Advanced Photochemical Synthesis of Benzo-Bicyclo Octane Derivatives for Commercial Scale
The pharmaceutical and fine chemical industries are constantly seeking robust synthetic routes for complex bicyclic scaffolds that serve as critical intermediates for bioactive molecules. Patent CN107586272B introduces a groundbreaking methodology for the preparation of polysubstituted benzo-bicyclo [2.2.2] octane derivatives, which are highly valued for their potential as calcium ion channel blockers and tumor necrosis factor-alpha inhibitors. This technology represents a significant leap forward in organic synthesis by leveraging photochemical activation to construct the rigid bicyclo [2.2.2] carbon skeleton without the need for harsh conditions. The strategic importance of this patent lies in its ability to generate high-value pharmaceutical intermediates through a process that is inherently safer and more environmentally benign than traditional thermal or metal-catalyzed approaches. For R&D directors and procurement managers, understanding the implications of this metal-free pathway is essential for optimizing supply chains and reducing the environmental footprint of API manufacturing. The versatility of the substituents allowed in this reaction further expands its utility across various therapeutic areas, making it a cornerstone technology for modern medicinal chemistry programs.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the benzo-bicyclo [2.2.2] octane framework has relied heavily on Lewis acid catalysis or transition metal-mediated cycloaddition reactions, which present substantial drawbacks for large-scale manufacturing. These conventional methods often necessitate stringent anhydrous conditions, elevated temperatures, and the use of expensive and toxic metal catalysts that complicate the downstream purification process. The presence of residual metals in the final product is a critical quality attribute that requires extensive and costly removal steps, such as specialized scavenging or recrystallization, to meet regulatory standards for pharmaceutical ingredients. Furthermore, the thermal conditions required for traditional Diels-Alder reactions can lead to side reactions, polymerization, or decomposition of sensitive functional groups, resulting in lower overall yields and a more complex impurity profile. From a supply chain perspective, the reliance on specific metal catalysts introduces vulnerability regarding raw material availability and price volatility, while the generation of metal-containing waste streams poses significant environmental compliance challenges for production facilities.
The Novel Approach
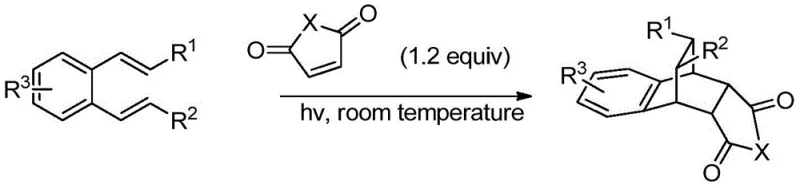
The novel approach detailed in the patent data utilizes a photochemical [4+2] cycloaddition strategy that fundamentally alters the energy landscape of the reaction, allowing it to proceed efficiently at room temperature under ultraviolet irradiation. This metal-free methodology eliminates the need for transition metal catalysts entirely, thereby removing the associated risks of metal contamination and the costly purification steps required to mitigate them. By operating under mild conditions in common organic solvents like acetonitrile or toluene, the process significantly reduces energy consumption and enhances operational safety, making it highly suitable for scale-up in standard chemical manufacturing plants. The simplicity of the work-up procedure, which involves basic rotary evaporation followed by silica gel chromatography, streamlines the production timeline and reduces the consumption of auxiliary materials. This shift from thermal to photochemical activation not only improves the sustainability profile of the synthesis but also offers a more predictable and controllable reaction environment, ensuring consistent product quality and batch-to-batch reproducibility which is paramount for commercial supply.
Mechanistic Insights into Photochemical [4+2] Cycloaddition
The core of this innovative synthesis lies in the photo-induced excitation of the o-phenylenediene derivative, which alters its electronic state to facilitate a concerted cycloaddition with the olefinic dienophile. Upon absorption of ultraviolet light, the diene system undergoes a transition to an excited state that lowers the activation energy barrier for the formation of the new carbon-carbon bonds, enabling the reaction to occur rapidly at ambient temperatures. This mechanism bypasses the high thermal energy requirements of ground-state reactions, preventing the thermal degradation of sensitive functional groups such as esters or amides that are often present in complex pharmaceutical intermediates. The stereoselectivity of the reaction is governed by the orbital symmetry constraints of the photochemical process, leading to the formation of the specific endo or exo bicyclic structures with high fidelity. Understanding this mechanistic pathway is crucial for process chemists as it allows for the fine-tuning of reaction parameters, such as wavelength and intensity of the UV source, to maximize conversion rates and minimize the formation of by-products. The absence of radical intermediates typically associated with photoredox catalysis further simplifies the impurity profile, ensuring a cleaner reaction mixture that is easier to purify.
Impurity control in this photochemical process is inherently superior due to the absence of metal catalysts which often promote side reactions such as oxidation or isomerization. The mild reaction conditions prevent the thermal decomposition of reactants and products, which is a common source of impurities in high-temperature synthesis routes. Additionally, the specificity of the photo-excitation ensures that only the desired chromophores are activated, reducing the likelihood of non-specific reactions with other functional groups present in the molecule. The purification strategy relies on standard silica gel chromatography, which is highly effective at separating the desired bicyclic product from any unreacted starting materials or minor photo-by-products. This robustness in impurity management translates directly to higher overall yields and reduced waste generation, aligning with the principles of green chemistry. For quality control teams, the predictable nature of the impurity profile simplifies the validation of analytical methods and ensures that the final product consistently meets the stringent purity specifications required for pharmaceutical applications.
How to Synthesize Polysubstituted Benzo-Bicyclo [2.2.2] Octane Efficiently
Implementing this synthesis route requires careful attention to the preparation of the reaction mixture and the optimization of the irradiation conditions to ensure maximum efficiency. The process begins with the dissolution of the o-phenylenediene derivative and the olefinic compound in a suitable organic solvent, followed by degassing to remove oxygen which can quench the excited states. The reaction mixture is then subjected to UV light for a specific duration, typically ranging from 4 to 10 hours, depending on the specific substrates and the intensity of the light source. Detailed standardized synthesis steps are provided in the guide below to ensure reproducibility and safety during scale-up operations. The simplicity of the protocol allows for easy adaptation to various flow chemistry setups or batch reactors, providing flexibility for manufacturing teams to optimize throughput based on their specific equipment capabilities.
- Dissolve o-phenylenediene derivative and olefin compound in organic solvent like acetonitrile at room temperature.
- Expose the mixture to ultraviolet light under nitrogen or air atmosphere until reaction completion.
- Purify the crude product via rotary evaporation and silica gel column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this photochemical synthesis route offers compelling advantages that directly address the cost and reliability concerns of procurement and supply chain managers. The elimination of transition metal catalysts results in significant cost savings by removing the need for expensive metal reagents and the specialized equipment required for their removal, such as filtration units or scavenger columns. This simplification of the manufacturing process reduces the overall production time and labor costs, allowing for faster turnaround times and improved responsiveness to market demand. The use of common solvents and ambient temperature conditions further lowers the energy footprint of the process, contributing to reduced utility costs and enhanced sustainability credentials which are increasingly important for corporate social responsibility goals. These factors combine to create a more resilient and cost-effective supply chain for high-purity pharmaceutical intermediates.
- Cost Reduction in Manufacturing: The removal of transition metal catalysts from the synthesis route eliminates the substantial costs associated with purchasing these precious metals and the subsequent purification steps required to meet residual metal limits. This reduction in material and processing costs allows for a more competitive pricing structure for the final intermediate without compromising on quality or purity standards. Furthermore, the simplified work-up procedure reduces the consumption of solvents and auxiliary materials, leading to additional savings in waste disposal and raw material procurement. The overall economic efficiency of this method makes it an attractive option for large-scale production where margin optimization is critical.
- Enhanced Supply Chain Reliability: By relying on readily available organic starting materials and avoiding specialized metal catalysts, the supply chain becomes less vulnerable to disruptions caused by raw material shortages or geopolitical issues affecting metal markets. The mild reaction conditions also reduce the risk of process deviations or safety incidents that could lead to production downtime, ensuring a more consistent and reliable supply of the intermediate. This stability is crucial for maintaining continuous manufacturing operations and meeting the strict delivery schedules required by downstream API manufacturers. The robustness of the process ensures that supply commitments can be met with high confidence.
- Scalability and Environmental Compliance: The photochemical nature of the reaction is highly amenable to scale-up using modern flow chemistry technologies or optimized batch reactors, allowing for seamless transition from laboratory to commercial production volumes. The absence of heavy metal waste streams simplifies environmental compliance and reduces the regulatory burden associated with waste treatment and disposal. This alignment with green chemistry principles not only mitigates environmental risk but also enhances the corporate image of the manufacturer as a sustainable partner. The ease of scaling ensures that production capacity can be expanded rapidly to meet growing market demand without significant capital investment in new infrastructure.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the photochemical synthesis of benzo-bicyclo octane derivatives, providing clarity for stakeholders evaluating this technology. These answers are derived directly from the patent data and practical experience with similar photochemical processes, ensuring accuracy and relevance for decision-makers. Understanding these details is essential for assessing the feasibility of integrating this route into existing manufacturing portfolios. The information provided here serves as a foundation for further technical discussions and feasibility studies.
Q: Does this synthesis method require transition metal catalysts?
A: No, the patented process operates without transition metal catalysis, utilizing UV light instead, which eliminates heavy metal residue concerns.
Q: What are the typical reaction conditions for this cycloaddition?
A: The reaction proceeds at room temperature and normal pressure under UV irradiation, ensuring mild and safe operating conditions.
Q: How is product purity ensured in this photochemical route?
A: High purity is achieved through simple silica gel column chromatography, avoiding complex purification steps associated with metal catalyst removal.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Benzo-Bicyclo Octane Derivative Supplier
NINGBO INNO PHARMCHEM stands at the forefront of translating innovative patent technologies like CN107586272B into commercial reality, offering unparalleled expertise in the scale-up of complex photochemical reactions. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial plant is seamless and efficient. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of benzo-bicyclo octane derivatives meets the highest international standards for pharmaceutical intermediates. Our commitment to technical excellence and quality assurance makes us the ideal partner for companies seeking to secure a reliable supply of these high-value building blocks.
We invite you to engage with our technical procurement team to discuss how this metal-free synthesis route can optimize your supply chain and reduce manufacturing costs. Request a Customized Cost-Saving Analysis to understand the specific economic benefits for your project, and ask for specific COA data and route feasibility assessments to validate the performance of our materials. Our dedicated support team is ready to provide the technical documentation and samples needed to accelerate your development timeline. Partnering with us ensures access to cutting-edge chemistry and a supply chain built on reliability and trust.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
