Scalable Production of High-Purity QTFMT Triazoles for Global Pharmaceutical Supply Chain Excellence
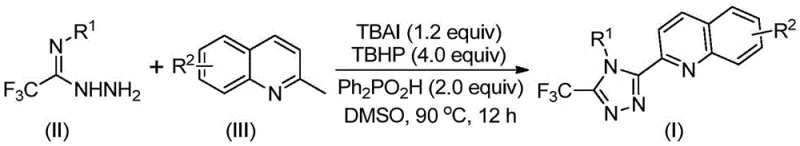
Patent CN113307790B introduces a groundbreaking methodology for synthesizing structurally diverse 3-quinolyl-5-trifluoromethyl substituted 1,2,4-triazole compounds through an innovative oxidative cyclization process that fundamentally redefines conventional synthetic approaches in heterocyclic chemistry. This patented technique leverages commercially accessible starting materials including tetrabutylammonium iodide and tert-butyl peroxide to facilitate a one-pot transformation that eliminates multiple intermediate steps while maintaining high regioselectivity and yield consistency across various substrate combinations. The elimination of stringent anhydrous and anaerobic reaction environments represents a significant operational advancement that reduces equipment complexity and enhances process robustness for industrial-scale manufacturing facilities worldwide. Furthermore, the complete avoidance of transition metal catalysts addresses critical regulatory concerns regarding heavy metal contamination in pharmaceutical intermediates while simultaneously lowering purification costs through simplified downstream processing workflows. This strategic innovation not only expands the synthetic toolbox available to medicinal chemists but also establishes new benchmarks for sustainable manufacturing practices within the fine chemical industry by integrating green chemistry principles into complex heterocycle synthesis.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthesis routes for quinolinyl-substituted triazoles typically rely on quinoline-2-formic acid as starting material requiring five sequential reaction steps under severe thermal conditions that demand specialized equipment and generate significant waste streams through multiple intermediate isolations. These multi-step processes suffer from cumulative yield losses resulting in unacceptably low overall yields around seventeen percent which render them economically unviable for commercial scale-up despite their theoretical utility in producing bioactive molecular frameworks. The stringent requirement for anhydrous and anaerobic environments introduces substantial operational complexity including specialized glovebox systems and continuous nitrogen purging that significantly increase capital expenditure while creating vulnerability points in production continuity due to moisture sensitivity issues during scale-up operations. Additionally, conventional methods often employ transition metal catalysts that necessitate expensive removal protocols to meet pharmaceutical purity standards while generating hazardous waste streams requiring costly disposal procedures that conflict with modern environmental sustainability objectives across global manufacturing facilities.
The Novel Approach
The patented methodology presented in CN113307790B revolutionizes this synthetic landscape by implementing a direct oxidative cyclization pathway using readily available starting materials including substituted quinolines and trifluoroethylimine hydrazides under mild thermal conditions between eighty to one hundred degrees Celsius without requiring inert atmosphere protection or specialized moisture control systems. This one-pot transformation leverages tetrabutylammonium iodide as an iodide source combined with tert-butyl peroxide as oxidant to facilitate radical-mediated conversion of methylquinoline precursors into reactive aldehydes that subsequently undergo condensation and cyclization with hydrazide components through carefully orchestrated mechanistic pathways. The elimination of transition metal catalysts represents a paradigm shift that removes both capital costs associated with precious metal procurement and operational expenses related to metal residue removal while simultaneously enhancing product purity profiles essential for pharmaceutical applications. Furthermore this approach demonstrates exceptional substrate tolerance allowing diverse functional group substitutions at multiple positions which significantly broadens its applicability across various therapeutic compound classes while maintaining consistent high yields across different structural variants.
Mechanistic Insights into Oxidative Cyclization with TBAI/TBHP
The reaction mechanism proceeds through a sophisticated radical-mediated pathway initiated by tetrabutylammonium iodide activation of tert-butyl peroxide which generates reactive iodine species that oxidize the methyl group of quinoline precursors into aldehyde intermediates under mild thermal conditions. This aldehyde then undergoes condensation with trifluoroethylimine hydrazide components forming dehydrated hydrazone intermediates that subsequently experience oxidative iodination followed by intramolecular electrophilic substitution reactions leading to ring closure through aromatization processes that form the characteristic triazole core structure. The diphenyl phosphoric acid additive plays a critical role in proton management throughout this cascade by facilitating key dehydration steps while maintaining optimal pH conditions that prevent undesired side reactions such as hydrolysis or decomposition of sensitive intermediates under thermal stress conditions. This carefully balanced mechanistic sequence operates efficiently within DMSO solvent systems which provide ideal polarity characteristics to stabilize radical species while solubilizing both organic precursors and inorganic additives throughout the entire reaction continuum without phase separation issues.
Impurity control is achieved through multiple built-in mechanistic safeguards including precise stoichiometric control of iodide promoter which prevents over-halogenation side products while the mild thermal profile between eighty to one hundred degrees Celsius minimizes thermal degradation pathways common in conventional high-energy processes. The absence of transition metals eliminates potential metal-catalyzed decomposition routes that typically generate complex impurity profiles requiring extensive purification efforts while the one-pot nature of this transformation reduces intermediate handling that often introduces contamination risks during multi-step syntheses. Substrate design flexibility allows strategic placement of electron-donating or electron-withdrawing groups that can be optimized to suppress specific impurity formation pathways through electronic effects on reactive intermediates while maintaining high regioselectivity during cyclization steps. Rigorous post-reaction processing through silica gel filtration followed by standard column chromatography provides additional purification leverage points that effectively remove any residual starting materials or minor side products ensuring final products consistently meet stringent pharmaceutical purity specifications required by global regulatory authorities.
How to Synthesize QTFMT Triazoles Efficiently
This innovative synthesis route represents a significant advancement over traditional methodologies by enabling direct conversion of commercially available precursors into high-value triazole compounds through carefully optimized oxidative cyclization parameters that eliminate multiple intermediate isolation steps while maintaining exceptional yield consistency across diverse structural variants as demonstrated in fifteen experimental examples within the patent documentation. The process operates effectively under ambient atmospheric conditions without requiring specialized inert gas handling equipment which substantially reduces operational complexity while enhancing process robustness during scale-up transitions from laboratory to manufacturing environments where environmental control systems may experience fluctuations during continuous production runs.
- Combine tetrabutylammonium iodide (TBAI), tert-butyl peroxide aqueous solution (TBHP), diphenyl phosphoric acid (DPPA), trifluoroethylimine hydrazide (II), and substituted quinoline precursor (III) in DMSO solvent under ambient atmospheric conditions without requiring specialized inert gas handling equipment.
- Heat the homogeneous reaction mixture to precisely controlled temperatures between 80–90°C and maintain thermal stability for extended periods of approximately twelve hours to ensure complete oxidative cyclization through radical-mediated mechanisms.
- Execute straightforward post-reaction processing by direct filtration through silica gel followed by standard column chromatography purification to isolate high-purity QTFMT triazole products without complex intermediate isolation steps.
Commercial Advantages for Procurement and Supply Chain Teams
This patented methodology delivers substantial strategic value across procurement and supply chain operations by addressing critical pain points inherent in traditional triazole synthesis routes through fundamental process innovations that enhance both economic viability and operational reliability while maintaining strict adherence to pharmaceutical quality standards required by global regulatory frameworks.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts removes associated procurement costs while simultaneously eliminating complex metal removal processes that typically require specialized equipment and generate hazardous waste streams requiring costly disposal protocols which collectively represent significant operational savings without compromising product quality or regulatory compliance standards.
- Enhanced Supply Chain Reliability: Utilizing globally available starting materials including commercially sourced quinoline derivatives and trifluoroacetic acid-based precursors ensures consistent raw material availability while avoiding supply chain bottlenecks associated with specialized reagents required by conventional methods thereby strengthening procurement resilience against market volatility.
- Scalability and Environmental Compliance: The straightforward reaction setup operating under ambient atmospheric conditions enables seamless scale-up from laboratory to industrial production volumes while generating minimal hazardous waste streams through simplified processing workflows that align with modern environmental sustainability objectives across global manufacturing facilities.
Frequently Asked Questions (FAQ)
The following technical questions address critical commercial considerations based on detailed analysis of patent CN113307790B's experimental data and mechanistic insights which provide essential decision-making information for procurement teams evaluating this innovative synthesis route against conventional alternatives.
Q: How does this method overcome limitations of traditional multi-step synthesis routes?
A: The patented process eliminates five-step conventional routes requiring harsh conditions by enabling direct one-pot conversion using commercially available starting materials under mild thermal conditions without anhydrous or anaerobic requirements while achieving significantly higher yields through optimized oxidative cyclization.
Q: Why is avoiding heavy metal catalysts critical for commercial production?
A: Eliminating toxic transition metals removes costly purification steps for metal residue removal while reducing environmental compliance risks and aligning with green chemistry principles essential for pharmaceutical manufacturing quality standards.
Q: What ensures scalability and supply chain reliability for this synthesis?
A: The use of inexpensive globally available raw materials combined with simple reaction setup enables seamless scale-up from laboratory to industrial production volumes while maintaining consistent quality through robust process parameters.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable QTFMT Triazole Supplier
Our company possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production while maintaining stringent purity specifications through rigorous QC labs equipped with advanced analytical instrumentation capable of detecting trace impurities at parts-per-billion levels essential for pharmaceutical applications requiring high-purity intermediates meeting global regulatory standards including ICH Q7 guidelines.
We invite you to request our Customized Cost-Saving Analysis which details specific implementation pathways tailored to your production requirements; contact our technical procurement team today to obtain specific COA data and route feasibility assessments demonstrating how this patented methodology can enhance your supply chain resilience while delivering superior quality performance metrics.
