Advanced Manufacturing of Varlitinib Intermediates via Green Amidation and Cyclization
Advanced Manufacturing of Varlitinib Intermediates via Green Amidation and Cyclization
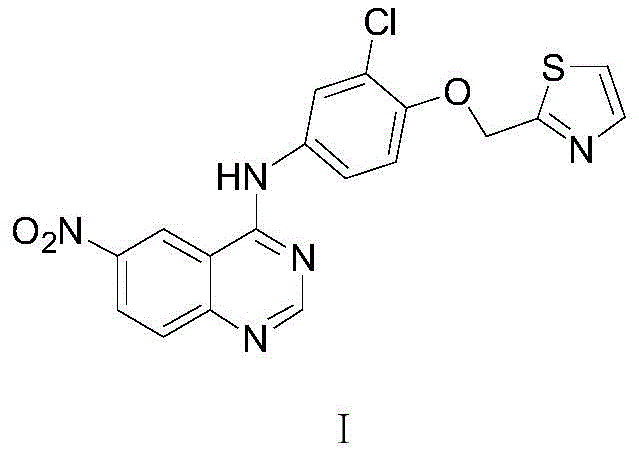
The pharmaceutical industry continuously seeks robust, scalable, and environmentally sustainable pathways for the production of complex kinase inhibitors. A pivotal development in this domain is disclosed in patent CN110577520B, which details a novel preparation method for 6-nitro-4-substituted amino quinazoline derivatives. This specific compound serves as a critical advanced intermediate in the synthesis of Varlitinib, a potent pan-HER inhibitor currently under development for various oncology indications including biliary tract and gastric cancers. The significance of this patent lies not merely in the chemical transformation itself, but in its strategic departure from legacy synthetic routes that have historically plagued manufacturers with high costs, toxic byproducts, and operational complexity. By establishing a streamlined two-step sequence involving Lewis acid catalyzed amidation followed by a one-pot cyclization, this technology offers a compelling value proposition for reliable pharmaceutical intermediate suppliers aiming to secure long-term contracts with global innovator companies.
For R&D directors and process chemists, the technical elegance of this route is immediately apparent. Traditional methods often rely on the reduction of nitro groups using stoichiometric amounts of iron powder, a practice that generates massive quantities of solid waste and complicates downstream purification. In stark contrast, the methodology outlined in CN110577520B leverages the intrinsic reactivity of 2-halo-5-nitrobenzoates, activated by electron-withdrawing nitro groups, to facilitate highly selective nucleophilic attacks. This approach eliminates the need for heavy metal reductants in the early stages and avoids the use of hazardous coupling reagents like carbonyldiimidazole (CDI) which can release toxic gases. The result is a process that delivers exceptional purity profiles, with liquid phase purity consistently exceeding 99.6%, thereby setting a new benchmark for high-purity pharmaceutical intermediates in the competitive oncology market.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
To fully appreciate the innovation presented in this patent, one must critically analyze the deficiencies of the prior art synthetic strategies for Varlitinib intermediates. Historical routes, such as those disclosed by the original research entities, often commence with 6-amino-4-substituted aminoquinazolines, necessitating the use of expensive and unstable starting materials. For instance, one prominent legacy route involves condensation with sulfo-CDI, a reaction that unfortunately generates hydrogen sulfide gas as a byproduct. From an industrial hygiene and environmental perspective, the evolution of H2S presents a severe liability, requiring specialized scrubbing equipment and rigorous safety protocols that drastically inflate capital expenditure. Furthermore, alternative literature methods describe a seven-step synthesis initiating from 2-chloro-4-nitrophenol, which suffers from a dismal overall yield of approximately 18.2%. This multi-step sequence relies heavily on iron powder reductions, creating substantial volumes of iron sludge that are costly to dispose of and environmentally damaging. Additionally, the Dimroth rearrangement steps often utilized in these older pathways are prone to forming isomeric side products, complicating the purification landscape and threatening the stringent impurity specifications required for clinical-grade materials.
The Novel Approach
The patented process fundamentally reengineers the synthetic logic by reversing the construction order and utilizing more robust building blocks. Instead of building the quinazoline ring first and then functionalizing it, this method constructs the core through a direct cyclization of a pre-formed amide. The process initiates with the amidation of readily available 2-halo-5-nitrobenzoates with 4-(thiazol-2-yl)methoxy-3-chloroaniline. This step is catalyzed by inexpensive Lewis acids such as ammonium chloride or zinc chloride, achieving yields greater than 96%. The subsequent transformation involves a clever one-pot substitution and condensation reaction with formamidine salts. By carefully controlling the temperature profile—initially maintaining mild conditions for substitution before ramping up for dehydration condensation—the process achieves a remarkable yield of over 90% for the cyclization step. This convergence of high yield, minimal waste, and operational simplicity represents a paradigm shift in cost reduction in pharmaceutical intermediates manufacturing, offering a viable path for commercial scale-up of complex quinazoline derivatives.
Mechanistic Insights into Lewis Acid Catalyzed Amidation and Cyclization
The success of this synthetic route hinges on a deep understanding of the electronic effects governing the reactivity of the nitro-substituted benzamide intermediate. In the first step, the presence of the nitro group at the 5-position of the benzoate ring significantly enhances the electrophilicity of the ester carbonyl carbon. When a Lewis acid catalyst, such as zinc chloride or ammonium chloride, is introduced into the reaction mixture, it coordinates with the carbonyl oxygen, further polarizing the bond and making it more susceptible to nucleophilic attack by the aniline nitrogen. This catalytic activation allows the amidation to proceed efficiently at moderate temperatures (90-120°C) without the need for harsh activating agents. Crucially, the solvent system, typically comprising toluene or xylene, facilitates the continuous removal of the alcohol byproduct (methanol or ethanol) via azeotropic distillation. This drives the equilibrium towards the product side according to Le Chatelier's principle, ensuring near-quantitative conversion and minimizing the formation of hydrolysis byproducts that could compromise the purity of the intermediate amide.
In the second stage, the mechanism shifts to an intramolecular nucleophilic aromatic substitution followed by cyclodehydration. The nitro group, now positioned ortho to the halogen atom on the benzamide ring, exerts a powerful electron-withdrawing effect that activates the carbon-halogen bond towards nucleophilic displacement. Upon addition of the formamidine salt and a base like sodium methoxide, the nitrogen of the formamidine moiety attacks the activated aryl halide position, displacing the halide ion. The resulting intermediate then undergoes an intramolecular condensation where the amide nitrogen attacks the imine carbon of the formamidine group. The patent highlights the importance of a staged temperature protocol; performing the substitution at lower temperatures (50-55°C) prevents side reactions, while the subsequent heating to 105-110°C drives the dehydration necessary to aromatize the pyrimidine ring. The concurrent distillation of low-boiling substances like methanol and water is critical here, as their presence at high temperatures could lead to the decomposition of the sensitive thiazole ring, a degradation pathway that this process meticulously avoids to maintain product integrity.
How to Synthesize 6-Nitro-4-Substituted Amino Quinazoline Efficiently
Implementing this synthesis requires precise control over reaction parameters to maximize the benefits of the patented methodology. The process is designed to be operationally straightforward, utilizing standard reactor configurations found in most fine chemical facilities. The initial amidation step sets the foundation for high purity, requiring careful monitoring of water content and efficient solvent recovery. Following the isolation of the amide intermediate, the one-pot cyclization demands strict adherence to the temperature gradient and vacuum distillation protocols to ensure the complete removal of volatiles that could otherwise degrade the thiazole moiety. For process engineers looking to translate this laboratory-scale success to production, the detailed standardized synthesis steps provided below outline the critical operational windows and workup procedures necessary to achieve the reported yields and purity specifications.
- Perform amidation reaction between 2-halo-5-nitrobenzoate and 4-(thiazol-2-yl)methoxy-3-chloroaniline using a Lewis acid catalyst in toluene or xylene at 90-120°C.
- Isolate the intermediate N-[4-(thiazol-2-yl)methoxy-3-chlorophenyl]-2-halo-5-nitrobenzamide via crystallization.
- Execute one-pot substitution and condensation with formamidine salt and base in DMF, distilling off low-boiling substances to yield the final quinazoline derivative.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain executives, the adoption of this synthetic route translates directly into tangible risk mitigation and cost optimization strategies. The shift away from multi-step sequences involving hazardous reagents simplifies the supply chain by reducing the number of distinct raw materials that need to be sourced and qualified. By eliminating the need for iron powder and the associated waste disposal logistics, manufacturers can significantly reduce their environmental compliance overhead. Furthermore, the high selectivity of the reaction minimizes the formation of difficult-to-remove impurities, which reduces the consumption of solvents and adsorbents during the purification phase. This efficiency gain is crucial for maintaining healthy margins in the competitive generic and contract manufacturing sectors, where reducing lead time for high-purity pharmaceutical intermediates is often a key differentiator in securing supply agreements.
- Cost Reduction in Manufacturing: The economic advantages of this process are driven primarily by the utilization of commodity chemicals and the elimination of expensive coupling reagents. Traditional routes often rely on specialized activators like CDI or carbodiimides, which carry a high price tag and require careful handling. By substituting these with inexpensive Lewis acid catalysts and formamidine salts, the direct material cost of goods sold (COGS) is substantially lowered. Additionally, the high yields achieved in both the amidation (>96%) and cyclization (>90%) steps mean that less raw material is wasted per kilogram of final product. The ability to recover and recycle solvents like toluene and DMF further enhances the economic viability, creating a lean manufacturing process that is resilient to fluctuations in raw material pricing.
- Enhanced Supply Chain Reliability: Supply continuity is often threatened by the reliance on niche reagents that have limited global production capacity. This patented method utilizes 2-halo-5-nitrobenzoates and substituted anilines, which are widely produced bulk chemicals with stable supply chains. The robustness of the reaction conditions also means that the process is less sensitive to minor variations in utility supply or operator error, leading to more consistent batch-to-batch performance. For supply chain heads, this reliability reduces the need for excessive safety stock and minimizes the risk of production stoppages due to quality deviations. The simplified workflow also shortens the overall cycle time, allowing for faster turnover and more responsive fulfillment of customer orders.
- Scalability and Environmental Compliance: As regulatory scrutiny on pharmaceutical manufacturing intensifies, the environmental footprint of a synthesis route becomes a critical factor in vendor selection. This process excels in sustainability metrics by avoiding the generation of heavy metal sludge and toxic gases. The absence of iron powder reduction eliminates a major waste stream that is increasingly difficult and expensive to dispose of in compliance with modern environmental regulations. Moreover, the one-pot nature of the cyclization step reduces the total volume of solvent required and minimizes the number of unit operations, leading to lower energy consumption. These factors collectively make the process highly scalable, enabling seamless transition from pilot plant trials to multi-ton commercial production without the need for significant re-engineering of waste treatment infrastructure.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and comparative examples provided in the patent documentation, offering a transparent view of the process capabilities and limitations. Understanding these nuances is essential for technical teams evaluating the feasibility of integrating this route into their existing manufacturing portfolios.
Q: How does this new synthesis route improve environmental compliance compared to traditional methods?
A: Unlike prior art methods that utilize iron powder reduction generating heavy metal sludge or CDI reagents releasing hydrogen sulfide gas, this patented process employs a Lewis acid catalyzed amidation and a clean cyclization step, significantly reducing hazardous waste generation and simplifying effluent treatment.
Q: What represents the critical quality attribute for this intermediate in downstream API production?
A: The liquid phase purity exceeding 99.6% is critical, as it minimizes the formation of isomeric impurities during the subsequent reduction and coupling steps required to produce Varlitinib, thereby ensuring consistent final API quality and reducing purification burdens.
Q: Is this manufacturing process scalable for commercial tonnage production?
A: Yes, the process utilizes common industrial solvents like toluene and DMF and avoids sensitive reagents requiring cryogenic conditions. The high yields (>90% in the cyclization step) and robust reaction conditions (up to 110°C) indicate excellent potential for scale-up from kilogram to multi-ton batches.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 6-Nitro-4-Substituted Amino Quinazoline Supplier
The technological breakthroughs detailed in patent CN110577520B underscore the immense potential for optimizing the supply chain of oncology intermediates. At NINGBO INNO PHARMCHEM, we recognize that translating such innovative chemistry into commercial reality requires more than just a patent; it demands deep process engineering expertise and a commitment to quality excellence. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive a consistent and reliable supply of critical materials. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of 6-nitro-4-substituted amino quinazoline delivered meets the exacting standards required for downstream API synthesis.
We invite forward-thinking pharmaceutical companies and procurement leaders to collaborate with us to leverage this advanced manufacturing capability. By partnering with our technical procurement team, you can access a Customized Cost-Saving Analysis tailored to your specific volume requirements and logistical needs. We encourage you to reach out today to request specific COA data and route feasibility assessments, allowing us to demonstrate how our optimized synthesis of Varlitinib intermediates can enhance your supply chain resilience and drive down your overall production costs.
