.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the method uses common solvents like THF and standard inorganic bases, avoiding exotic reagents, which facilitates straightforward scale-up from laboratory to commercial tonnage production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Polysubstituted Benzodihydrofuran Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that advanced heterocyclic intermediates play in the development of next-generation therapeutics. Our team of expert process chemists has thoroughly analyzed the technology disclosed in CN111205279B and is fully prepared to translate this academic innovation into commercial reality. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project can move seamlessly from pilot plant to full-scale manufacturing. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of polysubstituted benzodihydrofuran delivered meets the highest international standards for pharmaceutical applications.
We invite you to collaborate with us to leverage this cutting-edge synthesis for your specific drug discovery or process development needs. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your volume requirements, along with specific COA data and route feasibility assessments. Let us help you optimize your supply chain and accelerate your time-to-market with our reliable, high-quality chemical solutions.
